The Heart of Regulatory Operations
Replace Fragmented Tools With a Unified Regulatory Platform
Rimsys is the heart of regulatory operations - a unified regulatory management software platform purpose-built for MedTech. Rather than digitizing isolated tasks, Rimsys connects regulatory intelligence, product data, approvals, submissions, UDI, and change management into a living, continuously aligned system.
Proven Across Global Regulatory Operations
Six of the world’s top 12 medical device manufacturers rely on Rimsys to manage and streamline global regulatory operations
Product Philosophy
What You Get on Day One: Enterprise-Grade Foundation, Human-Centered Design
“Enterprise” often implies slow, rigid, and difficult to adopt. Rimsys proves otherwise.
Built specifically for MedTech, the platform combines:

As portfolios expand and global markets multiply, most organizations rely on:
Enterprise Architecture
- A structured, product-centric data model
- Robust APIs and BI integrations
- Event-driven architecture
- Single-tenant environments
- SOC 2 and ISO 27001 certification


As portfolios expand and global markets multiply, most organizations rely on:
User Experience
- An intuitive, modern interface
- Logical module design with optional AI power
- High adoption across RA teams
- Unlimited internal and external user licensing

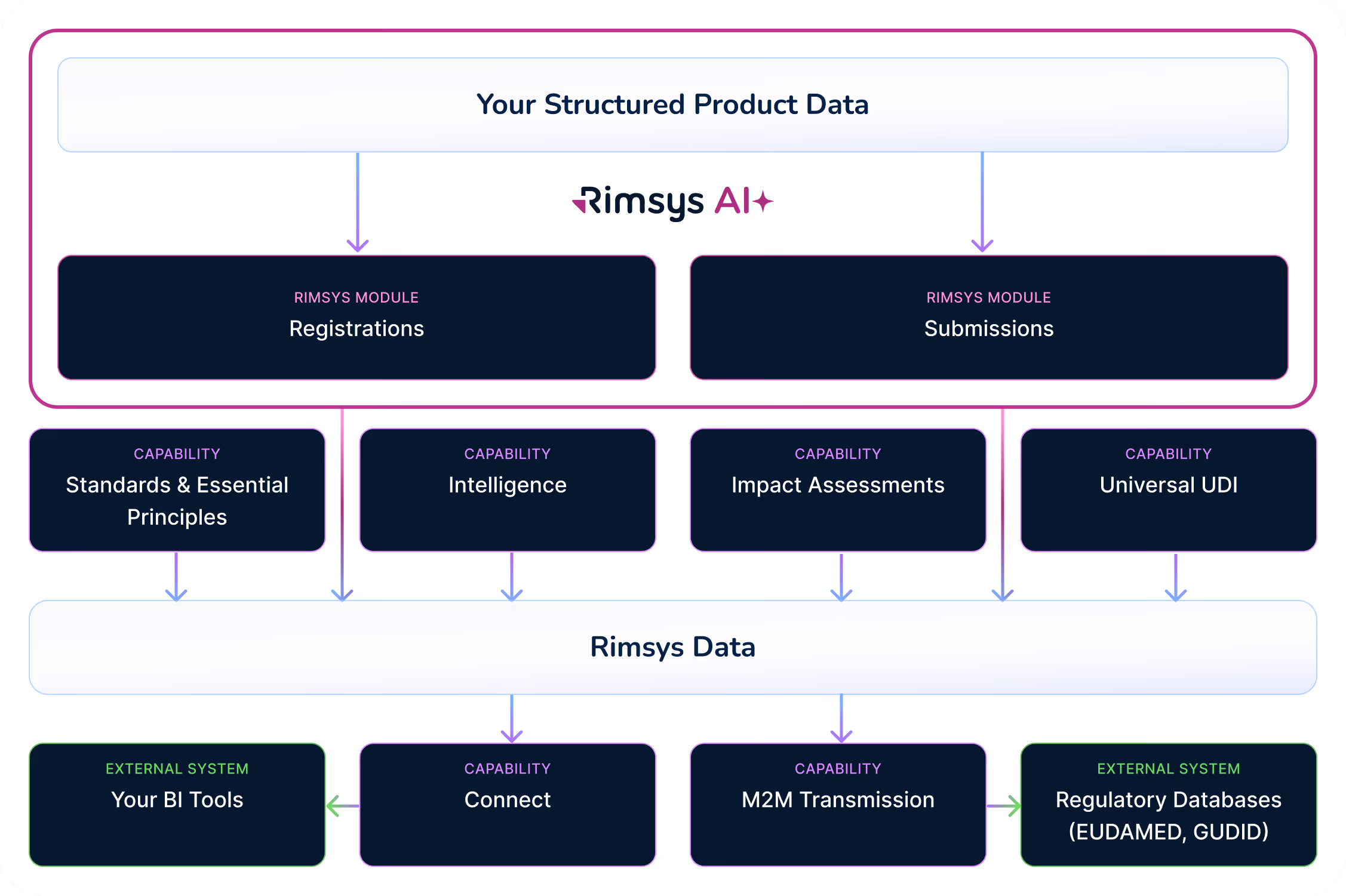
Platform Architecture
One Platform, Clearly Connected
At a glance, the Rimsys platform is simple to understand:

Modules
Drive key workflows

Capabilities
Extend value across module

Services
Reduce implementation risk

Integrations
Connect regulatory operations to the broader enterprise ecosystem
APIs and event-driven BI integrations connect Rimsys to ERP, PLM, CRM, and analytics systems—so regulatory intelligence informs commercial and operational decisions across the organization.
It is connected regulatory operations and the heart of your compliant growth.
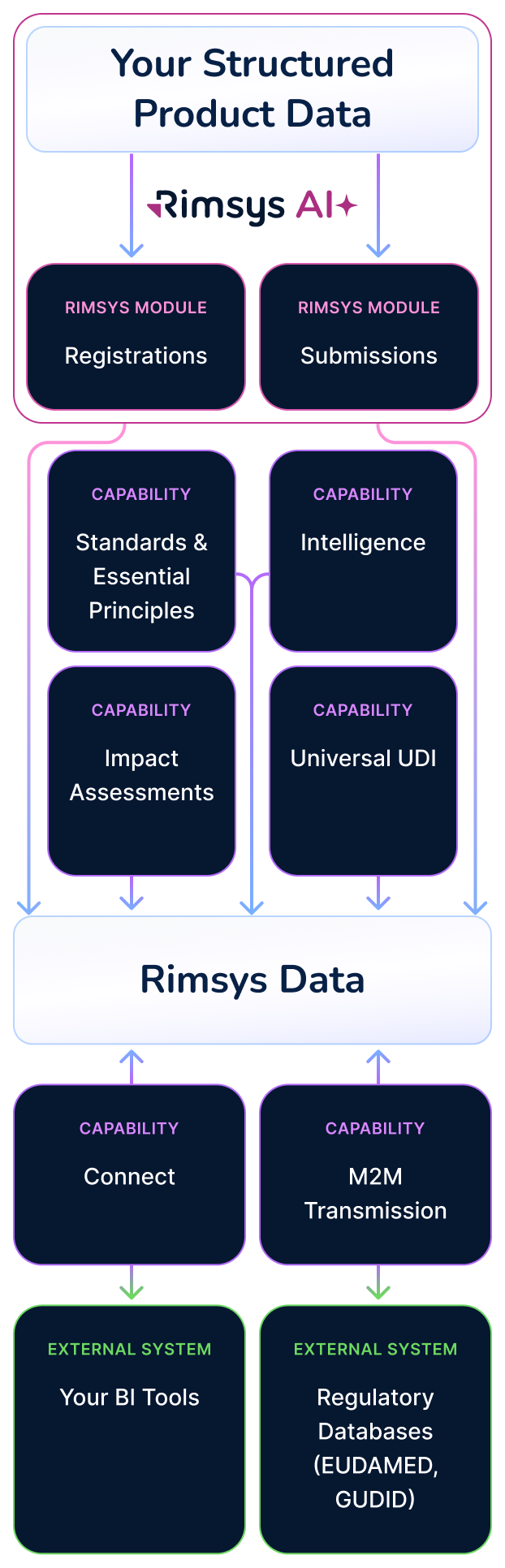
Core Modules
The Two Pillars of the Rimsys Platform
Registrations
Manage global registrations, approvals, renewals, and lifecycle tracking in one harmonized source of truth.
- Global registrations and approvals
- CE marks and dual registrations (e.g., ANVISA + INMETRO)
- Country-specific risk classifications
- Renewal timelines and reminders
- Real-time selling status visibility
Regulatory and commercial teams gain instant confidence in where products can be sold, renewed, or expanded.

Submissions
Plan, author, track, and execute submissions within a structured, collaborative workflow.
- Real-time co-authoring and embedded Word-compatible editing
- Linked product and country data
- Milestone tracking and task management
- AI-assisted submission preparation
- Structured data reuse across markets with linked product and country data.
From content preparation to final approval, accelerate submissions and remain aligned with your regulatory data.
Use Cases
Capabilities That Eliminate Manual Work
Rimsys focuses on jobs to be done-not feature lists.
Plan expansion, align stakeholders, and generate new registrations from structured product data.
Best for:
Regulatory leaders and growth-focused executives.
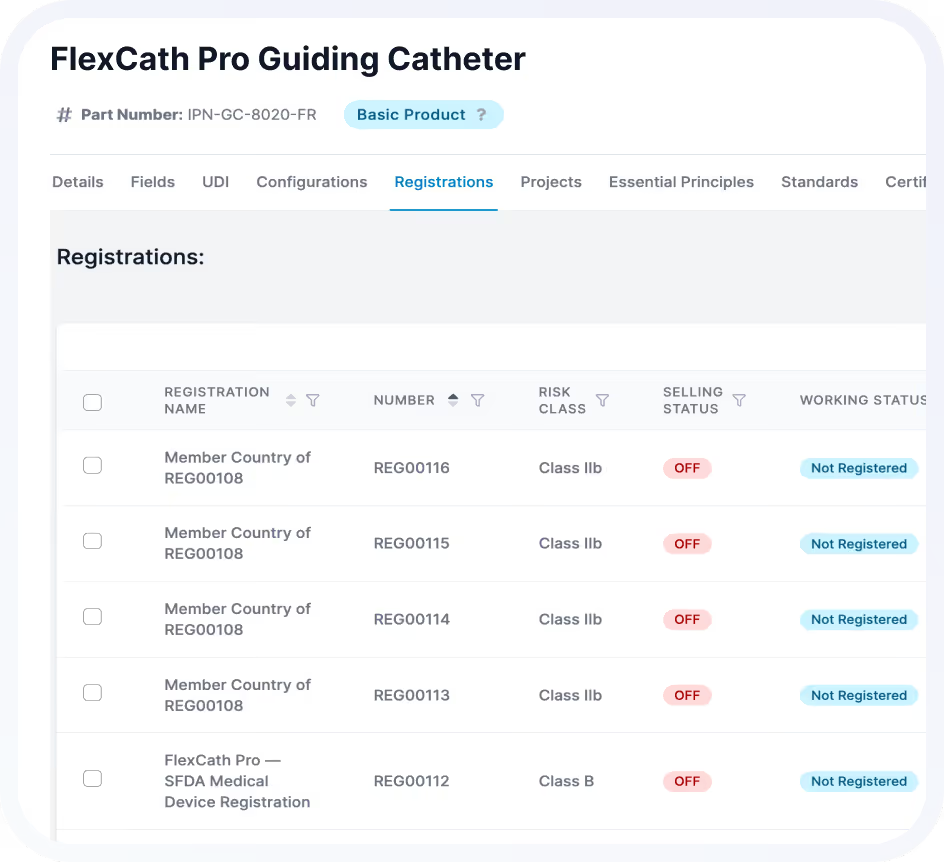
Centralize submission preparation, documentation, and review to reduce back-and-forth and eliminate data silos.
Best for:
Regulatory Affairs teams managing heavy submission volume.

Automated reminders and lifecycle tracking keep approvals current across 100+ markets.
Best for:
Global registration managers.

Assign tasks, conduct structured change assessments, and collaborate across teams when product or regulatory changes occur.
Best for:
Change control and cross-functional stakeholders.

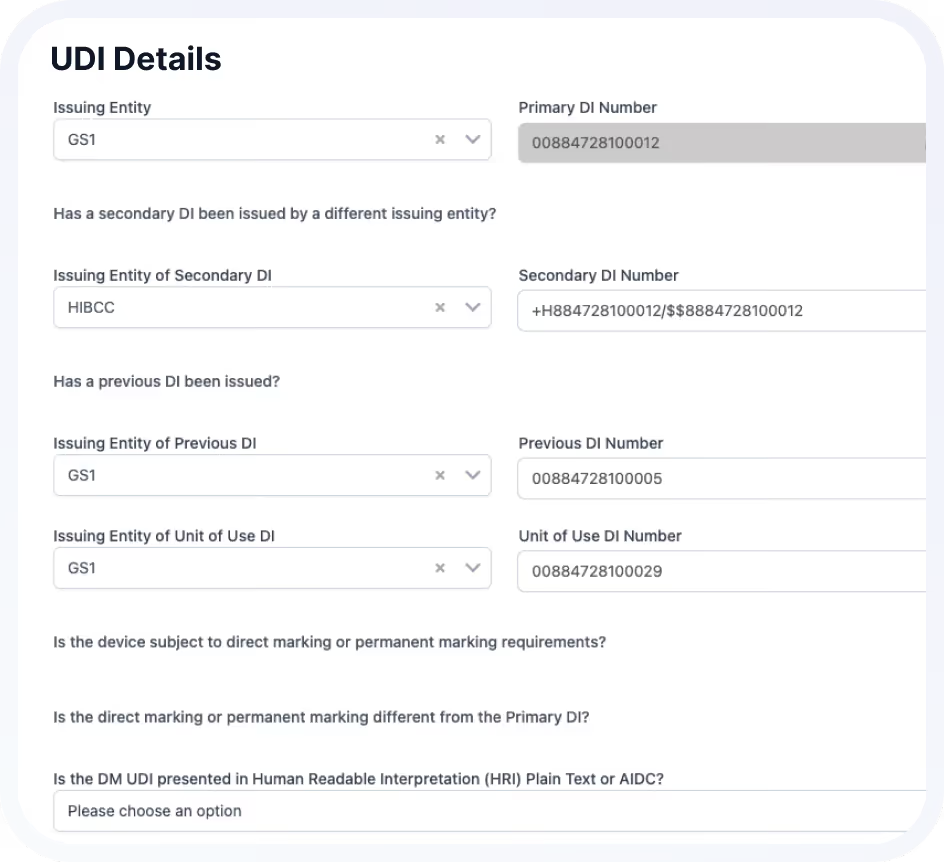
Native Universal UDI functionality supports structured global compliance, including EUDAMED requirements
Best for:
Teams managing complex device portfolios.

Accelerate Regulatory Work
AI Designed for Real Regulatory Workflows
AI in regulatory operations must be trustworthy, explainable, and controlled.

Rimsys AI is embedded directly within regulatory workflows and built on MedTech-specific data structures. It enhances human judgement—it does not replace it.
AI is available as a configuration option for organizations ready to accelerate regulatory work.
Accelerators Include:

Faster submission preparation through automated content support

Earlier identification of regulatory impact through automated content support

Intelligent tracking of evolving global requirements

Reduced time spent on repetitive regulatory tasks
Built for Every Stakeholder
One platform for everyone
RegOps/Regulatory Affairs
Emphasize visibility, fewer manual handoffs, audit readiness, and confidence in regulatory status.

Digital Transformation Leaders
Centralize regulatory data, standardize workflows, and measure efficiency across the organization
.avif)
IT
Leverage a structured, product-centric data model with robust APIs, developer support, AI-assisted code generation, BI integration, and single-tenant security architecture
.avif)
Start Building Connected Regulatory Operations
Regulatory complexity is not slowing down.
The organizations that win, treat regulatory operations as a strategic growth engine-not an administrative burden.

Rimsys transforms regulatory operations into the heart of compliant growth-bringing speed, visibility, and confidence to global expansion.
FAQs
A unified RIM platform connects registrations, submissions, product data, and change management into one structured operational system. Traditional RIM systems often digitize isolated tasks and serve as a system of record instead of optimizing workflows. Rimsys also uniquely includes UDI to create a continuously connected regulatory operation.
By centralizing global regulatory intelligence, approvals, risk classifications, and selling status, Rimsys enables faster, more confident market entry and expansion decisions.
Structured data models, governed workflows, and real-time traceability ensure compliance while eliminating manual bottlenecks.
Rimsys AI is explainable, context-aware, and embedded within regulatory workflows. It proposes and analyzes—your team reviews and approves
Yes. Robust APIs and event-driven BI integrations connect regulatory intelligence to enterprise dashboards and downstream systems
Rimsys uses a proven, high-touch implementation approach guided by a RIM & AI Maturity Model - helping organizations modernize in structured, incremental phases