The Regulatory Operations Platform for MedTech
Replace Fragmented Tools With a Unified Regulatory Platform
- Enterprise-ready yet intuitive to use, Rimsys is trusted by 6 of the top 12 global MedTech manufacturers to accelerate time to market, reduce compliance risk, and scale regulatory operations worldwide

Operational Impact
The Day-to-Day Impact of a Unified Platform
A unified platform doesn’t just improve reporting—it changes how regulatory teams operate every day.

Faster Regulatory Execution
When regulatory data is centralized and connected, teams can validate market release status in minutes—not chase approvals across emails and spreadsheets.
- Minutes instead of weeks for regulatory release authorization
- Faster submission assembly and approval cycles
- Shortened time to new market entry
When regulatory data is structured and connected, execution speeds up without increasing risk.

Reduced Manual Tracking
Eliminate spreadsheets, duplicate data entry, and disconnected trackers.
- Access critical regulatory information in seconds rather than hours
- More than 90% reduction in regional regulatory reporting time
- Automated reminders for renewals and expirations
The result: fewer administrative tasks and more time for strategic work.

Clear Visibility Across Products and Markets
Know exactly where every product stands—globally.
- Real-time selling status visibility by country
- Unified view of registrations, submissions, and UDI
- Portfolio-level dashboards for leadership and cross-functional teams
Regulatory data becomes transparent, auditable, and actionable.

Lower Compliance Risk
Governed workflows and precision data reduce the risk of missed renewals, inconsistent filings, or outdated information.
- Structured, product-centric data model
- Audit-ready traceability across approvals and changes
- SOC 2 and ISO 27001 certified environments
Compliance is built into the system—not layered on afterward.

Reduced Manual Tracking
Eliminate spreadsheets, duplicate data entry, and disconnected trackers.
- Access critical regulatory information in seconds rather than hours
- More than 90% reduction in regional regulatory reporting time
- Automated reminders for renewals and expirations
The result: fewer administrative tasks and more time for strategic work.
Core Capabilities
Core Capabilities of the Rimsys Platform
A unified platform doesn’t just improve reporting—it changes how regulatory teams operate every day.
Product Registrations
Manage global registrations, approvals, renewals, and lifecycle tracking in one harmonized source of truth.
Country-specific risk classifications
Dual registration support (e.g., ANVISA + INMETRO)
Expiration tracking and renewal alerts
Selling status visibility by market
Registrations are no longer static records—they are active, connected operational assets.

Submissions Management
Plan, author, track, and execute submissions within a structured, collaborative workflow.
Real-time co-authoring and embedded document tools
Linked product and country data
Milestone tracking and task management
AI-assisted submission preparation
From content preparation to final approval, submissions remain aligned with your regulatory data.

UDI and Device Identification
Native Universal UDI capabilities support EUDAMED, GUDID, and global UDI compliance.
Structured UDI data model
Alignment with registrations and submissions
Controlled validation logic
Audit-ready traceability
Manage UDI directly inside your RIM—no external database dependency required.

Standards and Essential Principles
Track standards applicability, essential principles, and regulatory requirements across markets.
Link standards directly to products and registrations
Maintain traceability for audit
Monitor regulatory alignment as products evolve
Standards management becomes proactive rather than reactive.

Regulatory Intelligence
Monitor, assess, and operationalize regulatory changes.
Track new regulations, guidances, and laws
AI-powered intelligence screening and triage
Market entrance requirements by country and risk class
Intelligence is embedded into workflows—not stored in isolation.

Impact Assessment
Evaluate the operational effect of product or regulatory changes.
Configurable impact survey templates
Structured change assessment projects
Task assignment and cross-functional collaboration
Link assessments to countries, registrations, and products
When change occurs, your organization responds with clarity and control.
Real-world Scenarios
Supporting Regulatory Work Across the Lifecycle
Rimsys supports the real-world scenarios MedTech companies face every day.
Managing Large Product Portfolios
Entering New Markets
Responding to Regulatory Change
Preparing Submissions and Audits
Reducing Reliance on Spreadsheets
Managing Large Product Portfolios
Handle complex product structures—kits, bundles, and variants—within a structured data model that scales. Bulk actions, governed approvals, and event-driven BI integrations allow enterprise teams to manage thousands of SKUs without losing control.
Entering New Markets
Quickly determine market entrance requirements by risk class and country. Align strategy, create registrations, and initiate submissions—all within one connected workflow.

Responding to Regulatory Change
When a new regulation emerges, regulatory intelligence connects directly to impact assessments, tasks, and submissions. Teams identify affected products, assign ownership, and track progress in one system.Change becomes manageable, not disruptive.

Preparing Submissions and Audits
Because product data, approvals, and standards are already structured and linked, teams prepare submissions and audits with greater speed and accuracy. Traceability is built in, not reconstructed manually.
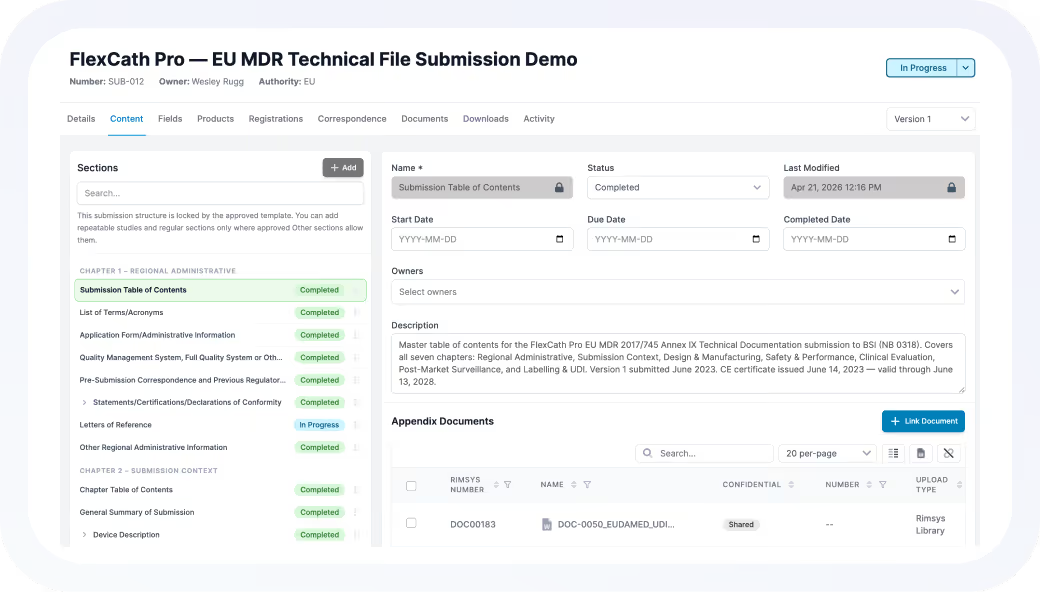
Reducing Reliance on Spreadsheets
Many customers adopt Rimsys to eliminate spreadsheet-based tracking. By centralizing registrations, submissions, and UDI, they gain consistency, reduce errors, and improve team adoption.
Platform Architecture
A Closer Look at the Rimsys Platform
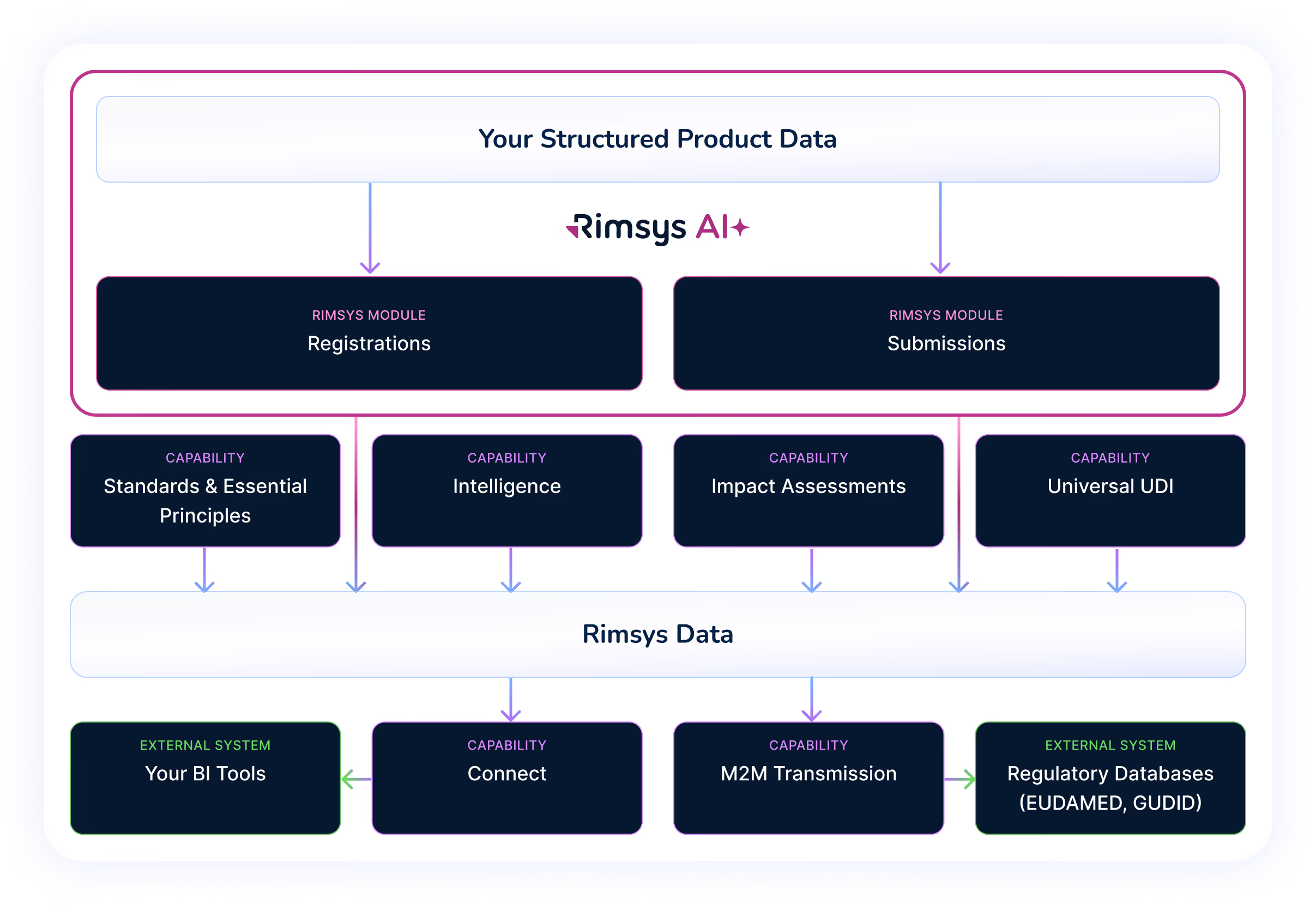
At its core, Rimsys is built on a structured, product-centric data model that begins at implementation. Every module—registrations, submissions, UDI, standards, intelligence, impact assessments—connects back to the same data foundation.
Key platform differentiators include:
Purpose-built for MedTech regulatory complexity
AI-powered workflows embedded inside operational processes
Event-driven BI integration for enterprise dashboards
Robust APIs with developer support portal (including AI-assisted code generation)
Single-tenant environments for enhanced security and control

Enterprise Infrastructure
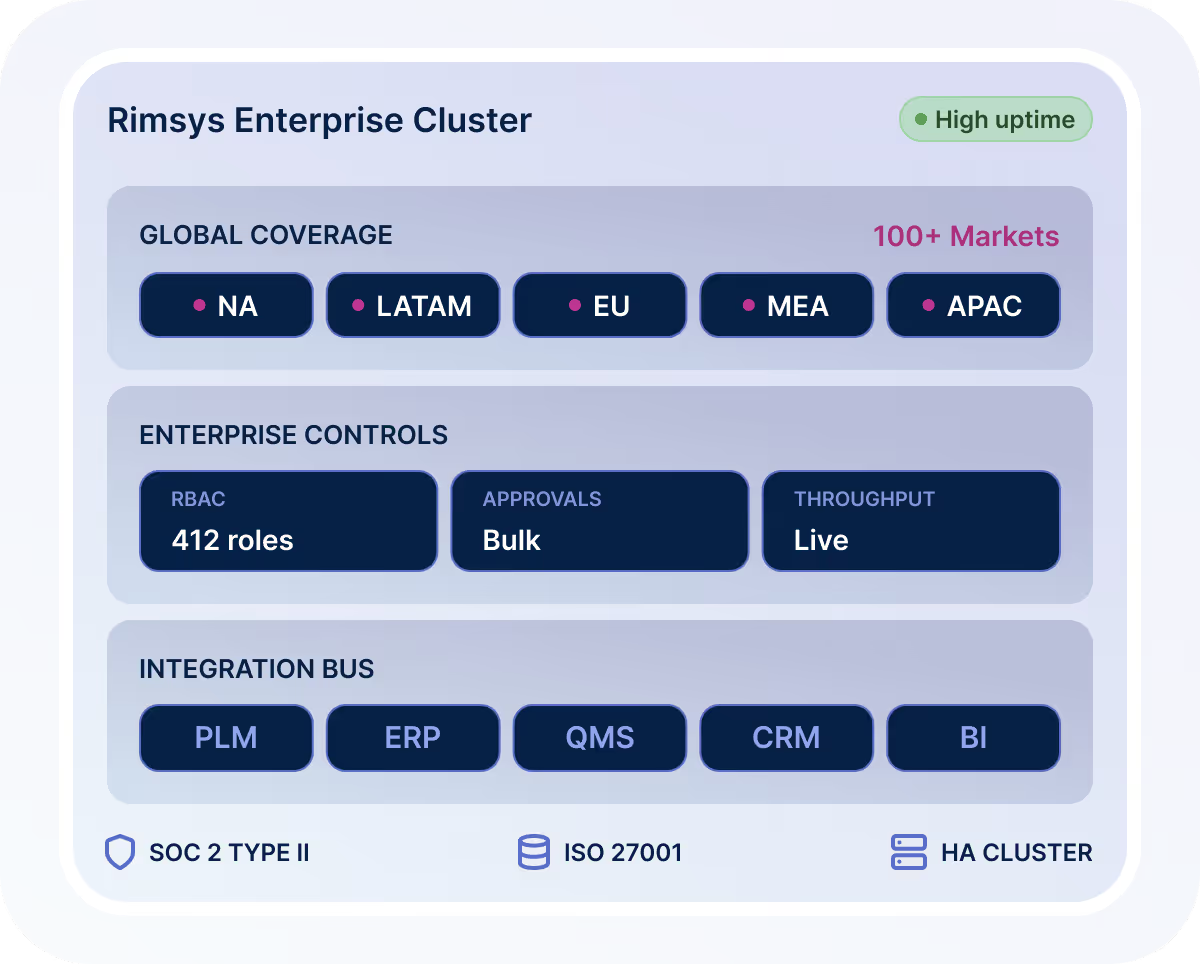
Enterprise-Ready Regulatory Management Software
Enterprise MedTech companies require more than usability—they require scale, governance, and integration.
Rimsys delivers:

Support for large global portfolios across 100+ markets

Role-based access controls and governed approval workflows

Bulk approvals and enterprise-scale processing

SOC 2 and ISO 27001 certification

High-uptime, enterprise-grade infrastructure
Proven Across Global Regulatory Operations
Six of the world’s top 12 medical device manufacturers rely on Rimsys to manage and streamline global regulatory operations



Experience Unified Regulatory Management
Regulatory complexity is not slowing down. Portfolios are expanding. Markets are evolving. Requirements are intensifying.

Rimsys transforms regulatory operations into the heart of compliant growth—keeping intelligence, product data, approvals, and change continuously connected so your organization can scale with confidence.
FAQs
The Rimsys RIM (Regulatory Information Management) platform connects regulatory intelligence, product data, submissions, registrations, UDI, and change management within a single structured data model.
Traditional RIM systems often digitize isolated tasks, such as submission tracking or document storage, without connecting the underlying data. A unified platform does more than store data, it enables regulatory teams to manage their work efficiently and keeps information synchronized across modules so that changes to a product, market, or regulation are reflected everywhere. The result is greater visibility, fewer inconsistencies, and stronger operational control.
Transforming regulatory operations means elevating regulatory from a reactive function into a strategic enabler of expansion. When intelligence, approvals, selling status, and change management are continuously connected, regulatory becomes a source of enterprise visibility and confidence.
Instead of slowing growth, regulatory becomes the foundation that enables faster market entry, stronger compliance, and scalable global execution.
Global expansion requires speed, clarity, and coordination. A modern RIM platform centralizes market entrance requirements, risk classifications, registrations, approvals, and selling status across countries.
With a single source of truth, organizations can evaluate opportunities more quickly, align cross-functional teams, and maintain real-time oversight of product availability worldwide. This allows companies to expand into new markets with confidence and control.
Regulatory risk increases when teams rely on spreadsheets, siloed systems, and manual tracking. Rimsys reduces risk through structured, product-centric data, governed workflows, automated renewal alerts, and full traceability across products and markets.
At the same time, intuitive design and AI-assisted workflows streamline repetitive tasks and reduce administrative burden. Compliance and speed are not trade-offs—they are outcomes of connected, well-governed operations.
AI in regulatory must enhance human expertise, not replace it. Rimsys AI is embedded directly inside regulatory workflows to assist with submission preparation, regulatory intelligence monitoring, and impact analysis.
It is secure, explainable, and context-aware. AI proposes and analyzes, while your team reviews and approves. AI functionality is configurable and optional, allowing organizations to adopt it at their own pace while maintaining full ownership of regulatory decisions.
Yes. Enterprise regulatory operations depend on connected systems. Rimsys provides robust APIs and event-driven integrations to connect with ERP, PLM, QMS, CRM, and business intelligence platforms.
This ensures regulatory approvals, product data, and selling status flow securely across the enterprise, improving cross-functional alignment and executive visibility.
Implementation timelines vary based on portfolio complexity, data maturity, and integration needs. Rimsys follows a structured, high-touch implementation approach guided by MedTech regulatory expertise.
Using our RIM & AI Maturity Model, organizations assess their current state and define a practical modernization roadmap before configuration begins. The focus is not just on deploying software, but on advancing regulatory maturity, improving data governance, and enabling scalable global operations.

