Rimsys Capabilities: Modern Regulatory Management
End-To-End Regulatory Information Management Built for MedTech Teams
Rimsys transforms regulatory operations into a connected execution system that drives compliant growth. By unifying regulatory intelligence, product data, submissions, and change management, teams gain real-time visibility, faster execution, and confidence across global markets.

Proven Results
Proven Results Across Global Regulatory Operations
90+
LESS MANUAL WORK
90%+ reduction in manual reporting time
Minutes
NOT WEEKS
Weeks to minutes for selling status visibility
98%
FASTER REGULATORY REPORTING
98% reduction in regulatory reporting time
100+
COUNTRIES
Thousands of SKUs managed across 100+ countries
Unified Execution
One System That Owns the Work
Rimsys is not a collection of tools. It is a single system where regulatory work happens.


All capabilities operate on a shared data model, ensuring that submissions, registrations, UDI, and regulatory intelligence remain continuously aligned.

Services support implementation and optimization, but execution stays inside your system. Your data, workflows, and regulatory knowledge remain centralized and controlled.

The result is consistent execution, audit readiness, and a foundation that scales with your organization.
Explore Our Platform
Capability Set
Explore the Full Rimsys Capability Set
Product Registrations
Submissions Management
UDI Management & M2M Trading
Standards & Essential Principles
Regulatory Intelligence
Regulatory Impact Assessment
New Market Entry
Connect (Change Data Capture & Integrations)
Product Registrations
Centralize global product registrations and maintain accurate selling status across markets.

Submissions Management
Plan, author, track, and publish regulatory submissions with structured workflows.
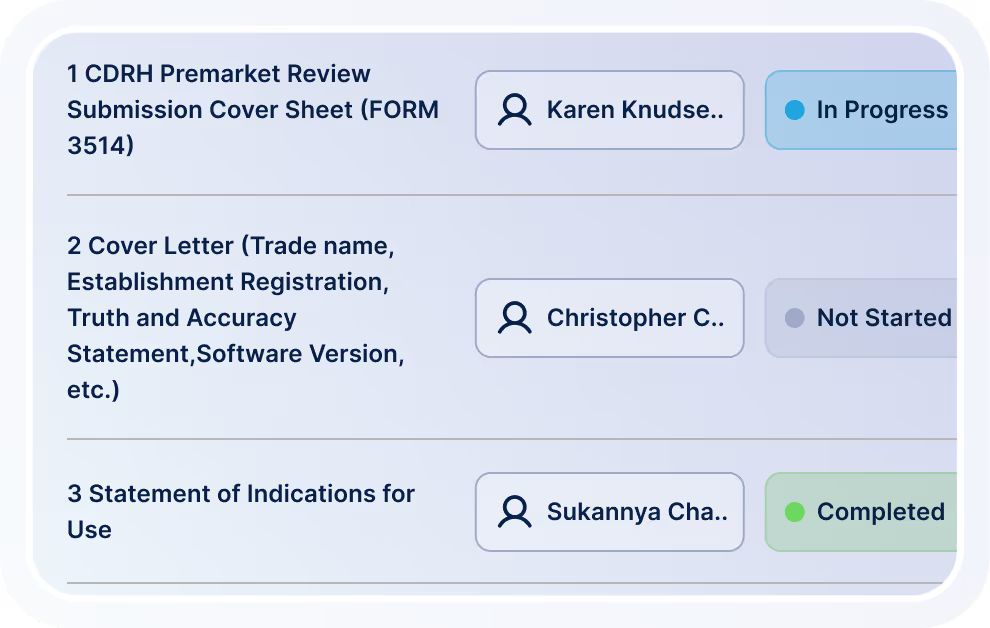
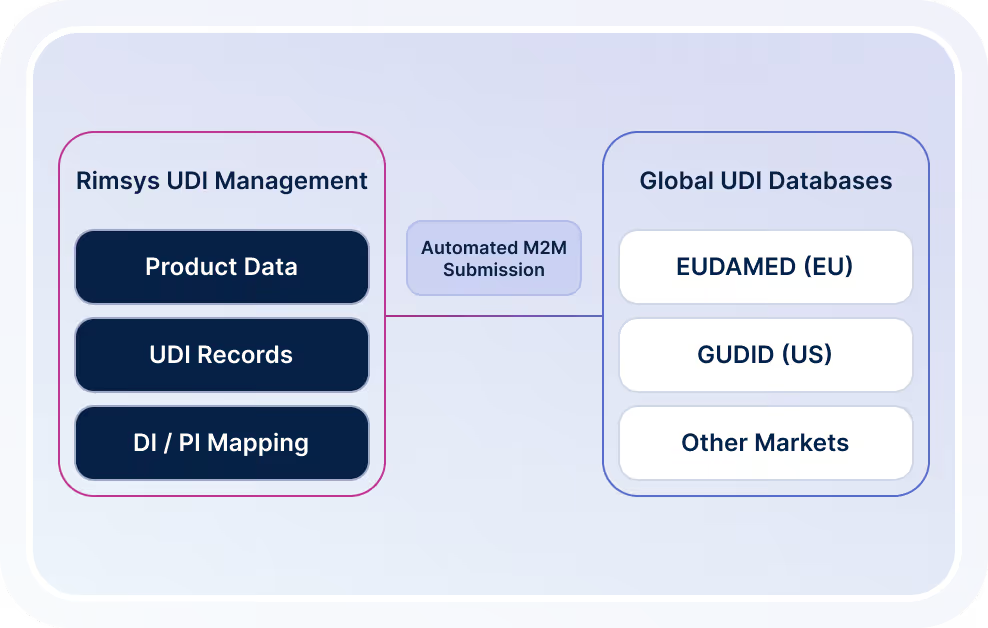
UDI Management & M2M Trading
Manage global UDI compliance and device identification requirements in one system.
Standards & Essential Principles
Track medical device standards and GSPR with automated monitoring and product-level links.

Regulatory Intelligence
Monitor global regulatory updates and link changes to impacted products and markets.
.avif)
Regulatory Impact Assessment
Assess the operational impact of regulatory change across submissions and portfolios.

New Market Entry
Coordinate regulatory requirements, submissions, and registrations to support expansion.
.avif)
Connect (Change Data Capture & Integrations)
Integrate with ERP, PLM, QMS, CRM, and BI systems for continuous regulatory data alignment.
.avif)
Built For Leaders
Designed for Global MedTech Regulatory Leaders
RegOps/Regulatory Affairs
Emphasize visibility, fewer manual handoffs, audit readiness, and confidence in regulatory status.

Digital Transformation & Operational Excellence
Centralize structured regulatory data. Streamline workflows. Measure efficiency gains and reduce administrative burden while enabling global growth.
.avif)
IT
Leverage a structured data model, role-based access control, APIs, and a secure single-tenant architecture
.avif)
Rimsys connects regulatory operations to the broader enterprise without compromising control or compliance.
Outcomes
What Customers Achieve With Rimsys
Organizations using Rimsys move from reactive coordination to controlled execution.
Faster approvals
Reduced compliance risk
Continuous visibility
Audit readiness
Stronger team alignment
They reduce administrative overhead, accelerate submission timelines, and maintain continuous visibility across global markets. Regulatory teams operate with precision, while leadership gains trusted insight into product readiness and market access.

The impact is measurable. Faster approvals, reduced compliance risk, and stronger alignment between regulatory and commercial teams.
Bring Your Regulatory Operations Into One System

Rimsys becomes the operational foundation for regulatory work. It connects data, workflows, and decisions so nothing falls out of sync.
FAQs
Regulatory Information Management software centralizes regulatory data, submissions, registrations, and compliance activities into a single system.
It provides visibility, structure, and control across regulatory workflows, helping teams manage global requirements, reduce risk, and accelerate time to market.
A structured data model, real-time visibility, system integrations, and the ability to execute regulatory work inside the platform.
All capabilities share a unified data foundation, ensuring that regulatory intelligence, submissions, registrations, and impact assessments remain continuously aligned.
Legacy systems act as static systems of record. Rimsys operates as an execution platform that keeps regulatory work moving and connected.
Yes. Rimsys supports complex product portfolios and global operations, enabling organizations to scale without increasing manual effort.
Through structured workflows, real-time data access, AI-assisted processes, and integrations with enterprise systems, Rimsys enables teams to operate efficiently at scale.

