Partnering with Industry on Regulatory Compliance
System-based implementation and ongoing enablement that transforms global regulatory compliance into a repeatable operational capability.
Global compliance is not a one-time effort. It’s an operational system that must evolve as your products, markets, and regulations evolve. Global compliance is not a one-time effort. It’s an operational system that must evolve as your products, markets, and regulations evolve.

The Challenge
Global Compliance Is Not A Project - It’s Infrastructure
As MedTech companies expand globally, regulatory complexity multiplies.
New markets introduce new classifications, evolving standards, UDI requirements, language variations, and country-specific nuances. Product portfolios grow. Regulatory change accelerates.
Spreadsheets and disconnected systems cannot scale with that complexity.
Sustainable global compliance requires:

Structured, product-centric data

Governed workflows

Continuous regulatory intelligence

Real-time operational visibility
Compliance must function as infrastructure -
not memory, not email chains, and not individual heroics.
Platform Delivery
What You Get With Rimsys
Structured Platform Implementation
Rimsys implementation is designed to build a regulatory foundation that reflects how your organization truly operates.
Our team works with you to establish:
- Compliance maturity assessment
- Data migration and structured system configuration
- Regional regulatory framework mapping
- Workflow design aligned to your operating model
- Role-based governance and permissions setup
Unlike generic system deployments, Rimsys begins with a structured, product-centric data model that supports enterprise-scale regulatory complexity.
The result: your compliance system is configured intentionally — not retrofitted later.
Implementation ensures the platform becomes the system of record for global regulatory operations.
Embedded Regulatory Operations
Once implemented, compliance lives inside the platform — not across disconnected files and inboxes.
Rimsys centralizes:
- Global product registrations and approvals
- Submission lifecycle tracking
- UDI management across regions
- Standards and GSPR alignment
- Regulatory intelligence monitoring
- Impact assessments and change workflows
By continuously linking regulatory intelligence, submissions, product changes, and impact assessments, Rimsys creates a connected operational foundation.
This structured visibility delivers:
- Faster release authorization
- Reduced regional reporting time
- Immediate access to selling status and approvals
Compliance becomes embedded in daily execution — not dependent on individual tracking.


Ongoing Customer Success & Platform Optimization
Regulatory environments change. Your system should evolve with them.
Rimsys provides:
- Dedicated customer success partnership
- Adoption monitoring and optimization
- Continuous workflow refinement
- Support for expansion into new markets
- Release training and feature enablement
We don’t disappear after go-live. We ensure your compliance infrastructure matures as regulations, products, and markets evolve.
Our Approach
Built for Infrastructure - Not Advisory
Rimsys is regulatory management software.
We are not a consulting firm.

Rimsys does:
Enterprise Architecture
- Provide structured regulatory management software
- Implement governed, scalable workflows
- Embed regulatory intelligence into daily operations
- Strengthen internal regulatory teams
- Support long-term platform success

Rimsys does not:
Enterprise Architecture
- Provide standalone regulatory advice
- Replace your internal regulatory team
- Deliver bespoke consulting engagements
Platform Capabilities
How The Platform Powers Global Compliance
Rimsys serves as a unified regulatory backbone across markets and product portfolios.
Unified RIM Database
A structured, product-centric data model connects registrations, submissions, intelligence, UDI, and change control in one system.
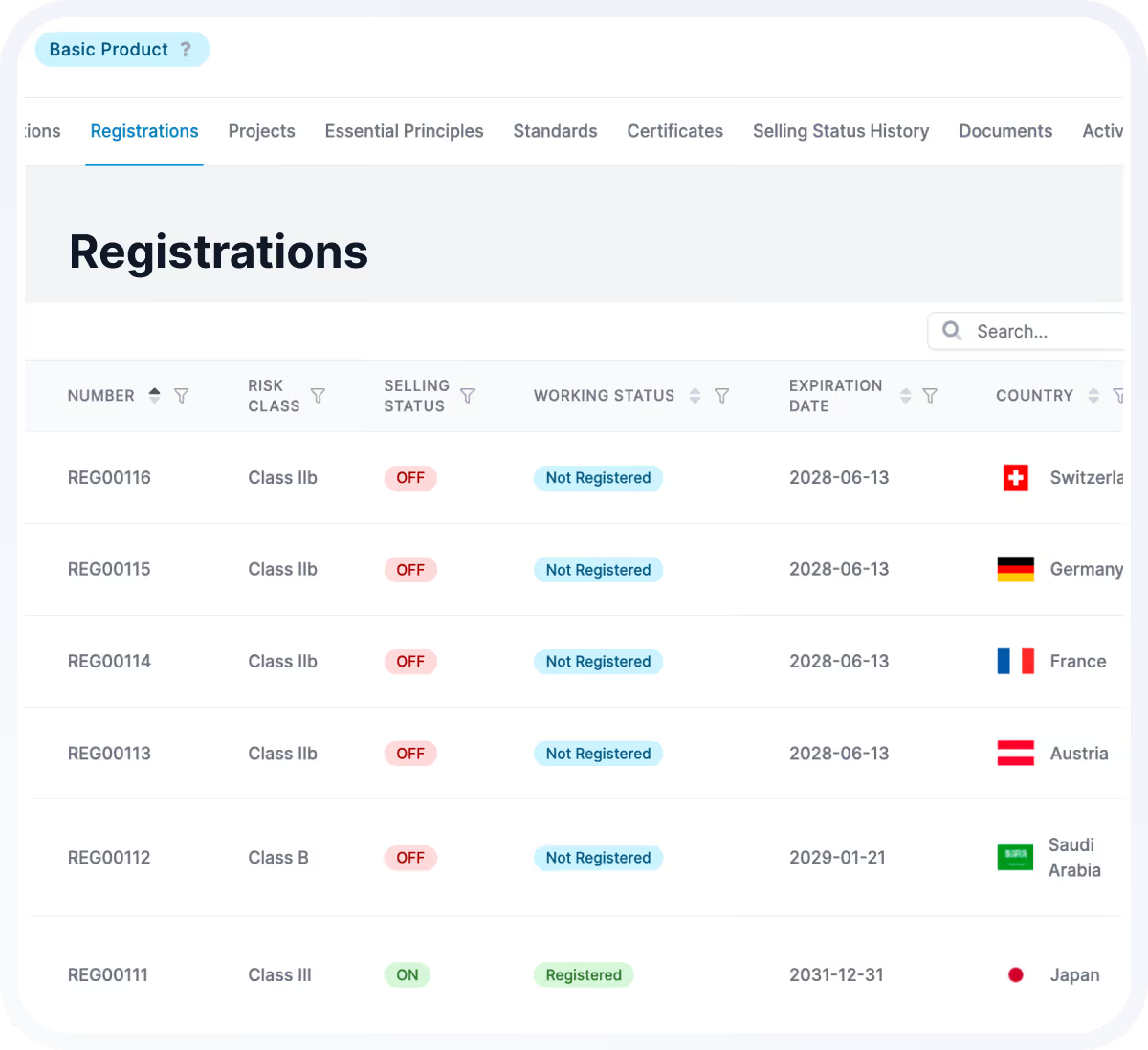
Registration & Submission Management
Manage global registrations and approvals, including complex structures such as kits, variants, bundles, and dual registrations.
Regulatory Intelligence Hub
Monitor changing regulations and understand impact directly within your workflows.
UDI & Market-to-Market Capabilities
Native UDI management supports global compliance and regional database requirements.
Audit-Ready Reporting
Governed workflows and traceable data ensure precision, transparency, and inspection readiness.
Enterprise Integration
Robust APIs and event-driven BI integrations connect regulatory data to enterprise dashboards and cross-functional systems.
Full Lifecycle
The Compliance Lifecycle Inside Rimsys
Rimsys supports the full regulatory lifecycle inside a single connected system:

Market Entry Planning
Evaluate new markets with structured intelligence and product context.
Submission Preparation
Centralize documentation, workflows, and collaboration.
Registration Tracking
Maintain global approval status with full visibility across regions.
UDI Management
Govern identifiers and submissions within the same operational system.
Post-Market Surveillance
Monitor compliance status across active markets.
Regulatory Change Monitoring
Track regulatory updates and link them directly to impacted products and markets.
Impact Assessment & Change Control
Assign tasks, manage cross-functional collaboration, and maintain traceable decisions.
Audit & Inspection Readiness
Deliver governed, reportable, and defensible regulatory data at any time.
Target Audience
Who this is Designed for
Global Regulatory Affairs Leaders
Gain enterprise-wide visibility and transform regulatory from reactive coordination into strategic enablement.

Regulatory Operations Teams
Replace spreadsheets and manual tracking with governed workflows and centralized execution.

MedTech Companies Expanding Internationally
Enter and scale in global markets with structured intelligence and operational confidence.

Organizations Replacing Fragmented Systems
Consolidate disconnected tools into a single source of truth.

Teams Preparing for Audit, Investment, or Transaction
Demonstrate audit readiness, traceability, and operational maturity.

Start Building Predictable Global Compliance
Global regulatory management should not depend on manual effort, tribal knowledge, or disconnected systems.

Replace fragmented processes with structured compliance infrastructure. Operationalize global regulatory management. Build a system - not dependency.
FAQs
It is a structured system that centralizes product registrations, submissions, regulatory intelligence, UDI management, and change control into one governed operational platform.
We assess your regulatory maturity, configure a structured data model, map workflows to your operating model, migrate data, and establish governance — ensuring long-term adoption.
No. Rimsys provides regulatory management software and structured implementation. We strengthen your internal regulatory capability rather than replace it.
We give you ideas to drive adoption, refine workflows, support expansion into new markets, and ensure your system evolves alongside regulatory changes.
Yes. The structured data model, enterprise-grade APIs, and secure architecture are built for global scale.
Governed workflows, traceable data, regulatory intelligence integration, and audit-ready reporting reduce manual error and increase visibility.
Rimsys continues to evolve with your organization through ongoing customer success, optimization, and feature enablement — ensuring compliance infrastructure remains resilient over time.


