Regulatory Submissions Management Software
Plan, author, track, and publish submissions with speed and control
Regulatory submissions are critical to bringing products to market—but too often, they are slowed by disconnected tools and manual processes. Advanced Submissions by Rimsys is the only regulatory submissions software that brings planning, content, review, and publishing into one controlled workflow.
The result is faster submissions, fewer errors, and greater confidence in execution—with optional AI capabilities available to accelerate work when teams are ready.

Outcomes
What teams achieve with Rimsys submissions management
Efficient
PREPARATION
Faster submission preparation
with structured workflows and reusable content
Controlled
QUALITY
Reduced errors
through controlled review and approval processes
Team
ALIGNMENT
Improved coordination
Improved coordination across regulatory, clinical, and quality teams
Live
TRANSPARENCY
Greater visibility
into submission status, readiness, and blockers
Faster
APPROVALS
Accelerated approvals
with clear traceability and ownership and AI-assisted workflows
The Problem
Why Regulatory Submissions Break Down At Scale
As organizations grow, regulatory submission management becomes increasingly complex—and fragile.
Common breakdowns include:
Disconnected authoring, review, and approval workflows
Manual coordination across SMEs, consultants, and global partners
Version control issues across documents and teams
Limited visibility into submission readiness and status
Rebuilding similar submissions repeatedly for different markets
The Approach
A Structured System For Regulatory Submission Execution
Regulatory submissions are inherently cross-functional. Advanced Submissions is designed to support real-world collaboration—not force teams into disconnected tools.
Work across Regulatory Affairs, clinical, quality, and external partners
Replace email and spreadsheets with task-based workflows
Maintain clear accountability throughout the submission lifecycle
Enable co-authoring and review directly within the platform
Teams can author and edit Word-compatible documents directly within the system, eliminating the need to move between tools
Platform Benefits
Benefits of Rimsys Regulatory Submissions Software

Clear submission plans with defined ownership
Eliminate ambiguity with structured submission plans, assigned responsibilities, and aligned timelines—so nothing falls through the cracks.
.avif)
Reusable, structured content
Move beyond document sprawl. Reuse approved content across markets and submissions to reduce duplication and accelerate execution.
.avif)
Real-time visibility into submission status
Track progress, identify blockers, and monitor readiness across all submissions in one system.
.avif)
Fewer errors through controlled workflows
Standardized review and approval processes reduce last-minute issues and ensure consistency across submissions.
.avif)
Faster publishing with submission-ready outputs
Generate submission-ready documents and outputs directly from the platform—without manual formatting or rework.
.avif)
Optional AI to accelerate execution
For teams ready to adopt AI, Advanced Submissions includes optional AI capabilities that can:
- Summarize and refine regulatory content
- Expand or rewrite sections into clearer regulatory language
- Assist with translation and deficiency response preparation
Real-World Workflow
Designed for the Reality of Submission Execution
Collaboration, not document chasing.
Regulatory submissions are inherently cross-functional. Advanced Submissions is designed to support real-world collaboration—not force teams into disconnected tools.

Full Lifecycle
Beyond Submissions: A Connected Regulatory System
Connected to registrations, intelligence, and change.
Submissions do not exist in isolation—they are part of a broader regulatory ecosystem.
Rimsys connects submissions to:
Product registrations and global market status
Regulatory intelligence and market entrance requirements
Impact assessments triggered by regulatory or product changes
Standards, essential principles, and compliance evidence
With optional AI embedded across workflows, teams can also identify potential impacts earlier and standardize processes across markets, helping reduce compliance risk and improve speed to market
How It Works
Inside the Rimsys Submission Workflow
Advanced Submissions provides a complete, end-to-end workflow:
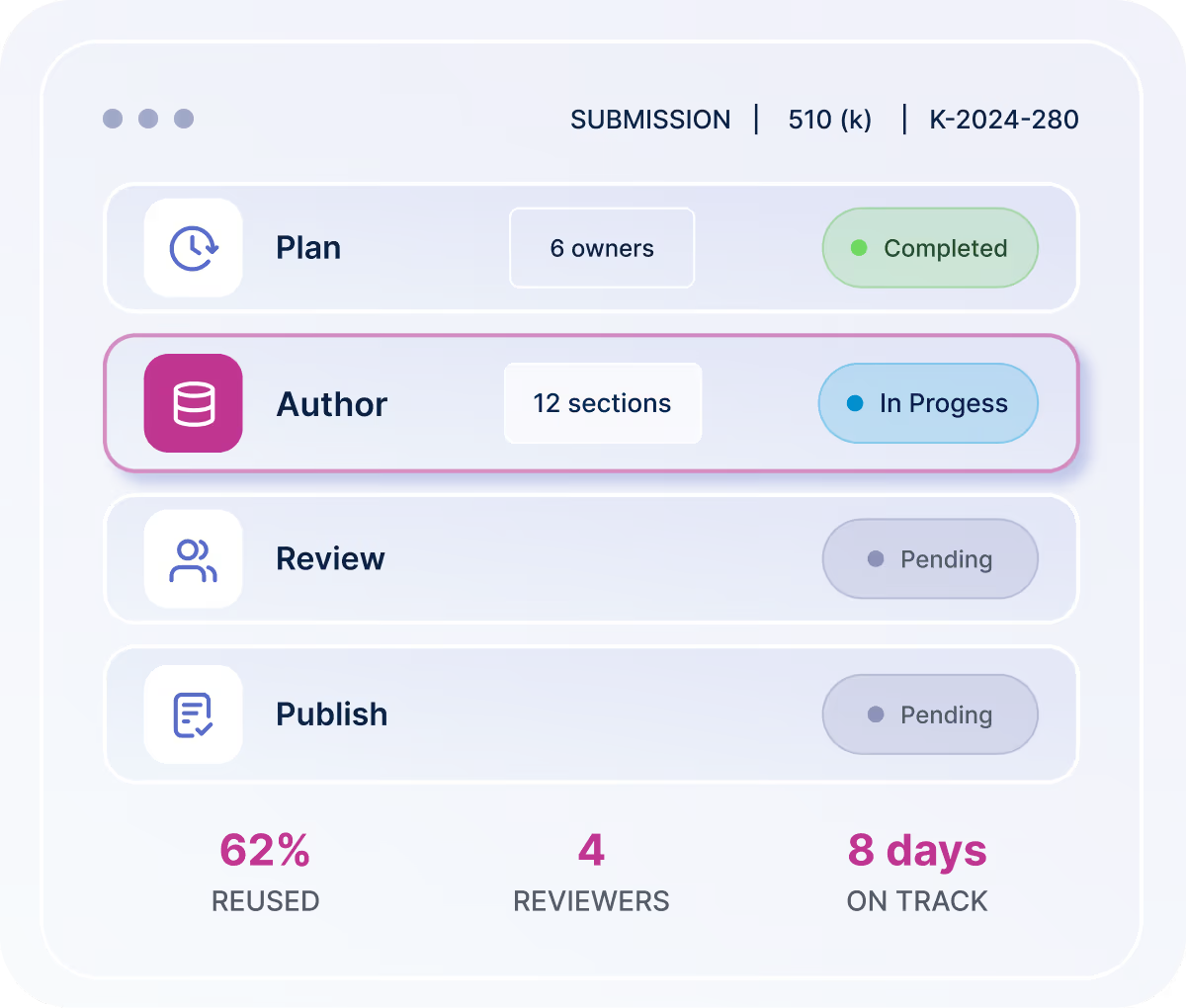

Plan submissions with structured timelines and ownership

Author content using word-compatible AI-assisted editing within the platform

Collaborate and review with version control and approval tracking

Reuse content across markets with automated mapping

Publish outputs in submission-ready formats
Enterprise Scale
Enterprise-Ready Submission Management
Built for global MedTech organizations, Advanced Submissions supports:
Multiple submission types (e.g., 510(k), PMA, STED, and more)
Submissions across regions, markets, and regulatory authorities
Full auditability and traceability of decisions and changes
Integration with enterprise systems and data environments
Who It’s For
Teams That Benefit Most From This Solution
Regulatory Affairs teams
Manage complex submission pipelines with greater speed, control, and visibility.

Regulatory Operations leaders
Coordinate global execution and standardize processes across regions.

Growing MedTech companies
Scale submissions efficiently as they expand into new markets or manage change-driven updates.

Experience Streamlined Regulatory Submissions

With Advanced Submissions, teams gain the structure, visibility, and control needed to execute faster—reduce submission stress—and bring products to market with confidence.
FAQs
Regulatory submissions management software is a system that helps teams plan, author, track, review, and publish regulatory submissions in a structured and controlled environment.
It standardizes workflows, enforces review and approval processes, and provides real-time visibility—reducing manual mistakes and last-minute issues.
Rimsys enables teams to manage submissions across multiple markets, reuse content, and align submissions with global regulatory requirements and product data.
Yes. The platform is designed to support high volumes of concurrent submissions with full visibility into status, ownership, and progress.
Submissions are directly linked to product registrations, ensuring alignment between submission activity and market approvals.
Implementation timelines vary, but Rimsys follows a proven, high-touch approach designed to accelerate adoption and deliver value quickly.
Typically, Regulatory Affairs or Regulatory Operations teams own submission execution, with collaboration from clinical, quality, and external partners.

