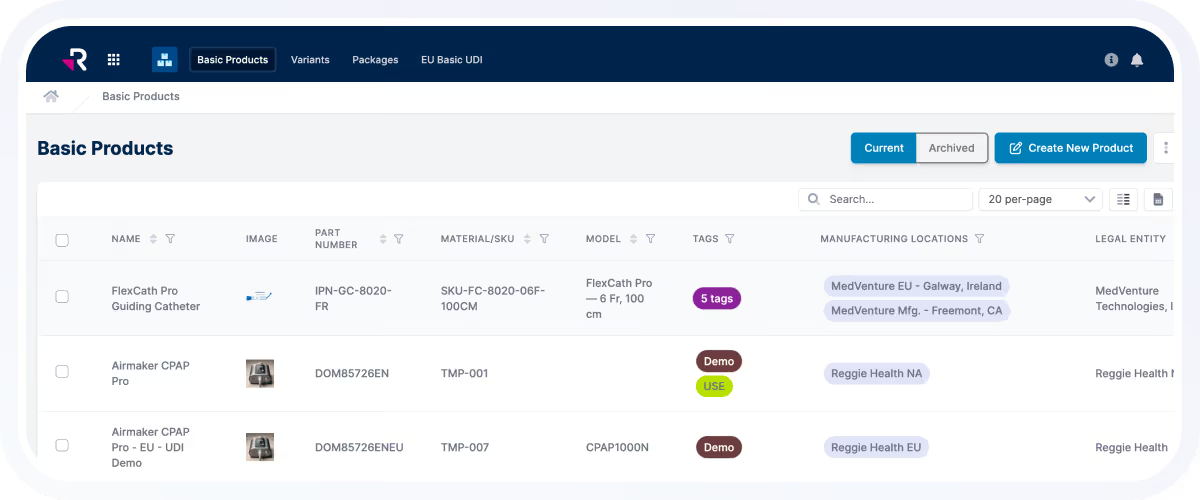
Medical Device Market Access Software
Plan, Execute, and Maintain Market Access With Regulatory Operations Software
Rimsys transforms regulatory operations into the heart of compliant growth. By connecting regulatory intelligence, product data, submissions, and approvals, teams gain the clarity and control to expand into global markets with speed and confidence.
This is not advisory support. It is enterprise software that enables repeatable, visible, and governed market entry at scale.

The Stakes
The Cost of Getting Market Entry Wrong
When global expansion breaks down, the consequences are immediate:
Delayed revenue from missed launch windows
Regulatory rework due to incomplete or misaligned submissions
Missed distributor or commercial commitments
Leadership uncertainty due to lack of visibility
Increased compliance risk across markets
The Gap
The Gap Between Strategy and Execution
Most MedTech companies have a global growth strategy. Few have an operational system to execute it at scale.
Common breakdowns include:
Each new market treated as a one-off initiative
Requirements scattered across documents and disconnected tools
Limited visibility into product-country readiness
Difficulty coordinating submissions, registrations, risk class, and evidence
No structured way to reuse intelligence across markets
The Rimsys Difference
Why Rimsys: A Platform-First Approach to Global Market Entry
Rimsys creates a living, connected regulatory operation.
As the system of record, it ensures:

A single source of truth for regulatory data

Connected workflows across intelligence, submissions, and registrations

Retained knowledge that compounds over time

Full control for internal teams
The result: faster execution, stronger compliance, and scalable global growth.
Inside the Platform
How Market Access Works Inside the Platform
Market access inside Rimsys is not a disconnected project—it is a connected operational flow.

Regulatory Intelligence: Understand Requirements Before You Move
Rimsys AI-powered regulatory intelligence provides structured visibility into market entrance requirements by country and risk class. Teams gain clarity on documentation, timelines, classification, and local nuances before committing resources.
AI-driven intelligence is embedded directly into workflows—secure, explainable, and optional for organizations ready to accelerate execution.

Submissions: Execute With Structure and Control
Submissions are built and managed inside a governed system:
- Centralized document and data management
- Clear ownership and status tracking
- Structured approval workflows
- Real-time visibility across regions
By eliminating silos between product data and submission activity, Rimsys reduces rework and accelerates approval cycles.
Registrations: Secure and Track Market Authorization
Rimsys manages market registrations globally, including complex dual registrations (e.g., ANVISA + INMETRO in Brazil)
Teams gain:
- Country-level selling status visibility
- Renewal tracking and automated notifications
- Structured lifecycle management
- Traceable audit history
Minutes instead of weeks for regulatory release authorization becomes possible when information is centralized and governed

Standards, GSPR, and UDI: Support Ongoing Compliance
Market entry does not stop at approval.
Rimsys connects:
- Standards and regulatory requirements
- GSPR documentation
- Universal UDI for EUDAMED compliance
- Ongoing change management
Compliance stays alive inside the system, rather than living in static documents.
Compounding Value
A Unified Platform for Global Expansion
Rimsys connects every stage of market access:
Key platform differentiators include:
Product data is reusable across markets
Risk classifications are country-specific and structured
Submissions reference existing data instead of recreating it
Approvals flow into ongoing registration management
Intelligence improves with each expansion
As you expand, the system becomes more valuable, not more complex.
Full Lifecycle
From Planning to Execution toto Maintenance
Rimsys supports the full lifecycle.

Understand Requirements
Leverage regulatory intelligence and risk class data to determine entry pathways and documentation needs.

Prepare and Submit
Coordinate evidence, documentation, and workflows inside a governed submission framework.

Secure Registrations
Track approvals, country authorizations, and selling status across global markets.

Maintain Compliance
Manage renewals, amendments, additional information requests, and regulatory changes from a central system.
Trusted for Global Market Entry Execution
MedTech manufacturers rely on Rimsys to bring structure and confidence to global expansion.
Global
Authorizations
Global Enterprise Manufacturer
Challenge:
Disconnected approval tracking and manual reporting.
Result:
Harmonized source of product approval information and unified lifecycle tracking—reducing reporting effort by over 90%.
Global
Authorizations
“Rimsys is intuitive and well-structured, making it easy to manage global market authorizations without getting lost in administrative complexity.”
- Senior Regulatory Affairs Specialist, Mid-Sized MedTech Manufacturer
UDI
Submissions
Global Enterprise Manufacturer
Challenge:
Disconnected approval tracking and manual reporting.
Result:
Harmonized source of product approval information and unified lifecycle tracking—reducing reporting effort by over 90%.
UDI
Submissions
“Rimsys allows us to manage UDI submissions along with registrations and regulatory submissions in one system.”
- Enterprise MedDevice User
Organizations report:

Faster launches across multiple regions

Fewer last-minute submission surprises

Clearer visibility into global readiness

Reduced manual tracking and spreadsheet reliance

Improved cross-functional collaboration
Take Control of Global Market Expansion
Global market entry should feel controlled.

Rimsys transforms regulatory operations into the operational foundation for compliant growth. By keeping regulatory intelligence, product data, submissions, and approvals continuously connected, your team gains the clarity and confidence to scale.
FAQs
Medical device market access refers to the structured process of gaining regulatory approval and authorization to sell a medical device in a specific country or region. It includes understanding regulatory requirements, preparing submissions, securing approvals, and maintaining ongoing compliance.
Market entry typically refers to the initial approval or registration that allows a device to be sold in a region. Market access is broader—it includes entry, lifecycle maintenance, renewals, regulatory updates, and ongoing selling status management.
Each country has unique regulatory frameworks, risk classifications, documentation standards, and renewal requirements. Without a centralized system, managing these differences across multiple markets becomes fragmented and error-prone.
Common delays include incomplete documentation, misaligned risk classification, poor coordination between teams, lack of visibility into readiness, and manual tracking across disconnected systems.
Rimsys serves as the system of record for regulatory operations. It connects regulatory intelligence, submissions, registrations, UDI, and compliance activities into one governed platform—accelerating execution while reducing risk
Product data, risk classification, and regulatory intelligence flow directly into submission workflows. Once approved, registrations are tracked within the same platform, ensuring continuity from planning through lifecycle maintenance.
Yes. Rimsys is built for enterprise-scale portfolios, supporting global product structures, bulk actions, and cross-region visibility. Teams can manage concurrent market launches while maintaining centralized control and audit-ready traceability

