UDI Compliance Software
Manage Global UDI Requirements, Reporting, and Change in One System
Rimsys is UDI compliance software built for MedTech. It systematizes how UDI data is created, managed, updated, and reported across global markets.
Instead of managing UDI in disconnected tools, Rimsys connects identifiers, product data, registrations, and regulatory workflows in a single platform. The result is consistent, audit-ready UDI compliance at scale, with less manual effort and lower risk.

The Results
UDI Compliance Results You Can Measure
90%
REDUCTION
Reduce manual UDI tracking effort by 90%+
Eliminate spreadsheets and duplicate entry so teams focus on execution, not administration.
Minutes
NOT WEEKS
Update UDI across markets in minutes, not weeks
Changes to product data automatically flow through the system, reducing delays and rework.
Audit Ready
DATA
Improve accuracy and audit readiness
Centralized, structured data reduces inconsistencies that lead to audit findings.
Real-time
VISIBILITY
Gain real-time visibility into UDI status
Know what is submitted, approved, or at risk across markets without manual reporting.
Faster
EXECUTION
Accelerate global compliance execution
Connected workflows reduce bottlenecks and keep regulatory operations moving.
Key Challenges
Common UDI Compliance Challenges in MedTech
UDI compliance becomes difficult as portfolios and markets grow.
Requirements vary across FDA GUDID, EU MDR, China, Saudi Arabia, and others.
Labeling, packaging, and configuration updates require ongoing UDI maintenance.
Spreadsheets, portals, and local processes create inconsistency.
The result is risk.
Inaccurate data, missed updates, and audit exposure increase as complexity grows.
How It Works
UDI Compliance Managed in One Platform
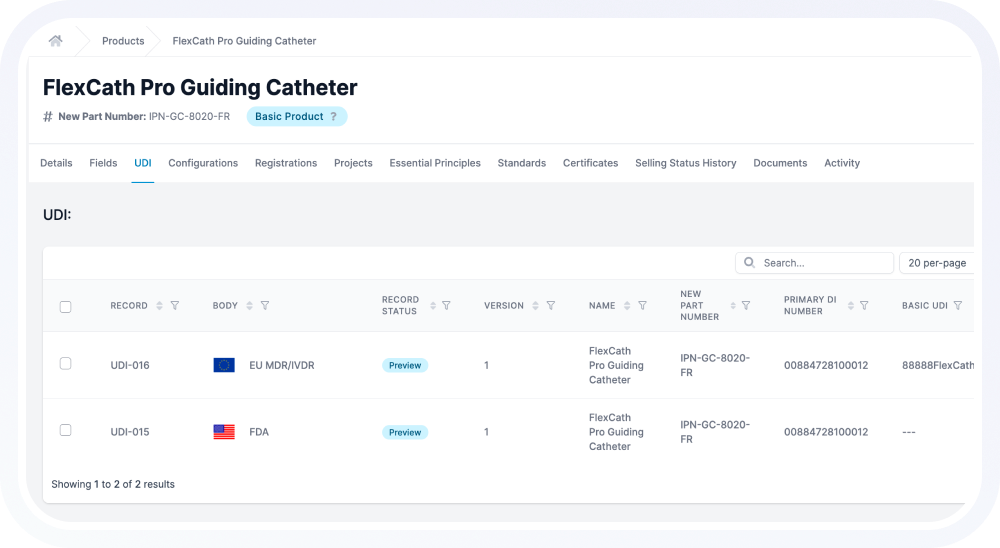
Rimsys acts as the system of record for UDI.
.avif)

UDI data is centralized at the product level and structured for global use. Each identifier, attribute, and relationship is managed in a controlled environment.

When product data changes, UDI updates are triggered automatically. Teams no longer rely on manual tracking or memory.

The platform supports both data management and submission workflows, ensuring UDI compliance is maintained continuously, not just at submission.
Connected System
One System That Keeps UDI in Sync
UDI does not exist in isolation. It is part of a broader regulatory operation.

Product Registrations
Rimsys connects UDI data to product registrations, ensuring alignment between identifiers and market approvals.

Regulatory Submissions
Rimsys links to regulatory submissions, so UDI content is consistent with submitted documentation.

Regulatory Intelligence & Change Management
Rimsys integrates with regulatory intelligence and change management, so updates are assessed and acted on in context.
UDI Lifecycle
The Core Steps of UDI Compliance
UDI compliance follows a repeatable lifecycle inside Rimsys.
Define UDI requirements by market
Identify country-specific rules and required attributes.
Manage identifiers and product data
Maintain UDI-DI, UDI-PI, and associated attributes in a structured system.
Update UDI when products change
Trigger updates automatically when product, labeling, or packaging changes occur.
Support reporting and audits
Generate submission-ready data and maintain a full audit trail.

.avif)

UDI Compliance at Scale, From Customers
Take Control of Global UDI Compliance

Rimsys gives you a single system to manage UDI data, maintain accuracy, and execute compliance across markets with confidence.
FAQs
UDI compliance requires assigning and maintaining unique device identifiers for medical devices and submitting that data to regulatory authorities such as FDA GUDID and EUDAMED.
UDI compliance refers to meeting requirements in a single market. Global UDI compliance involves managing different requirements, formats, and submissions across multiple countries.
UDI software centralizes identifier data, manages updates, supports submissions, and ensures consistency across products and markets.
By linking UDI data to product records and change workflows, updates are triggered automatically and tracked through completion.
UDI data must align with product registrations and submission content. A connected system ensures consistency across all regulatory records.
Yes. Enterprise platforms support global UDI requirements across regions including the US, EU, China, and others.
Look for centralized data management, automated updates, global coverage, audit traceability, and integration with broader regulatory workflows.

