Medical Device Product Registration Software
Manage, track, and maintain global registrations in one unified system
Transform product registration from manual tracking into a connected, enterprise-ready regulatory operation.
Rimsys is the heart of regulatory operations—bringing registrations, approvals, product data, and intelligence into one living system. Instead of managing country trackers and spreadsheets gain real-time visibility into global selling status at scale.
- Trusted by 6 of the top 12 global MedTech manufacturers, Rimsys helps organizations accelerate compliant growth while reducing operational risk.

Measurable Impact
Key Registration Outcomes Teams Achieve with Rimsys
Minutes
not weeks
Minutes Instead of Weeks for Regulatory Release Authorization
Instant visibility into where products are approved, pending, restricted, or expired—across every market.
90%
REDUCTION
Up to 90% reduction in manual registration tracking effort
Eliminate spreadsheet reconciliation and manual follow-ups with structured, governed workflows.
Faster
RENEWALS
Faster renewal and expiration management
Automated alerts and lifecycle tracking reduce the risk of missed renewals and revenue disruption.
100+
COUNTRIES
Support for thousands of SKUs across 100+ countries
Manage complex portfolios—including kits, variants, and bundles—inside one structured system.
Real-time
VISIBILITY
Enterprise-wide visibility for leadership
Answer executive questions about product availability, compliance status, and expansion readiness in seconds—not days.
The Challenge
Why Medical Device Product Registration Becomes Unmanageable
Registration management rarely fails because teams lack expertise. It fails because the operating model cannot scale.

As portfolios expand and global markets multiply, most organizations rely on:
Enterprise Architecture
- Fragmented spreadsheets by region or distributor
- Country-specific trackers maintained by individual team members
- Manual renewal reminders in calendars or inboxes
- Disconnected submission documentation stored in shared drives
- Static reports that are outdated the moment they are created


The result is operational friction:
Enterprise Architecture
- No single source of truth for selling status
- Risk of missed renewals or expired approvals
- Inconsistent data between submissions and registrations
- Difficulty answering basic leadership questions quickly
- Heavy dependence on tribal knowledge

The Solution
Centralized Product Registration Tracking, Built for MedTech
Rimsys centralizes registrations, certificates, approvals, and supporting documentation at the individual product level—creating a structured, product-centric source of truth.
Instead of treating registrations as standalone documents, Rimsys connects them to:

Products and variants

Country-specific classifications

Submission records

Certificates and approvals (including dual registrations such as ANVISA + INMETRO)

UDI data

Regulatory intelligence
Because the platform is purpose-built for MedTech, it understands complex product hierarchies, country nuances, and risk classifications. The structured data model begins at implementation - giving organizations an enterprise-grade foundation that scales as portfolios grow.
The result is clarity. Registrations are no longer buried in trackers—they are visible, connected, and continuously maintained as part of a living regulatory operation.
Platform Benefits
Benefits of Rimsys Registration Software

Always-Current Global Selling Status
Know exactly where every product can be sold, across every market. Real-time status tracking eliminates guesswork and provides confidence to regulatory, commercial, and executive teams.
What becomes easier:
- Supporting market expansion decisions
- Providing instant answers to sales and leadership
- Reducing delays caused by uncertainty
.avif)
Automated Tracking of Expirations and Renewals
Rimsys monitors registration lifecycles and alerts teams to upcoming expirations, renewals, and additional information requests.
What becomes safer:
- Reduced risk of missed renewals
- Fewer last-minute escalations
- Greater continuity of market access
.avif)
Faster Access to Registration Data for Audits and Planning
Audit readiness is built into the system through governed workflows, structured records, and traceable history.
What becomes easier:
- Responding to health authority inspections
- Preparing internal compliance reports
- Supporting due diligence and M&A activity
.avif)
Reduced Dependency on Tribal Knowledge and Spreadsheets
Institutional knowledge becomes structured data—not institutional memory.
What becomes stronger:
- Onboarding new team members
- Cross-regional collaboration
- Long-term process resilience
.avif)
Improved Collaboration with Distributors and In-Country Partners
Unlimited user licensing allows controlled access for internal teams and external stakeholders.
What becomes faster:
- Managing country-specific updates
- Sharing documentation securely
- Coordinating renewals and amendments
Connected Ecosystem
More Than Just Registration Management
A Foundation Connected to Submissions, Intelligence, and Change
Registrations do not exist in isolation. They are directly impacted by:
Regulatory Submission
Changing country requirements
Product design changes
Labeling updates
UDI and standards evolution
Rimsys connects registration data to the broader regulatory ecosystem:
Regulatory Submissions
Build and manage submissions directly alongside the registrations they support—reducing duplicate entry and silos.
Regulatory Intelligence
Track evolving global requirements and understand how changes affect existing registrations and market access.
Impact Assessment and Change Control
When a product or regulation changes, teams can assess impact, assign tasks, and manage updates directly within the system.
UDI and Standards Management
Registration records link to structured UDI data and global standards, ensuring alignment between approvals and device identification.
Rimsys AI (Optional)
AI-assisted workflows accelerate assessments, content preparation, and impact analysis—while keeping human judgment in control. Designed specifically for MedTech regulatory structures, Rimsys AI enhances execution without compromising compliance.
REGULATORY EVENTS
Regulatory submissions
Changing country requirements
Product design changes
Labeling updates
UDI and standards evolution
CENTRAL HUB
Product Registration
Approvals · Certificates · Selling Status
CONNECTED capabilities
Regulatory Submission
Regulatory Intelligence
Impact Assessment and Change Control
UDI and Standards Management
Rimsys AI
(Optional)
A connected regulatory operating system
Platform Experience
What Managing Registrations Looks Like Inside Rimsys
Inside Rimsys, registration management is structured, visual, and connected.Should these product sections all be videos eventually?
Teams can:

View global product hierarchies with linked country registrations

Filter by market, status, risk class, or expiration date

Access supporting certificates and documentation instantly

Track submission progress alongside approval status

Monitor dashboards showing renewal risk and selling status
Bulk actions allow updates at scale, while governed approval workflows ensure changes are controlled and traceable.

Enterprise Infrastructure
Built for Enterprise Regulatory Teams
Global MedTech organizations require more than tracking tools. They need infrastructure. Rimsys delivers:
Support for Complex Portfolios
Manage thousands of SKUs, variants, kits, and bundles across global markets.
Speak With Our Team
Controlled Access Across Stakeholders
Provide secure access to internal teams, distributors, and partners without sacrificing governance.
Speak With Our Team
Audit Readiness and Traceability
Structured data, version control, and real-time history ensure inspection confidence.
Speak With Our Team
Enterprise Integration
Robust APIs and event-driven BI integration connect registration data to ERP, CRM, and analytics platforms—bringing regulatory visibility into enterprise dashboards.
Speak With Our Team
Security and Compliance
SOC 2 and ISO 27001 certified, with single-tenant environments and enterprise-grade uptime.
Speak With Our Team
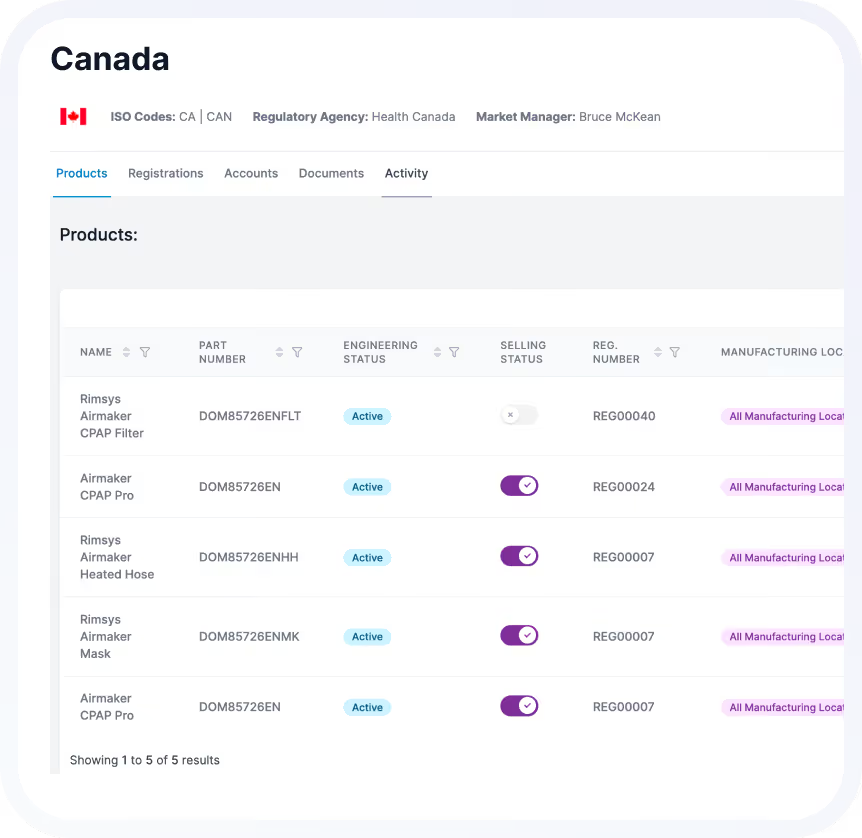

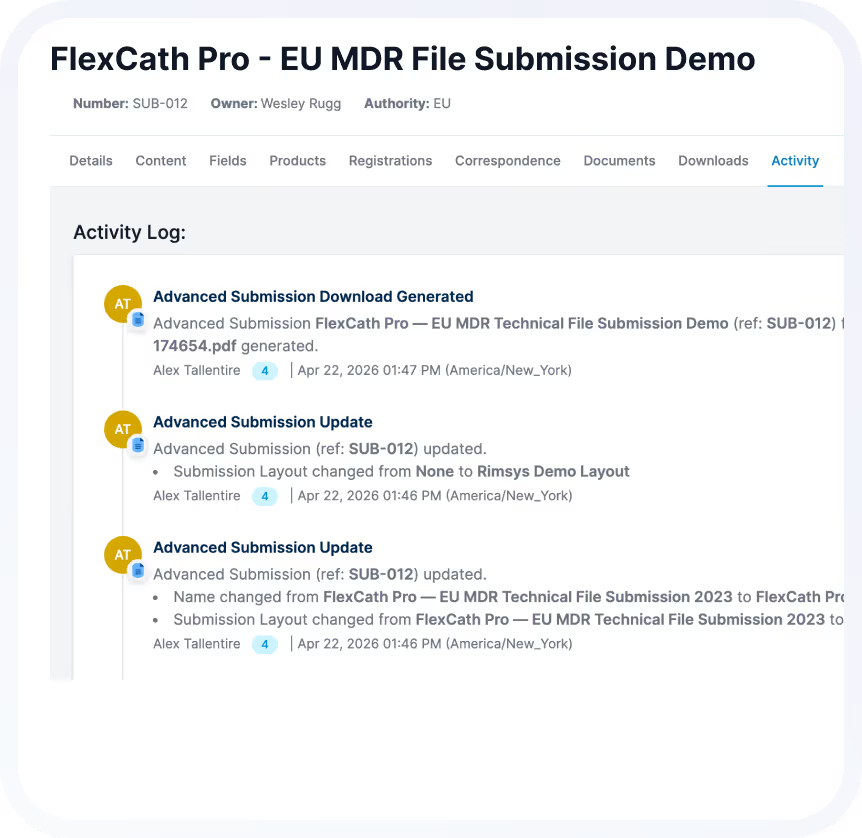

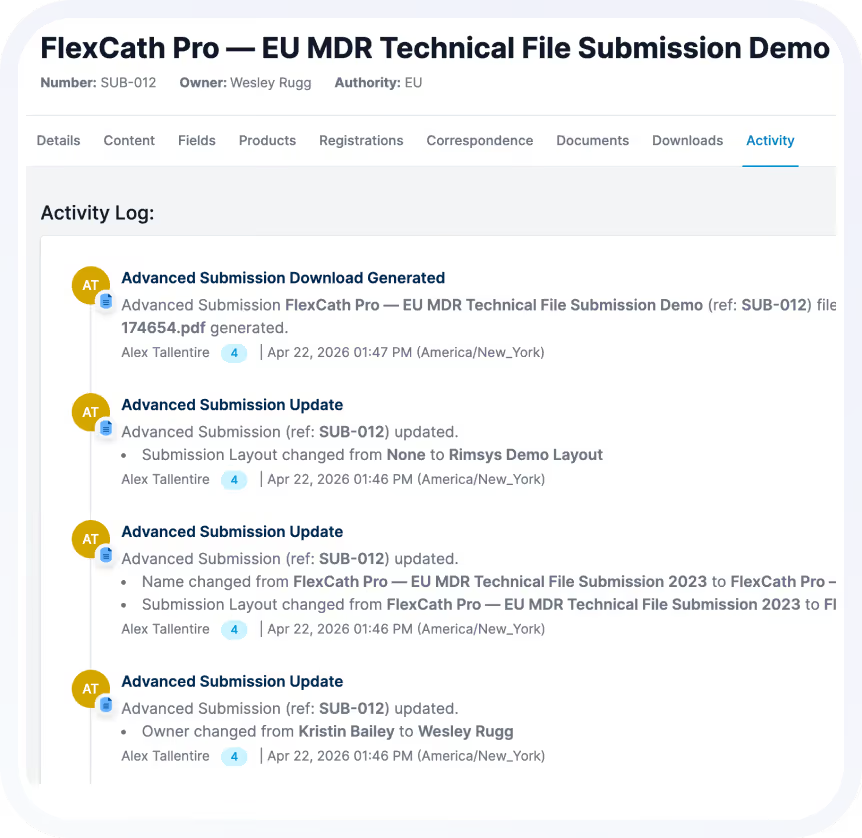
See How Teams Manage Global Registrations with Confidence
When registrations are connected, visible, and continuously maintained, regulatory teams shift from reactive tracking to strategic enablement.

Rimsys helps organizations:
Enter new markets with confidence
Provide enterprise-wide regulatory clarity
Maintain uninterrupted global selling status
Reduce compliance risk
FAQs
Medical device product registration software centralizes the tracking and management of market authorizations, certificates, approvals, and renewals across global markets. It replaces spreadsheets and disconnected trackers with a governed, structured system.
By automating renewal alerts, centralizing documentation, and providing real-time visibility into selling status, the software reduces the risk of missed expirations, inconsistent data, and audit gaps.
Rimsys connects registrations directly to products, submissions, certificates, UDI, and regulatory intelligence within a structured data model. This creates a continuously updated source of truth for global market access.
Yes. Rimsys is designed to eliminate fragmented country trackers and manual spreadsheets by providing centralized lifecycle management and automated workflows.
Registration records in Rimsys are linked to submission modules and impact assessment workflows. When a product or regulation changes, teams can assess impact and manage updates directly within the platform.
Implementation timelines vary based on portfolio size and data maturity. Rimsys follows a proven, high-touch implementation approach guided by MedTech regulatory expertise to ensure structured data from day one.
Ownership typically sits within Regulatory Affairs, often in collaboration with regional regulatory leads and in-country partners. With Rimsys, collaboration extends securely across teams while maintaining centralized governance.

