Rimsys Connect: Real-Time Change Data Capture For RIM
Synchronize regulatory data across your enterprise with change data capture solutions built for MedTech. Rimsys Connect streams structured regulatory data in near real-time to BI, ERP, QMS, and data platforms so your organization operates from a single source of truth.
Combine regulatory data with revenue, product, and operational KPIs to drive faster, more confident decisions.

Live Operational Intelligence
Turn Regulatory Data Into Actionable Insight
Rimsys Connect transforms regulatory data from static records into live operational intelligence.

Regulatory updates are captured and streamed automatically across systems, including:
- Product registrations and approvals
- Submission status and milestones
- UDI datasets and identifiers
- Certificates and expiration timelines

This enables:
- Near real-time synchronization across systems
- Automatic updates when regulatory records change
- Reduced reporting lag for leadership
- Consistent data across BI, ERP, and QMS
- Faster, more confident decision-making
Instead of exporting spreadsheets and reconciling reports, your teams work from continuously updated data. This solves common enterprise challenges:
This solves common enterprise challenges:
Regulatory teams
relying on manual exports
BI teams
working from outdated snapshots
IT teams
maintaining fragile custom scripts
Architecture
Connect Rimsys To Your Enterprise Stack
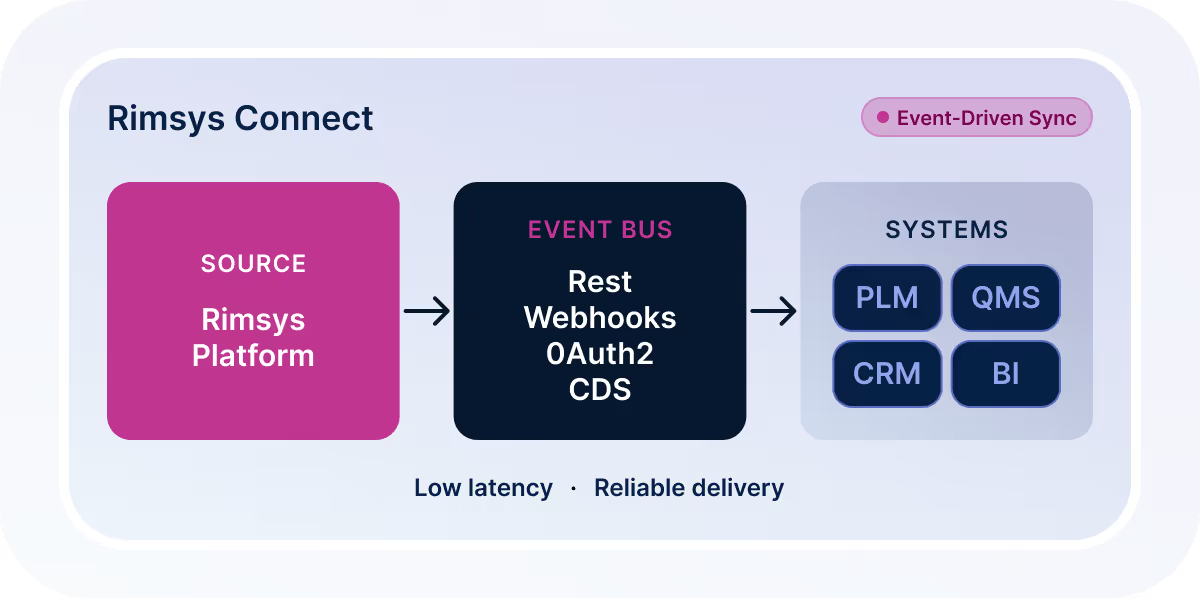
Rimsys Connect is built for enterprise integration at scale.
It delivers secure, event-driven data synchronization using modern architecture:
API-first platform for structured data access
Event-based updates for real-time change data capture
OAuth 2.0 authentication for secure access
Single-tenant architecture with full data isolation
Platform Continuity
Part Of The Complete Rimsys Platform
Rimsys Connect extends the value of the entire regulatory system by keeping data continuously connected across workflows.
As the heart of regulatory operations, Rimsys ensures regulatory intelligence, product data, submissions, and compliance activities remain synchronized across systems.

Connect enhances:
Enterprise Architecture
- Executive dashboards with live regulatory data
- Global compliance tracking across markets
- Market expansion planning with real-time visibility


It integrates directly with:
Enterprise Architecture
- UDI Management
- Submissions
- Standards and Essential Principles
- Regulatory Impact Assessment
- Rimsys AI

Use Cases
Where Rimsys Connect Delivers Immediate Value
Executive Dashboards
Submission Tracking
Registration Analytics
Certificate Monitoring
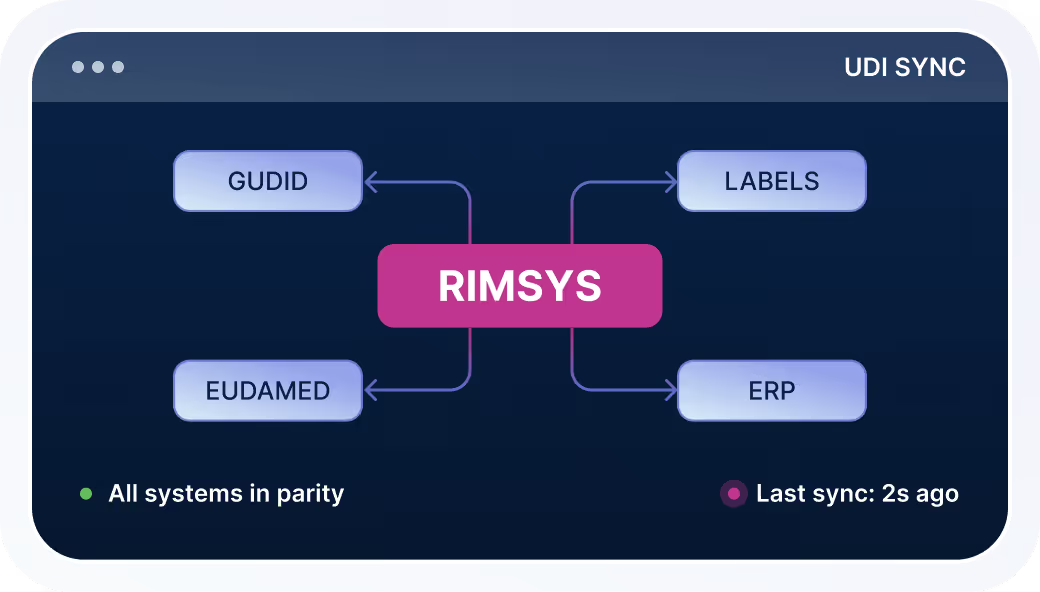
UDI Synchronization
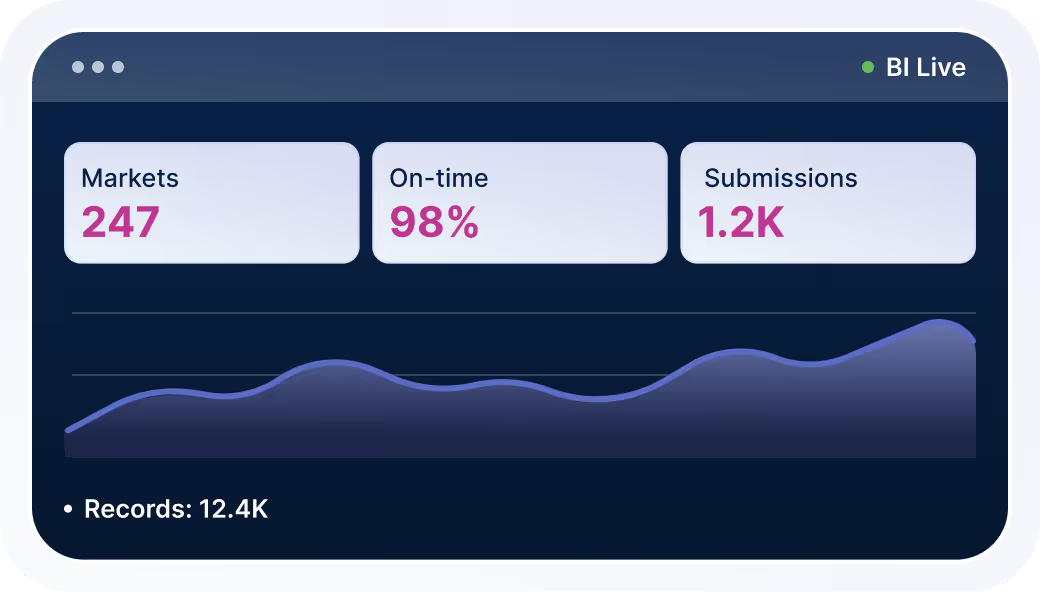
Executive Dashboards
Real-time regulatory data flows into BI platforms for accurate, up-to-date reporting.
Submission Tracking
Automatically monitor submission status across markets without manual updates.
Registration Analytics
Analyze country-level approvals, timelines, and trends with live data.

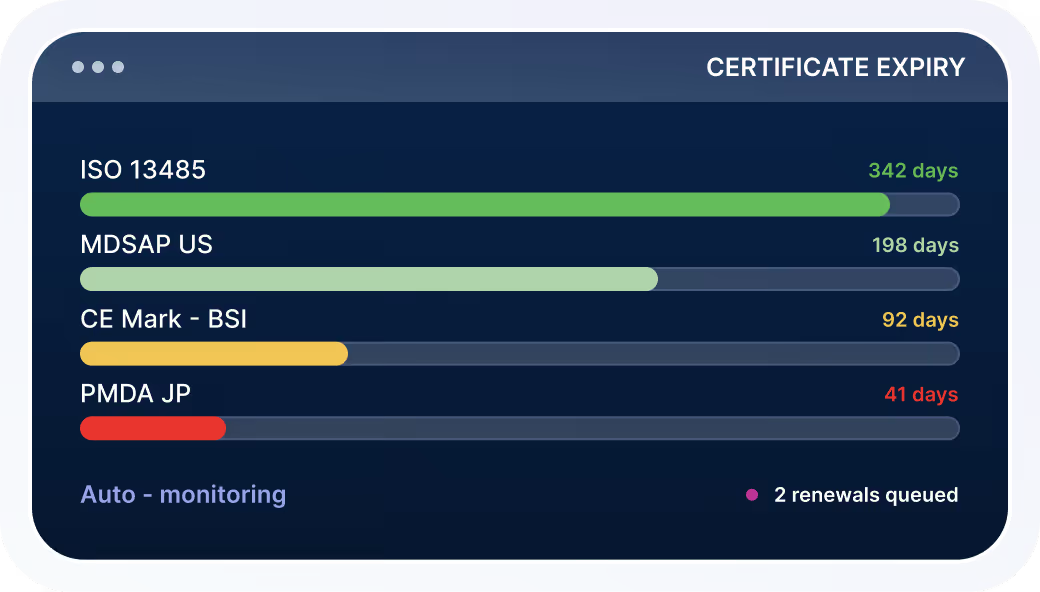
Certificate Monitoring
Track expirations and renewals without relying on static reports.
UDI Synchronization
Keep labeling and downstream systems aligned with current UDI datasets.
Trusted By Global MedTech Teams
Synchronize Your Regulatory Data Today

Rimsys Connect ensures your systems stay aligned, your data stays accurate, and your teams move faster with confidence.
FAQs
Real-time data ensures leadership, regulatory, and operational teams are working from the same information. This reduces delays, improves reporting accuracy, and supports faster decisions.
Change data capture automatically updates downstream systems when regulatory records change. This eliminates version mismatches and manual reconciliation.
Traditional exports create static snapshots. Rimsys Connect continuously synchronizes data, ensuring systems stay aligned without manual intervention.
Rimsys Connect is API-first and designed for enterprise integration. It reduces the need for custom scripts while supporting flexible integration architectures.
Rimsys Connect integrates with BI platforms, ERP systems, QMS, data lakes, and other enterprise applications.
Yes. Rimsys supports secure authentication, tenant isolation, and enterprise-grade security standards.
Yes. It is designed to handle complex global regulatory data across products, markets, and authorities.
Rimsys Connect is available as part of enterprise platform tiers or as an add-on capability based on your integration needs.