Regulatory Intelligence Software for MedTech
Rimsys Intelligence is an AI-powered regulatory intelligence software database purpose-built for medical device manufacturers.
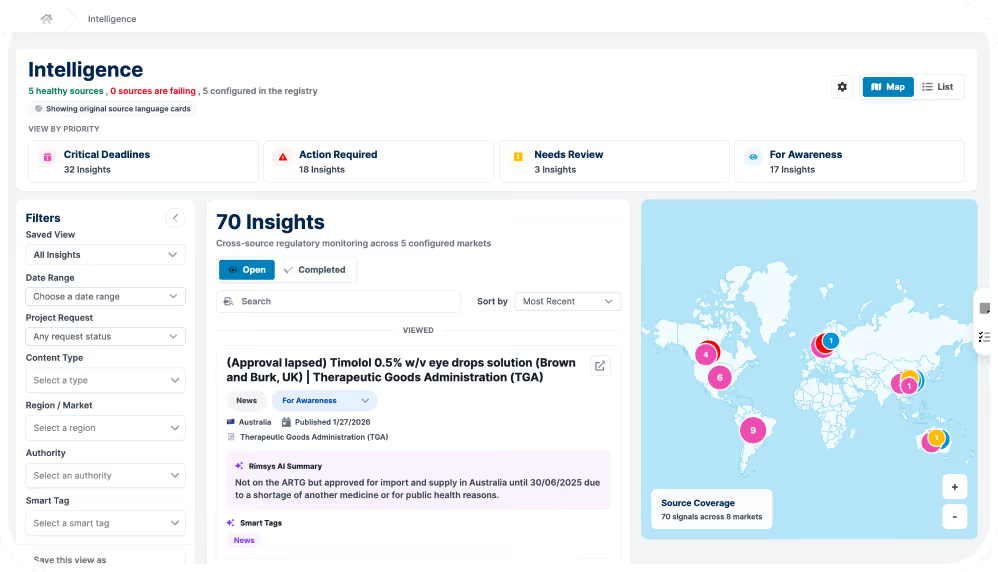
Intelligence transforms how regulatory teams identify what matters, prioritize and take action, and move faster with confidence.

Open Intelligence Layer
Regulatory Intelligence Should Be Open and Accessible
Regulatory intelligence is only valuable when it is accessible, structured, and continuously evolving. Intelligence delivers a centralized hub powered by AI to help teams cut through noise and focus on what matters.
.svg)
A centralized regulatory intelligence database built on a structured data model
.svg)
Access to more than 90 global market profiles
.svg)
Clear visibility into legislation, regulatory pathways, and UDI requirements
.svg)
Detailed risk classification requirements by country
.svg)
Standardized market access checklists
Global Intelligence Network
Built With and For the Regulatory Community
90+
countries and regulatory jurisdictions monitored
2-4
hours per person/day saved
4
product designations
Medical Device
IVD
SaMD
Combination Products
More Coming
6
content types tracked
Guidances
Product Recalls
Safety Alerts
News & Media
Combination Products
More Coming
Decision Clarity
How Rimsys Intelligence Reduces Risk
Regulatory teams don’t lack information—they lack clarity on what is relevant.
Rimsys Intelligence uses AI to filter, prioritize, and connect regulatory changes directly to your products, markets, and submissions—eliminating manual triage and reducing risk.
Operational Impact:
Faster country-level regulatory research with structured data
Reduced misinterpretation risk through summarized and contextualized content
AI-driven prioritization of “action required” updates
Elimination of manual monitoring across multiple sources
Commercial Impact:
Supports proactive market access strategy
Reduces time-to-decision for expansion
Improves confidence before regulatory investment decisions
Enables faster entry into new markets
Smarter Regulatory Decisions
Core Capabilities of the Regulatory Intelligence Database
Rimsys Intelligence combines structured regulatory data with AI-powered analysis to deliver actionable intelligence.
Country & Region Profiles
Legislation Summaries
Risk Class Requirements
AI-Powered Monitoring & Smart Tagging
Country & Region Profiles
Structured, AI-enhanced profiles of regulatory requirements across global markets.
Country Profile: Germany (DE)
→ MDR Classification: Class IIb
→ Notified Body: Required
→ Languages: DE + EN
✓ Profile Ready
Legislation Summaries
AI-assisted summaries of complex regulations and guidance documents for rapid understanding.
EU MDR Article 52 — Summary
→ 14 key obligations identified
→ Key changes highlighted
→ Mapped to GSPR requirements
✓ Summary ready
Risk Class Requirements
Clear classification rules by country, aligned to device type and regulatory frameworks.
Classifying device P-2491
→ BR · Class III
→ US · Class II (510(k))
→ EU · Class IIb
✓ Classification matrix ready
AI-Powered Monitoring & Smart Tagging
Continuous monitoring of regulatory sources with AI-driven tagging, prioritization, and relevance scoring—so teams see what matters first.
Global regulatory monitoring (90+ sources)
→ 2,418 updates analyzed (last 30 days)
→ Tagged, scored, and ranked by relevance
→ 6 high-priority items surfaced
✓ Teams notified
From Insight to Execution
Turning Regulatory Research Into Action
Rimsys Intelligence goes beyond research—it connects intelligence directly to execution.
Integrated within the Rimsys platform, Intelligence powers:
Registrations
Align intelligence with product approvals and market status

Submissions
Inform submission planning with real-time requirements
Impact Assessments
Automatically trigger assessments from regulatory changes

Standards & GSPR
Map intelligence to compliance frameworks

UDI Workflows
Ensure accurate and complete data submission
Integrated within the Rimsys platform, Intelligence powers:
Correlating regulatory changes to products, registrations, and submissions
Automatically identifying impacted markets and portfolios
Enabling one-click creation of impact assessments
What the MedTech Community Says

Explore Rimsys Intelligence
FAQs
Regulatory intelligence software is a solution that centralizes and structures global regulatory data. AI-powered tools like Rimsys Intelligence enhance this by filtering, prioritizing, and connecting intelligence to real regulatory workflows.
Rimsys Intelligence is an AI-powered regulatory intelligence tool designed specifically for medical device companies, combining structured data, continuous monitoring, and workflow integration.
Rimsys Intelligence stands apart by combining:
- AI-driven prioritization and relevance scoring
- Structured, queryable regulatory data
- Continuous monitoring of 90+ verified global sources
- Direct integration with regulatory workflows
Yes. Rimsys Intelligence is fully integrated, enabling intelligence to directly inform submissions, registrations, and impact assessments.
Yes. Rimsys Intelligence is purpose-built for MedTech, incorporating device classifications, UDI requirements, and global regulatory complexity unique to the industry.

