

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

CE marking guide for medical devices in the EU
This article is an excerpt from the CE marking guide for medical devices in the European Union.
Table of Contents
- What is CE marking?
- Why is CE marking important?
- CE marking responsibilities
- What countries require or accept CE marking?
- Which medical devices require a CE mark?
- Technical documentation
- What are the costs associated with CE marking?
- How do you apply the CE marking?
- CE mark and UDI
- Does the CE mark expire?
- Do I need to CE mark my software?
- Final steps
CE marking is a symbol that consists of “CE, “ which is the abbreviation of the French phrase "Conformité Européene" meaning "European Conformity". The term initially used to describe “CE” was "EC Mark" but it has officially been replaced by "CE marking" according to the EU Directive 93/68/EEC. CE marking is used in all EU official documents, although you will still see "EC Mark" being used in common language. If you are using EC Mark in your documentation, you should change that terminology to CE marking in the future.
The letters ‘CE’ appear on many products traded on the Single Market in all the member states of the European Union plus Iceland, Liechtenstein, Norway and Switzerland. Simply put, The CE mark is a mandatory compliance mark, informing the consumer that the product is compliant with all applicable EU directives and regulations where the CE mark is required.
The Single Market was established in 1993 and is still considered one of the most significant achievements of the European Union. The main goal was to ensure the movement of goods and services freely within all the member states and to establish high safety standards for consumers. The CE mark indicates that goods and services do not need to be verified when shipping into another member country. To further support this movement, in April 2011, the Single Market Act was established to boost growth and strengthen confidence in the economy even further.
CE marking is required for many types of products, not just medical devices. The CE symbol can be found on bicycle helmets, toys, laptop batteries, wheelchairs, construction equipment, gas appliances and cell phone chargers - to name a few. CE marking is required for products manufactured anywhere that are sold in the EU, and only for those products for which EU specifications exist and require CE marking. The CE marking signifies that the product has been found to meet the general safety and performance requirements (GSPRs) of the European health, safety and environmental protection legislation and allows the product to be sold in the EU.
Manufacturer responsibilities for CE marking
Medical device manufacturers are responsible for properly and legally CE marking products before they leave the warehouse.
Most Class II and III medical devices, along with IVDs and some Class I devices, require a conformity assessment performed by a Notified Body to ensure that all legislative requirements are met before it can be placed on the market. Manufacturers of most Class I devices can self assess conformity. This process needs to demonstrate that all the legislative requirements are met, including any testing and inspections, and that all necessary certifications are obtained.
The European Commission lists 6 steps that manufactures should follow to affix a CE marking to their devices:
- Identify the applicable directive(s) and harmonized standards - see EU standards for Medical Devices, In Vitro Diagnostic (IVD) devices, and Implantable Medical Devices.
- Verify product specific requirements using the essential principles identified in the above standards.
- Identify whether an independent conformity assessment by a Notified Body is necessary. Notified bodies will be required to verify compliance with relevant Essential Requirements for most medical devices classified as IIa, IIb, or III - along with sterile class I devices. See the Notified and Designated Organization (NANDO) database for available notified bodies.
- Test the product and check its conformity.
- Create and keep available the required technical documentation.
- Affix the CE marking and create the EU Declaration of Conformity.
Importer responsibilities for CE marking
If you are importing medical devices into the EU, it is your responsibility to review all the technical documentation and maintain a copy, or to make sure that it’s available to you upon request.
You should verify:
- That the device has been CE marked and that the EU declaration of conformity has been completed.
- That the manufacturer has designated and established an authorized representative.
- That the device is labeled appropriately and contains instructions for use (IFU).
- When applicable, that a UDI has been assigned to the product.
- Whether or not the product is registered in EUDAMED (registration is currently voluntary).
Take action:
- List your name and address on the device or packaging, in addition to the manufacturer’s information.
- Keep records of complaints, non-conformities, recalls, etc. on file.
- Report any noticed non-conformity or product complaints from end users to the manufacturer and authorized representative immediately.
- Maintain a copy of the EU declaration of conformity and any other relevant certificates.
Distributor responsibilities for CE marking
If you are a distributor, you are responsible for reviewing the technical documentation provided to you so that you can verify the product is safe to put on the local market. You must also be sure the product is labeled correctly with the CE marking symbol clearly visible. The technical file documentation contains all of the information that is necessary to show conformity of the product to the applicable requirements.
You should verify:
- That the device has been CE marked and that the EU declaration of conformity has been completed.
- That the device includes all the appropriate labeling, including instructions for use.
- That if imported, the importer has complied with all the EU regulations.
- When applicable, that a UDI has been assigned to the product.
Take action:
- Report any noticed non-conformity to the manufacturer, importer, and authorized representative immediately.
- If a product appears to be out of compliance to the regulations and could pose a serious risk, the information should be reported to the Competent Authority, and to the manufacturer, importer and authorized representative.
- Any complaints or reports from end users about the product should be reported to the manufacturer and, if necessary, to the importer and authorized representative.
Important note: If the importer or distributor markets the product under their own company name, then they become responsible for CE marketing, and take over that role from the manufacturer.
CE marking is mandatory when importing products into the European Union, which is part of the larger European Economic Area (EEA). The EEA Agreement, established in 1992 and made official in 1994, is an international agreement that enables the extension of the European Union’s single market to non-EU members. It consists of the 27 EU countries plus the four European Free Trade Association (EFTA) countries - Iceland, Liechtenstein, Norway and Switzerland. Today, the EFTA has 29 Free Trade Agreements (FTAs) with 40 countries and territories outside the EU. Because these countries operate in the single market, this allows free movement of goods and services across all of the EEA.

Source: European Environment Agency (EEA).
All medical devices sold in the EU require a CE mark. While a CE mark is not required for items such as chemicals and pharmaceuticals, it can be required for combination devices and medical device software. For these two situations, how do you know if your product requires a CE mark?
To continue reading this ebook, including an overview of CE mark costs, and the associated technical documentation/general safety and performance requirements (GSPRs) that manufacturers are required to maintain please register to download the full version

FDA Class II medical devices
What are medical device classes within the FDA?
In the United States, the Food and Drug Administration (FDA) categorizes medical devices into three classes. They base these classes largely on the level of perceived risk the device may have. The level of perceived risk determines the data requirements and controls that need to be put into place to ensure safety for patients and/or users.
The FDA determines the level of oversight necessary for each device based on three factors. According to the FDA’s guidance on how to classify your device, device classification depends on the intended use of the device and also upon indications for use. The third and most important criterion is the device's risk to patients and users. The higher the risk associated with the device, the higher the class of that device will be, i.e Class I devices represent the lowest risk, and Class III devices pose the highest risk.
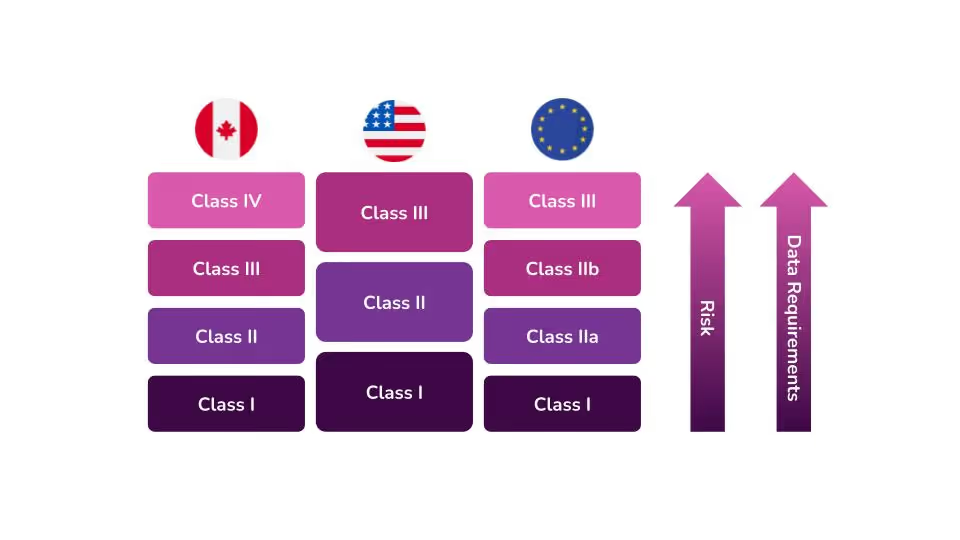
What are Class II Medical Devices?
Class II medical devices, which pose a medium to high risk to patients and users, account for 43 percent of all medical devices in the United States. Some common examples of Class II devices are syringes, pregnancy test kits, electric wheelchairs, and catheters. Class II medical devices must adhere to the provisions of the General Controls as mandated by the Food, Drug, and Cosmetic (FD&C) Act, which applies to all classes of devices.
The FDA also has its own Product Classification Database that can be used to assist in your device’s classification, finding consensus standards and submission types, such as a 510(k) or PMA.
What is the approval process for Class II Medical Devices?
All non-exempt Class II medical devices must go through the premarket notification process, also known as the 510(k). The 510(k) is a premarket submission process used to demonstrate that a device is safe and effective based on its substantial equivalence to a device already on the market, known as a predicate device.
Determining the substantial equivalence of your new device boils down to two things:
- Establishing that your device has the same intended use as the legally marketed predicate device
- Establishing that any technological differences between your device and the predicate device have no negative impact on the effectiveness and safety of the device
Any device with no identified predicate device, including those with a lower risk profile, is automatically classified as a Class III device and must use the more rigorous premarket approval (PMA) submission to receive market approval. Lower-risk devices can request reclassification, however, through a De Novo submission.
Class II medical devices in other countries
Device classification is different in each country. With that in mind, you should not make any assumptions regarding classification in other countries because your device is a Class II device in the United States. Each country with medical device regulations has its own classification scheme that may cause your device to be regulated differently.
During the initial phase of planning for the global commercialization of a product, it is imperative to consider international regulations, their classification schemes, and the registrations that each country will require.
The process of getting a Class II medical device to the market is arduous, and regulatory professionals must navigate disjointed manual data systems and processes. Plus, ever-evolving regulations make it difficult to ensure your new products, and even ones already on the market, are compliant.
Regulatory information management (RIM) software is an almost invaluable tool that can help your company get products to market more quickly and cost-effectively by digitizing and automating regulatory activities in a single system. The right RIM software can make the 510(k) process simpler and more efficient than ever before.
For more information on the 510(k) process, read our Beginner’s guide to the 510(k).

What's up with the duck?
If you’ve seen us online or at tradeshows, you might be wondering, “What’s up with the duck?” His name is Reggie by the way, and we’re pretty fond of him around here. Rimsys is more than software. It’s a team built from the ground up who understand the importance and challenge of managing regulatory affairs in the ever-evolving medical device industry. Our goal here at Rimsys is to help your company get its regulatory ducks in a row, and from that concept, “Reggie”, the regulatory duck was born.

If you could be a part of our company meetings, you’d see that we all love Reggie. After all, he’s more than a mascot to us. He symbolizes our mission to improve global health by accelerating delivery and increasing the availability of life-changing medical technologies. Reggie is our promise to our customers that you’re getting more than software with Rimsys. You’re getting a team of dedicated regulatory experts who understand the urgent need for better regulatory information and processes. That's why we love Reggie, our regulatory duck, and we believe you will too.

Brexit overview for medical device manufacturers
What is Brexit?
Brexit - meaning "British exit" is the process of the official withdrawal of the United Kingdom (UK) from the European Union (EU) on January 31, 2020, at 11:00 PM (GMT). The UK had been a member state of the EU since January 1, 1973, when it was known as the European Communities.
Leaving the EU was no easy task for the UK and it was six years in the making before it was final. When they “left” in January 2020, a discussion and negotiation transition period with the EU began. The negotiation process was both politically challenging and deeply isolating. Under the UK Prime Minister, Boris Johnson, the country continued to participate in many EU institutions during the one-year transition period in order to ensure frictionless trade until a long-term relationship could be agreed upon. Trade deal negotiations continued up until just days before the scheduled end of the transition period (December 31, 2020) and the EU–UK Trade and Cooperation Agreement was signed on December 30, 2020.
Following Brexit, EU law and the Court of Justice of the European Union no longer have power over British citizens and companies, with the exception of select areas related to Northern Ireland. Under the terms of the Brexit withdrawal agreement, Northern Ireland continues to participate in the European Single Market in relation to goods and to be a de facto member of the EU Customs Union.
The European Union Referendum Act 2015 set the wheels in motion. This Act of the Parliament of the United Kingdom made a legal provision for a referendum to be held on whether it should remain a member state of the European Union or leave it completely. The related legislation was introduced to the House of Commons in May of 2015, and it was passed on its third review in the House of Commons in early September 2015 and approved by the House of Lords in December 2015. The Act went into full legal force on February 1, 2016.
The initial withdrawal in March 2019 was delayed by the deadlock in the British parliament after the June 2017 general election. This deadlock then led to three extensions of the UK’s Article 50 of the Treaty of European Union (TEU), which began the member states’ withdrawal, known as Brexit.

Brexit’s effect on medical device regulations
All medical devices, including IVDs, custom-made devices, and systems or procedure packs must be registered with the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA) before they can be placed on the UK market. The MHRA is developing a new regulatory framework for medical devices, designed to safeguard and advance the health of its people by enabling early access to a high-quality supply of safe, effective, and innovative medical products. CE marked devices will initially be recognized in the UK, but manufacturers will have to obtain UK Conformity Assessment (UKCA) certification and follow the marking requirements per UK MDR 2002, beginning July 1, 2023. CE-marked devices will be allowed in the UK market until June 30, 2023, as long as they have been CE marked under one of the following:
- Directive 90/385/EEC - active implantable medical devices
- Directive 93/42/EEC
- Directive 98/79/EC - in vitro diagnostic medical devices
- Regulation 2017/745
- Regulation 2017/746
Medical device manufacturers continue to be able to self-certify CE marked devices until June 30, 2023 - providing the certificates remain valid for the EU market under the transitional timelines listed in the EU MDR and IVDR.
From January 1, 2022, non-UK manufacturers will require a UK Responsible Person for the purposes of registering devices. A Northern Ireland-based Authorized Representative will no longer be able to register devices on a manufacturer’s behalf for Great Britain.
You will need to use the new UKCA marking before January 1, 2023, if your product:
- will be marketed in Great Britain, and
- is covered by legislation that requires the UKCA marking, and
- requires mandatory third-party conformity assessment, and
- conformity assessment has been carried out by a UK conformity assessment body.
This does not apply to existing stock if your goods were fully manufactured, CE marked, and placed on the market before January 1, 2021.
Northern Ireland – the rules are different
In some circumstances, it is a requirement of the UK MDR 2002 that you inform the MHRA when you first place your device on the Northern Ireland market. Under the Northern Ireland Protocol, different rules apply than those in Great Britain. The precise requirements depend on the location of the manufacturer, the location of the Authorized Representative, and the device class. For more information on the regulatory system for medical devices in Northern Ireland, refer to the Regulation of medical devices in Northern Ireland.
The future of medical device regulations under Brexit
As you can see, the rules have changed and may continue to be refined until June 30, 2023. Currently, the CE marking is only valid in the UK for areas where both UK and EU rules remain the same. The UKCA marking is not recognized in the EU market, therefore, products will need the CE marking to sell in the EU. The UK MDR 2002 is up to date with all changes known to be in force on or before June 28, 2022. However, there may be changes that will be brought into force at a future date.
For more information on the EU MDR and IVDR requirements, read our Ultimate guide to the EU MDR/IVDR unique device identifier (UDI) system and Ultimate guide to the EU MDR GSPR - general safety and performance requirements.

The RegUP rundown
What is a RegUP?
The medical device industry is, by its very nature, a high-stress one. New medical technologies are being created all the time, and as a result, regulations are constantly changing. Being a regulatory professional in the medtech industry requires extraordinary attention to detail and nerves of steel. Rimsys was founded by regulatory pros for regulatory pros, and we understand the need to unwind in a professional environment with other leaders in the industry. That’s why we came up with the idea of the RegUP.
So, what’s a RegUP? It’s short for “Regulatory Meetup,” and it’s a small gathering of regulatory professionals, not a large conference. We’ll enjoy delicious food and beverages as we get to know each other and discuss regulatory trends and best practices.
When and where?
Boston - Sept 29th
Our first RegUP event will be held on the East Coast in Boston, MA, on September 29, 2022, at Democracy Brewing. Democracy Brewing, nestled in historic Downtown Crossing (one of the oldest neighborhoods in the United States), is the first cooperative brewery in Boston. Their menu features plenty of delicious craft beers and an eclectic selection of cuisines ranging from Mediterranean to classic American bar food.
San Jose - Oct 13th
Our second event will be held on the West Coast in the San Jose area on Oct 13, 2022, at Big Dog Vineyards. Big Dog Vineyards is a locally owned and operated winery in the picturesque Milpitas Hills, CA. Their Winery & Tasting Room opened in 2009, right beside the estate vineyard, which was planted in 1997.
What should you expect at a RegUP event?
As mentioned, we understand that regulatory professionals in the medical device industry are under constant pressure to ensure they keep up with ever-changing trends and best practices. And if you are like us, the opportunity to hone your trade and sharpen your skills and knowledge in a more casual setting will be welcome.
Here’s the agenda for RegUP Boston to give you an idea of what to expect at our events:
- 12:00 - 12:30 pm Welcome: Increasing confidence in regulatory planning. Many regulatory challenges are driven by a lack of visibility into regulatory timelines, resources, and needs. A comprehensive regulatory data strategy can help RA teams build more resilient plans, and shift their organizational posture from reactive to proactive.
- 12:30 - 1:15 pm Catered lunch
- 1:15 - 1:45 pm TBD Regulatory best practices case study
- 1:45 - 2:15 MDR/IVDR information essentials: MDR/IVDR compliance remains top of mind for RA teams. New information requirements including GSPRs, UDI/EUDAMED, and Post-market surveillance and reporting require a new approach to regulatory information management.
- 2:15 - 4:00 pm Brewery tour, tasting, and networking
Rimsys Speakers
- James Gianoutsos - Founder & CEO
- Bruce McKean - Director of Regulatory (Boston)
- Adam Price - Director of Product, Post Market (San Jose)
We hope to see you there!
Our RegUP events in Boston and San Jose will be the first of many opportunities for us to network, further regulatory knowledge and processes, and even enjoy each other’s company over tasty beverages. Best of all, these events are free for Rimsys customers and qualified guests.
Of course, you can also expect these events to get bigger and better with time. Nonetheless, you don’t want to miss out on the first of these opportunities to get to know us and our industry better. We hope to see you there! If you are unable to attend though, keep an eye on our LinkedIn page for future RegUP events in a city near you.
To get more information and register for our upcoming events, click the links below:
- RegUP Boston: https://regup-boston.eventbrite.com/
- RegUP San Jose: https://regup-sanjose.eventbrite.com/

IVDR: In Vitro Diagnostic Regulation within the European Union
What is the IVDR In Vitro Diagnostic Regulation?
The European Union’s In Vitro Diagnostic Regulation (IVDR) 2017/746 applies to IVD devices and came into force on May 26, 2022. The IVDR is a major change over the In Vitro Diagnostic Directive (IVDD), which it supersedes. The new regulation is designed to better protect patients and address changes in IVD technology through new requirements and stricter controls. As a Regulation, unlike a Directive, the IVDR becomes a requirement in every EU country on the same day, which means that compliance with the IVDR is paramount for market access within the EU.
Major changes in the IVDR
The IVDR includes changes to IVD classification, increased documentation requirements, and UDI specifications. It is critical for manufacturers and their European representatives to understand and implement the new regulations as soon as possible. There is significant concern that notified bodies will not have sufficient resources to handle the increased need for their services.
The major changes between the IVDR and its predecessor, IVDD, are as follows:
Changes to the classification system
Under the IVDD only a small percentage of IVD products were assigned a higher risk classification (based on a predefined list of products). All other products were exempt from notified body oversight and allowed for manufacturers to self-certify through conformity procedures. Under the IVDD, only 10%-20% of IVD products were subject to notified body oversight, whereas, under the IVDR, 80%-90% of IVD products will be subjected to notified body oversight.
Unlike the IVDD, the IVDR defines 4 risk classes based on risk profile:
- Class A: Lowest risk (e.g. specimen containers)
- Class B: Low to moderate risk (e.g. pregnancy tests)
- Class C: Moderate to high risk (e.g. cancer screening products)
- Class D: High risk (e.g. HIV tests)
It is important that IVD manufacturers re-classify their products using the new classification rules as soon as possible. Notified body oversight is required for IVDs in class B through class D.
Increased technical documentation requirements
Under the IVDD, technical documentation requirements were vague and left largely to the discretion of the manufacturer. The IVDR, however, details specific requirements for the content of technical documentation. Annex I details the General Safety and Performance Requirements, Annex II details primary technical documentation requirements, and Annex III details requirements for technical documentation for post-market surveillance.
Unique Device Identification (UDI) requirements
IVDs now require UDI labeling and registration in the European Union Database of Medical Devices, more commonly known as EUDAMED, in the same way as other medical devices. A UDI must be assigned to all medical devices, with some exceptions for custom-made and investigational devices.
Each UDI consists of multiple elements, including a Basic UDI-DI (also known as “BUDI”), a device identifier (DI) with static device information, and a product identifier (PI) with dynamic information provided by the manufacturer (such as serial number and manufacturing date). In addition, manufacturers can now register their device in EUDAMED, though this is not yet required.
Quality management system requirements
All IVD manufacturers are expected to have a quality management system in place, even though only those manufacturing class B to D devices require certification of conformity assessments by a notified body.
Person Responsible for Regulatory Compliance (PRRC)
Manufacturers will now be required to designate at least one person to monitor compliance with regulatory requirements. A PRRC must be qualified by way of a relevant degree and a minimum of one year of professional experience, or at least 4 years of experience in medical device regulatory affairs or quality management. Note that smaller manufacturers do have the option to subcontract their PRRC functions, but one person cannot act as a PRRC for a manufacturer based outside of the EU and for an Authorized Representative.
IVDR deadlines
Manufacturers who were able to self-declare conformity under the IVDD, but require a notified body under IVDR may continue to place IVDs on the market assuming they had issued a declaration of conformity prior to the original May 26, 2022 deadline, introduce no significant changes to the product, meet all post-market surveillance and vigilance requirements, and register in EUDAMED as specified in the IVDR. Assuming that these requirements are met, the new transition dates are as follows:
- May 26, 2022 - All new devices and non-sterile self-declared devices
- May 26, 2025 - Class D devices
- May 26, 2026 - Class C devices
- May 26, 2027 - Class B devices and Class A sterile devices
IVD regulations outside of the EU
This article is specific to requirements in the EU. It is important to note that other countries, such as the United States, have their own regulations related to In Vitro Diagnostics that are covered by the country’s medical device regulations. Each country with medical device regulations has its own classification scheme that may cause your device to be regulated in a different way. During the initial phase of planning for global commercialization of a product and throughout the product life cycle, it is imperative that you consider international regulations, their classification schemes, and the registrations that each country will require.
For additional information, The ultimate guide to EU MDR and IVDR general safety and performance requirements and also The Ultimate Guide to EU MDR/IVDR UDI.

CE marking guide for medical devices in the European Union
This article is an excerpt from the CE marking guide for medical devices in the European Union.
Table of Contents
- What is CE marking?
- Why is CE marking important?
- CE marking responsibilities
- What countries require or accept CE marking?
- Which medical devices require a CE mark?
- Technical documentation
- What are the costs associated with CE marking?
- How do you apply the CE marking?
- CE mark and UDI
- Does the CE mark expire?
- Do I need to CE mark my software?
- Final steps
CE marking is a symbol that consists of “CE, “ which is the abbreviation of the French phrase "Conformité Européene" meaning "European Conformity". The term initially used to describe “CE” was "EC Mark" but it has officially been replaced by "CE marking" according to the EU Directive 93/68/EEC. CE marking is used in all EU official documents, although you will still see "EC Mark" being used in common language. If you are using EC Mark in your documentation, you should change that terminology to CE marking in the future.
The letters ‘CE’ appear on many products traded on the Single Market in all the member states of the European Union plus Iceland, Liechtenstein, Norway and Switzerland. Simply put, The CE mark is a mandatory compliance mark, informing the consumer that the product is compliant with all applicable EU directives and regulations where the CE mark is required.
The Single Market was established in 1993 and is still considered one of the most significant achievements of the European Union. The main goal was to ensure the movement of goods and services freely within all the member states and to establish high safety standards for consumers. The CE mark indicates that goods and services do not need to be verified when shipping into another member country. To further support this movement, in April 2011, the Single Market Act was established to boost growth and strengthen confidence in the economy even further.
CE marking is required for many types of products, not just medical devices. The CE symbol can be found on bicycle helmets, toys, laptop batteries, wheelchairs, construction equipment, gas appliances and cell phone chargers - to name a few. CE marking is required for products manufactured anywhere that are sold in the EU, and only for those products for which EU specifications exist and require CE marking. The CE marking signifies that the product has been found to meet the general safety and performance requirements (GSPRs) of the European health, safety and environmental protection legislation and allows the product to be sold in the EU.
Manufacturer responsibilities for CE marking
Medical device manufacturers are responsible for properly and legally CE marking products before they leave the warehouse.
Most Class II and III medical devices, along with IVDs and some Class I devices, require a conformity assessment performed by a Notified Body to ensure that all legislative requirements are met before it can be placed on the market. Manufacturers of most Class I devices can self assess conformity. This process needs to demonstrate that all the legislative requirements are met, including any testing and inspections, and that all necessary certifications are obtained.
The European Commission lists 6 steps that manufactures should follow to affix a CE marking to their devices:
- Identify the applicable directive(s) and harmonized standards - see EU standards for Medical Devices, In Vitro Diagnostic (IVD) devices, and Implantable Medical Devices.
- Verify product specific requirements using the essential principles identified in the above standards.
- Identify whether an independent conformity assessment by a Notified Body is necessary. Notified bodies will be required to verify compliance with relevant Essential Requirements for most medical devices classified as IIa, IIb, or III - along with sterile class I devices. See the Notified and Designated Organization (NANDO) database for available notified bodies.
- Test the product and check its conformity.
- Create and keep available the required technical documentation.
- Affix the CE marking and create the EU Declaration of Conformity.
Importer responsibilities for CE marking
If you are importing medical devices into the EU, it is your responsibility to review all the technical documentation and maintain a copy, or to make sure that it’s available to you upon request.
You should verify:
- That the device has been CE marked and that the EU declaration of conformity has been completed.
- That the manufacturer has designated and established an authorized representative.
- That the device is labeled appropriately and contains instructions for use (IFU).
- When applicable, that a UDI has been assigned to the product.
- Whether or not the product is registered in EUDAMED (registration is currently voluntary).
Take action:
- List your name and address on the device or packaging, in addition to the manufacturer’s information.
- Keep records of complaints, non-conformities, recalls, etc. on file.
- Report any noticed non-conformity or product complaints from end users to the manufacturer and authorized representative immediately.
- Maintain a copy of the EU declaration of conformity and any other relevant certificates.
Distributor responsibilities for CE marking
If you are a distributor, you are responsible for reviewing the technical documentation provided to you so that you can verify the product is safe to put on the local market. You must also be sure the product is labeled correctly with the CE marking symbol clearly visible. The technical file documentation contains all of the information that is necessary to show conformity of the product to the applicable requirements.
You should verify:
- That the device has been CE marked and that the EU declaration of conformity has been completed.
- That the device includes all the appropriate labeling, including instructions for use.
- That if imported, the importer has complied with all the EU regulations.
- When applicable, that a UDI has been assigned to the product.
Take action:
- Report any noticed non-conformity to the manufacturer, importer, and authorized representative immediately.
- If a product appears to be out of compliance to the regulations and could pose a serious risk, the information should be reported to the Competent Authority, and to the manufacturer, importer and authorized representative.
- Any complaints or reports from end users about the product should be reported to the manufacturer and, if necessary, to the importer and authorized representative.
Important note: If the importer or distributor markets the product under their own company name, then they become responsible for CE marketing, and take over that role from the manufacturer.
CE marking is mandatory when importing products into the European Union, which is part of the larger European Economic Area (EEA). The EEA Agreement, established in 1992 and made official in 1994, is an international agreement that enables the extension of the European Union’s single market to non-EU members. It consists of the 27 EU countries plus the four European Free Trade Association (EFTA) countries - Iceland, Liechtenstein, Norway and Switzerland. Today, the EFTA has 29 Free Trade Agreements (FTAs) with 40 countries and territories outside the EU. Because these countries operate in the single market, this allows free movement of goods and services across all of the EEA.

Source: European Environment Agency (EEA).
All medical devices sold in the EU require a CE mark. While a CE mark is not required for items such as chemicals and pharmaceuticals, it can be required for combination devices and medical device software. For these two situations, how do you know if your product requires a CE mark?
To continue reading this ebook, including an overview of CE mark costs, and the associated technical documentation/general safety and performance requirements (GSPRs) that manufacturers are required to maintain please register to download the full version






