

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

An overview of 21 CFR Part 812: Investigational Device Exemptions (IDEs)
This document is a summary of the regulation and does include wording taken directly from the regulation itself. The original regulation should be referenced directly, however, for complete information when submitting an IDE request.
What is an investigational device exemption?
An investigational device exemption (IDE) allows a device to be used in a clinical study prior to obtaining market approval to collect safety and effectiveness data. Clinical studies are typically required to support a Premarket Authorization (PMA), but a small percentage of 510(k) applications also require clinical data when a predicate device comparison is inappropriate for the submission.
Before a clinical study is initiated, an investigational device must have an approved IDE, unless it is exempt. Devices may be exempt from IDE requirements if they are noninvasive diagnostic devices, being used for consumer preference testing unrelated to device safety or efficacy, or intended solely for veterinary use or research with laboratory animals. Refer to the full text of 21 CFR 812 for details and additional exemptions.
Part 812 – General Provisions (Subpart A)
Scope
This regulation is applicable to all clinical investigations of devices used to determine safety and effectiveness, except where exempt.
Applicability
Abbreviated Requirements
Investigations are considered automatically approved for IDEs if the device is not considered a significant risk, unless the FDA has specifically notified the sponsor otherwise. In these cases, the sponsor must still obtain IRB approval for the investigation and must comply with other requirements of Part 812, including proper labeling, record keeping, and conformed consent requirements.
Exempted investigations
IDEs are not required for devices that fall into one of the following categories:
- Devices that were in commercial distribution prior to May 28, 1976 that were used or investigated according to requirements in effect at that time. Devices that were introduced after May 28, 1976 but which have been found to be substantially equivalent to devices introduced earlier may also be exempt. This exemption is limited for Class II and III devices from the date an FDA regulation or order calls for the submission of a PMA (in the case of an unapproved Class III device) or establishes a performance standard for a Class II device.
- Diagnostic devices for which the testing is noninvasive and without significant risk, and is not used as a diagnostic procedure without confirmation through another product or procedure.
- Devices undergoing consumer preference testing, or testing of a device modification or combination of already distributed devices, if the testing is not for purposes of determining safety or efficacy and does not put subjects at risk.
- Devices solely for veterinary use.
- Devices intended for research on or with laboratory animals.
- A custom device that is not being used to determine safety or efficacy for commercial distribution.
Definitions
21 CFR Part 812.3 provides definitions for many terms, some of which are listed here.
Institution: A person, other than an individual, who engages in the conduct of research on subjects or in the delivery of medical services to individuals as a primary activity or as an adjunct to providing residential or custodial care to humans. For example, a hospital, retirement home, confinement facility, academic establishment, and device manufacturer. The term has the same meaning as “facility.”
Institutional Review Board (IRB): Any board, committee, or other group formally designated by an institution to review biomedical research involving subjects and established, operated, and functioning in conformance with part 56. The term has the same meaning as “institutional review committee.”
Investigational device: A device, including a transitional device, that is the object of an investigation.
Investigator. An individual who actually conducts a clinical investigation, i.e., under whose immediate direction the test article is administered or dispensed to, or used involving, a subject, or, in the event of an investigation conducted by a team of individuals, is the responsible leader of that team.
Monitor: When used as a noun, this term means an individual designated by a sponsor or contract research organization to oversee the progress of an investigation. The monitor may be an employee of a sponsor or a consultant to the sponsor, or an employee of or consultant to a contract research organization. Monitor, when used as a verb, means to oversee an investigation.
Significant risk device means an investigational device that:
- Is intended as an implant and presents a potential for serious risk to the health, safety, or welfare of a subject;
- Is purported or represented to be for a use in supporting or sustaining human life and presents a potential for serious risk to the health, safety, or welfare of a subject;
- Is for a use of substantial importance in diagnosing, curing, mitigating, or treating disease, or otherwise preventing impairment of human health and presents a potential for serious risk to the health, safety, or welfare of a subject; or
- Otherwise presents a potential for serious risk to the health, safety, or welfare of a subject.
Sponsor: A person who initiates, but who does not actually conduct the investigation, that is, the investigational device is administered, dispensed, or used under the immediate direction of another individual. A person other than an individual that uses one or more of its own employees to conduct an investigation that it has initiated is a sponsor, not a sponsor-investigator, and the employees are investigators.
Sponsor-investigator: An individual who both initiates and actually conducts, alone or with others, an investigation, that is, under whose immediate direction the investigational device is administered, dispensed, or used. The term does not include any person other than an individual. The obligations of a sponsor-investigator under this part include those of an investigator and those of a sponsor.
Subject: A human who participates in an investigation, either as an individual on whom or on whose specimen an investigational device is used or as a control.
Labeling of investigational devices
An investigational device or its packaging requires a label with the following information:
- Name and place of business of the manufacturer, packer, or distributor (per 801.1).
- Quantity of contents.
- Statement: “CAUTION—Investigational device. Limited by Federal (or United States) law to investigational use.”
- Any additional relevant contraindications, hazards, adverse effects, interfering substances or devices, warnings, and precautions.
- If used for animal research, a statement of “CAUTION—Device for investigational use in laboratory animals or other tests that do not involve human subjects” must be on the label.
IDE Labeling cannot have any statement that is false or misleading and cannot represent the device as safe nor effective.
Prohibition of promotion and other practices
An investigational device is intended for use only within an investigation. Therefore, until a device is approved for commercial distribution by the FDA, the sponsor or investigator (or those working on their behalf) cannot:
To continue reading this Regulatory Brief, please download the full document here.

The Real Cost of “We’ll Build It Ourselves”
If you are reading this from inside a large MedTech organization, you may be thinking: we have ten times the engineering staff. Why can’t we just build this ourselves?
We-Should-Just-Build-This-Ourse…
It is a fair question.
But software has a well-known paradox. Adding more people to a complex project does not make it go faster. It usually makes it go slower. More coordination. More handoffs. More meetings about meetings. More surface area for misalignment
A large IT organization is optimized for breadth — supporting dozens of systems, managing infrastructure, keeping the lights on across the enterprise
That is valuable work.
But it is fundamentally different from building and sustaining a deep vertical product over a decade.
The people on your team have day jobs. They run devices through regulatory pathways, manage quality systems, support manufacturing, and commercialize products globally
Building a regulated platform is not a side quest.
It is a second company
What the Numbers Actually Look Like
When people compare license fees to internal builds, they stop at the wrong baseline
The real comparison is:
Licensing a specialized platform
versus
Standing up and operating a regulated software company inside your enterprise
Product management.
UX research.
Engineering.
Regulatory SMEs.
Validation and QA.
Security operations.
Compliance programs.
24/7 support.
Infrastructure.
Multi-year modernization
AI makes some of that faster.
It does not make any of it optional
With a specialized vendor, that investment is amortized across an entire customer base.
With an internal build, the full long tail of ownership falls on you
And most of that spend ends up recreating the 80 percent that has already been solved — all because someone decided the remaining 20 percent justified building from scratch
The return on that 20 percent rarely survives honest scrutiny.
The Questions That Should Keep You Honest
It is easy to get excited about how fast something can be built.
The harder exercise is asking what happens in year three, year five, year eight
When your VP of Regulatory Affairs leaves, who maintains validation documentation?
When regulations change across jurisdictions simultaneously, who redesigns workflows and pushes a validated release before the deadline?
When an auditor asks for change control history and disaster recovery test results, who is accountable?
Internal initiatives often stumble not because engineers cannot prototype, but because sustaining them for a decade is brutally hard
Sponsors move on. Budgets change. Teams reorganize.
Regulatory systems do not get to pause.
They must remain inspection-ready through acquisitions, divestitures, and leadership turnover
Systems of record are commitments, not experiments
AI Changed the Tools, Not the Gravity
I am genuinely excited about what AI enables. It will reshape regulatory operations, reduce headcount growth, compress timelines, and raise expectations for every vendor in this space
What it has not done is repeal gravity.
Most of what AI replaces today is busy work. That is enormously valuable. But busy work was never the strategic bottleneck
The hard parts remain.
- Deciding submission strategy.
- Interpreting regulator feedback.
- Designing defensible workflows.
- Staying inspection-ready.
- Running global rollouts
Agents help teams move faster.
They do not decide what is safe, defensible, or durable
In MedTech, software is not just built.
It is designed, governed, operated, and defended
And gravity still applies.

Day Zero Is Easy. Day One Is Where It Gets Hard
There is something I keep coming back to in these conversations.
You can go from idea to prototype incredibly fast right now. That is the day-zero problem, and AI has essentially solved it. You can spit out working code, scaffold an integration, and stand up a proof of concept in a week
But the nuance around an actual business workflow — the day one and beyond activities — those are dramatically harder than day zero ever was
Software engineering done well is craftsmanship.
There is more to it than generating code and turning a prototype into something a regulated enterprise can depend on. It means thinking about edge cases, failure modes, upgrade paths, observability, and long-term operability. It means deleting as much as adding. Simplifying interfaces. Collapsing concepts down to what actually matters
Inside my own teams, I see impressive first versions all the time.
That is not the hard part anymore.
The hard part is everything that comes after
We-Should-Just-Build-This-Ourse…
Faster Engineering Just Pushes Work Somewhere Else
There is a tradeoff that rarely makes it into the first ROI spreadsheet.
AI compresses build cycles. In regulated companies, that speed shows up downstream. More releases mean more validation, more SOP updates, more training, more compliance review, and more audit prep
Engineering gets cheaper.
Governance becomes the constraint
There is also a subtler version of this problem.
Agents make it easy to generate output at scale. More workflows. More automation. More code.
But in regulated environments, every new service or automation path increases surface area. More things to secure. More things to validate. More things to explain to auditors
Speed without discipline creates complexity faster.
For CTOs, that is an architectural concern.
For Regulatory leaders, that is an inspection risk.
Are You Trying to Be a Software Company?
This is the part of these conversations that most often gets skipped.
A MedTech company is not a software shop. Most are largely outsourced IT organizations, and there is nothing wrong with that. The core business is devices, science, R&D, manufacturing quality, clinical programs, and global commercialization
When internal teams talk about building major regulatory platforms, the question is not whether they can spin up a prototype.
It is whether they want to operate a full-time software company inside their enterprise
Building software at scale is a people problem. It is not a technology problem. The constraint is coordination, judgment, institutional knowledge, and sustained focus over years
The people problem does not get fixed by agents and AI.
Regulatory platforms are deeply vertical. They encode jurisdiction-specific rules, regulator expectations, submission templates, QMS integrations, inspection trails, and post-market obligations
That knowledge is earned slowly.
It lives in product decisions, data models, operating procedures, and support playbooks.
AI will reshape how these platforms evolve.
It does not remove the learning curve that created them
AI Agents and the Confidence Shift Inside MedTech IT
In some MedTech IT planning meetings, a new kind of confidence has started to show up.
Not everywhere. Not in every organization. But often enough that it is worth paying attention to.
It is subtle. Casual. The kind that appears when something new begins to feel inevitable
A VP of IT or a CIO sits in a planning meeting. Someone pulls up a demo. An AI agent drafts a regulatory summary, generates a workflow, and scaffolds an integration. It looks impressive. It is impressive
Then someone says it:
Why are we paying for a platform when we could build this ourselves?
I understand the impulse.
SaaS valuations are volatile. Boards are pressing on efficiency. Hiring is under scrutiny everywhere. AI arrives, and suddenly there is a clean story. Automate friction. Avoid headcount growth. Modernize everything
Some of that is real.
I am optimistic about AI. In the right hands, it is a genuine superpower
But hope, cost pressure, aggressive marketing, and very human psychology are colliding right now. That collision is shaping how executives talk about technology strategy
In regulated industries, that matters.
The Confirmation Bias Problem
When leaders already feel pressure to reduce costs or flatten organizations, they naturally gravitate toward stories that validate those instincts. Flashy demos and headlines about agents replacing departments reinforce the belief that a breakthrough must be right around the corner
Once that belief sets in, messy operational details get discounted. Risk gets deferred.
That does not make the technology fake.
It does explain why ambition so often outruns delivery reality
For CTOs and Regulatory leaders, this is the moment to slow the conversation down.
Because prototypes are not platforms.
What AI Actually Changes
Years ago, Harvard Business Review wrote about the “hidden data factory,” the idea that organizations accumulate thousands of small one-off efforts to clean data, reconcile systems, patch workflows, and keep operations moving. No single fix ever justifies a major initiative. In aggregate, it quietly costs millions
That concept maps directly to what AI is good at today.
Inside engineering organizations, we call this work toil.
The repetitive, manual, low-judgment effort that keeps systems running but should not consume the time of highly trained people. Environment setup. Data reconciliation. Migration scripts. Test generation. Documentation drafts. Classification lookups. Compliance artifacts
AI is excellent at eliminating toil. It removes friction, collapses queues, and gives teams back time
In regulated environments, that is meaningful.
But here is the distinction that matters:
Eliminating toil does not eliminate accountability
It does not remove the need for architecture, UX design, validation strategy, regulatory interpretation, or operational ownership.
What it does is allow smaller, more senior teams to focus on the work that actually differentiates platforms.
That is very different than from saying agents replace the platforms themselves

Why MedTech Regulatory Teams Are Delegating EUDAMED to IT
And Why That Creates Bigger Problems Over Time
As EUDAMED implementation accelerates and the UDI/Devices module becomes mandatory in May of 2026, many MedTech companies have made a seemingly practical decision. They hand EUDAMED compliance to IT.
At first glance, the logic feels sound. EUDAMED is a system. It requires integrations, data transmission, and technical connectivity. IT already owns those capabilities, so the project lands there.
But this handoff reveals a deeper misunderstanding of what EUDAMED actually represents. It is a tool that enables manufacturers to meet ongoing regulatory obligations that touch product data, submissions, post-market activities, and lifecycle management. EUDAMED also enables manufacturers’ ACTOR partners like Notified Bodies, Authorized Representatives, Importers, and Distributors to meet their obligations under those EU regulations. Treating it as an isolated, one-time IT project creates risks to EU regulatory compliance that grow and spread across partners over time. MDR/IVDR regulatory compliance cannot be established and maintained with a one-time technical integration.
The first problem with delegating EUDAMED to IT is what it signals internally. It frames the regulation as a single event rather than a continuous program.
EUDAMED is not just about getting data into a database. It requires ongoing updates tied to regulatory changes, product modifications, vigilance activities, certificates, and market status. Every change across the product lifecycle can trigger downstream updates in EUDAMED.
When EUDAMED is positioned as a one-time event, organizations underestimate the scope, effort, and ownership required to maintain compliance over time. That gap does not show up immediately. It appears months later when updates are missed; data falls out of sync, or responsibilities become unclear.
IT teams often take on EUDAMED with the expectation that once the pipes are built, the work is largely done. In reality, the opposite happens.
As regulatory data changes, IT becomes the default escalation point for updates they do not own and cannot validate. They are asked to manage regulatory timelines, interpret data requirements, and support continuous updates that fall outside their core mandate.
This creates friction on both sides. Regulatory teams feel blocked by technical dependencies. IT teams feel burdened by compliance work they were never meant to manage. Over time, updates slow down, workarounds emerge, and risk quietly increases.
The most damaging consequence of delegating EUDAMED to IT is architectural. When EUDAMED operates outside of a centralized Regulatory Information Management system, organizations lose the opportunity to reuse data and reduce burden across the business.
Most of the data required for EUDAMED already exists within product information management and resource planning systems. Product registrations, certificates, submissions, UDI, and post-market data are not new. They are part of the regulatory lifecycle. When EUDAMED is disconnected from RIM, teams are forced to duplicate work, reconcile inconsistencies, and manually manage updates across systems.
Instead of becoming a natural extension of regulatory operations, EUDAMED turns into another silo. One that increases workload rather than streamlining it.
Establishing and maintaining regulatory information in EUDAMED is a regulatory obligation, not a technical one. While IT plays a critical role in enablement and integration, there should be a strong partnership between regulatory and IT (or a third-party submitter), but IT shouldn’t own it completely.
When EUDAMED is managed as part of a centralized RIM approach, organizations gain consistency, traceability, and reuse. Regulatory teams can leverage existing data, control updates at the source, and reduce the ripple effects of change across departments. IT supports the infrastructure, but regulatory owns the process.
This shift also changes how organizations think about compliance. Instead of reacting to EUDAMED as a standalone requirement, they treat it as part of a broader regulatory operating model that supports long-term compliance and growth.
Delegating EUDAMED to IT is rarely a conscious strategy. It is usually a symptom of fragmented regulatory operations and unclear ownership.
As MedTech companies scale globally and regulatory expectations continue to evolve, these handoffs become harder to sustain. EUDAMED exposes the cost of treating regulatory compliance as a series of isolated projects rather than an ongoing operational discipline.
The companies that navigate EUDAMED successfully are not the ones with the most complex integrations. They are the ones that anchor EUDAMED within regulatory operations, supported by centralized RIM systems that establish data consistency and reduce duplication, improve visibility, and spread the burden across the organization in a controlled way.

Agentic AI and the Future of Regulatory Operations
Why Regulatory Operations Is Ready for Agentic AI
Regulatory operations teams are under increasing pressure. Global regulatory complexity is rising, data volumes continue to grow, and teams are expected to move faster, often without additional headcount. At the same time, employee turnover and fragmented systems make it harder to maintain continuity and institutional knowledge.
As outlined in the RIM & AI Maturity in MedTech Executive Guide, many organizations are still operating with scattered regulatory data, reactive processes, and manual workflows. These conditions increase compliance risk and slow growth.
This environment has created the conditions where a more advanced form of AI can deliver meaningful value. That is where agentic AI comes into play, not as a replacement for regulatory expertise, but as a way to strengthen how regulatory operations function day to day.
What Is Agentic AI and Why It Matters
Most AI used in regulatory environments today is assistive. It helps classify documents, extract text, or answer questions when prompted. Agentic AI goes further by operating within defined workflows and processes.
Agentic AI systems can monitor structured regulatory data continuously, identify upcoming risks or deadlines, recommend actions based on rules and historical context, and surface next steps within governed processes. Instead of responding to requests, agentic AI supports execution by working alongside regulatory teams inside their operational systems.
The distinction is important. In regulated environments, value does not come from generative output alone. It comes from intelligence that is embedded, auditable, and aligned with how regulatory work actually gets done.
Moving Regulatory Teams Off the Data Treadmill
The executive guide describes early-stage regulatory teams as being stuck on a back-office data treadmill. Highly skilled professionals spend a disproportionate amount of time searching for information, reconciling spreadsheets, and repeating manual tasks rather than applying their expertise strategically.
Agentic AI helps reduce this burden by continuously organizing and validating regulatory data, identifying missing metadata or inconsistencies early, and reducing reliance on individual memory or tribal knowledge. Over time, this improves not just efficiency, but operational resilience. Teams become less vulnerable to audits, turnover, and last-minute regulatory surprises.
Why Agentic AI Depends on Operational Maturity
One of the most important insights from the paper is that AI value scales with RIM maturity. Advanced AI capabilities are not effective without centralized regulatory information and standardized processes .
At higher maturity levels, AI can surface upcoming risks across markets and renewals, analyze submission history to recommend reusable content, and identify bottlenecks before they impact timelines. At this stage, agentic AI begins to function as an operational partner, helping teams anticipate issues rather than react to them.
This is also where many organizations encounter friction. Skipping foundational steps may create the appearance of progress, but it limits reliability and long-term impact. Agentic AI is only as effective as the data, governance, and workflows it operates within.
From Task Automation to Predictive Compliance
At the most mature stage of regulatory operations, AI becomes fully embedded in daily work. The guide describes this level as one where real-time monitoring, predictive analytics, and continuous improvement are standard practice .
In this environment, agentic AI supports predictive compliance by identifying emerging risks, highlighting resource constraints, and improving visibility across submissions and renewals. These insights allow teams to act earlier and with greater confidence.
The paper is clear on one point. AI enhances regulatory expertise, but it does not replace it. Human judgment remains essential for interpretation, decision-making, and accountability. The real value of agentic AI is that it frees regulatory professionals from low-value work so they can focus on the decisions that matter most .
Regulatory Operations as the Heart of Compliant Growth
The most significant impact of agentic AI is not automation alone. It is the elevation of regulatory operations from a reactive support function to the heart of compliant growth.
Organizations that invest in strong RIM foundations, data governance, and workflow integration are better positioned to apply AI in a way that is safe, scalable, and durable. When implemented thoughtfully, agentic AI helps regulatory operations keep pace with growth, reduce risk, and support faster, more confident decision-making across the business.
The Future of MedTech Compliance: How AI Is Transforming Regulatory Affairs
MedTech regulatory affairs teams are facing a turning point. Regulations are expanding in number and complexity, resources are limited, and manual processes cannot keep up. At the same time, artificial intelligence (AI) has become a serious topic of discussion in regulatory circles. Leaders are beginning to ask: How can AI help us manage change, reduce risk, and accelerate compliance efforts?
The answer is clear: AI is no longer just a buzzword. When combined with effective regulatory information management (RIM), it can be a powerful enabler of efficiency, accuracy, and strategic decision-making.
Why AI is Trending in Regulatory Affairs
The Surge of Regulatory Data
Regulatory teams must now track requirements from multiple global markets. Each regulator frequently updates its regulations, guidances, templates, and recognized standards, which creates large volumes of data to organize and analyze. AI can scan and classify this information, highlight changes, and prepare it for structured use within RIM systems.
Doing More with Limited Resources
Most teams are expected to deliver more without additional staff. High turnover makes continuity difficult, and according to the 2024 RAPS Global Workforce Report, the number of professionals “open to work” has grown in North America and Europe. AI offers relief by taking on repetitive tasks such as document formatting or data entry, allowing experts to focus on higher-value work.
Global Complexity and Diverging Standards
No two markets are exactly alike. AI can help by flagging differences, surfacing potential risks, and recommending reusable content drawn from a company’s submission history. Faster, more accurate submissions directly improve time-to-market and compliance outcomes.
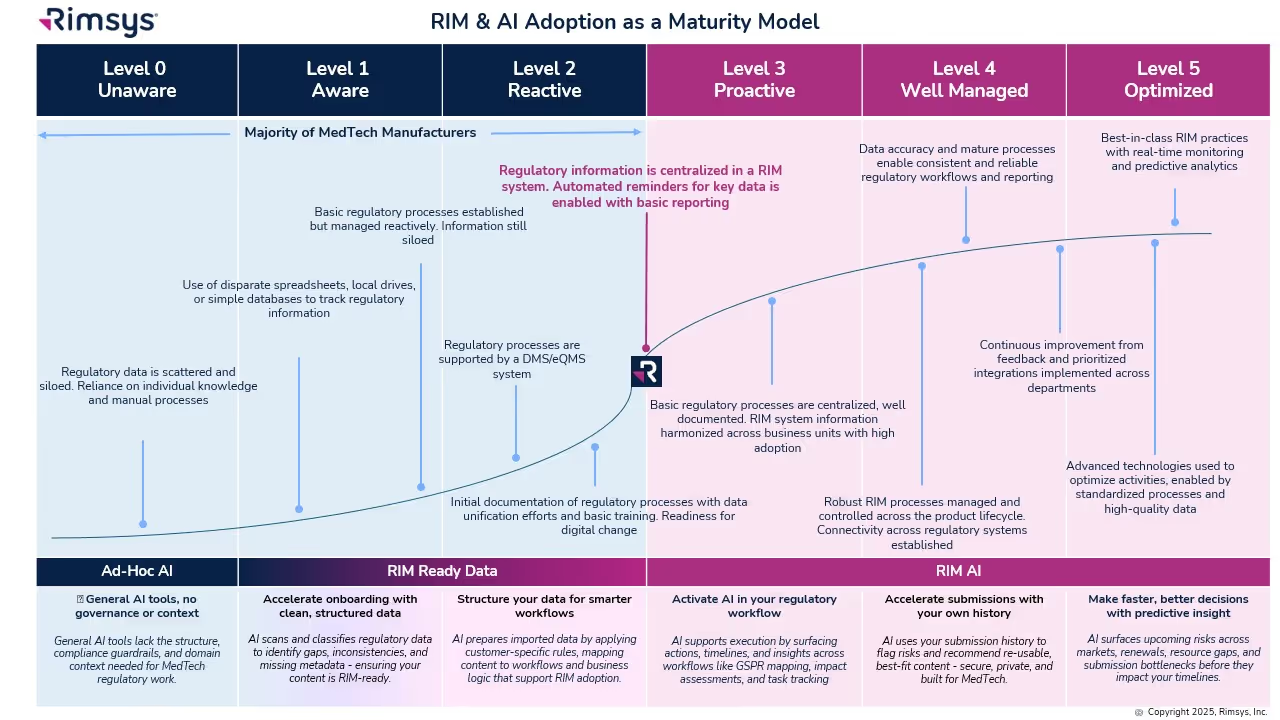
The RIM & AI Adoption Maturity Model
Not every organization is ready to fully embrace AI. Success depends on RIM maturity: how structured and centralized your regulatory processes and data are. The RIM & AI Adoption Maturity Model provides a roadmap from basic to optimized states.
- Levels 0–2: Early Stage
- Data is siloed and processes are ad hoc. AI provides value in isolated ways, such as cleansing records or scanning for regulatory changes.
- Level 3: Proactive
- A RIM system centralizes information. AI begins to surface reminders, deadlines, and global impact assessments.
- Level 4: Well Managed
- Processes are standardized across the lifecycle. AI generates insights, monitors KPIs, and supports reuse of regulatory content.
- Level 5: Optimized
- AI is fully embedded, delivering predictive analytics, continuous monitoring, and smarter decision-making.

Practical Applications of AI Today
Today, regulatory teams see the greatest opportunities in:
- Regulatory submissions: Automatically detecting changes in templates and suggesting updates.
- Document classification: Using natural language processing to tag and organize regulatory documents.
- Regulatory intelligence: Monitoring health authority updates and highlighting what matters most.
- Impact assessments: Linking changes (e.g., regulations/standards/design) directly to the affected products and registrations and evaluate the potential impact.
- Content reuse: Recommending approved content to accelerate submissions.
How to Start Your AI Journey in Regulatory Affairs
Adopting AI is not about jumping to the most advanced capabilities overnight. Instead, consider these steps:
- Assess your RIM maturity. Where does your organization sit on the 0–5 scale? What foundational gaps (data centralization, process standardization) need to be addressed first?
- Identify quick wins. Focus on repetitive, rules-based tasks where AI can add value without major disruption.
- Implement governance. Establish policies for safe, compliant AI use, particularly around data privacy and model training.
- Pilot in phases. Start small, validate results, and expand AI use as confidence and maturity grow.
- Keep people at the center. AI should enhance the expertise of regulatory professionals, not replace it.
Building a Smarter Future for MedTech Compliance
AI is becoming a trending topic in regulatory affairs not just because it’s new, but because it directly addresses the challenges teams face: rising complexity, limited resources, and scattered data.
For organizations that take this approach, the benefits are clear: lower compliance risk, faster execution, and stronger competitive positioning. AI does not replace regulatory professionals. Instead, it enables them to spend less time on manual tasks and more time on strategic contributions that improve patient access to life-changing technologies.
In other words, AI isn’t about futuristic transformation. It’s about helping regulatory teams step off the “data treadmill” and reclaim their time for what matters most: bringing safe, life-changing medical technologies to patients faster.






