eBooks
RIM & AI Maturity in MedTech: Executive Guide to Modernization Success
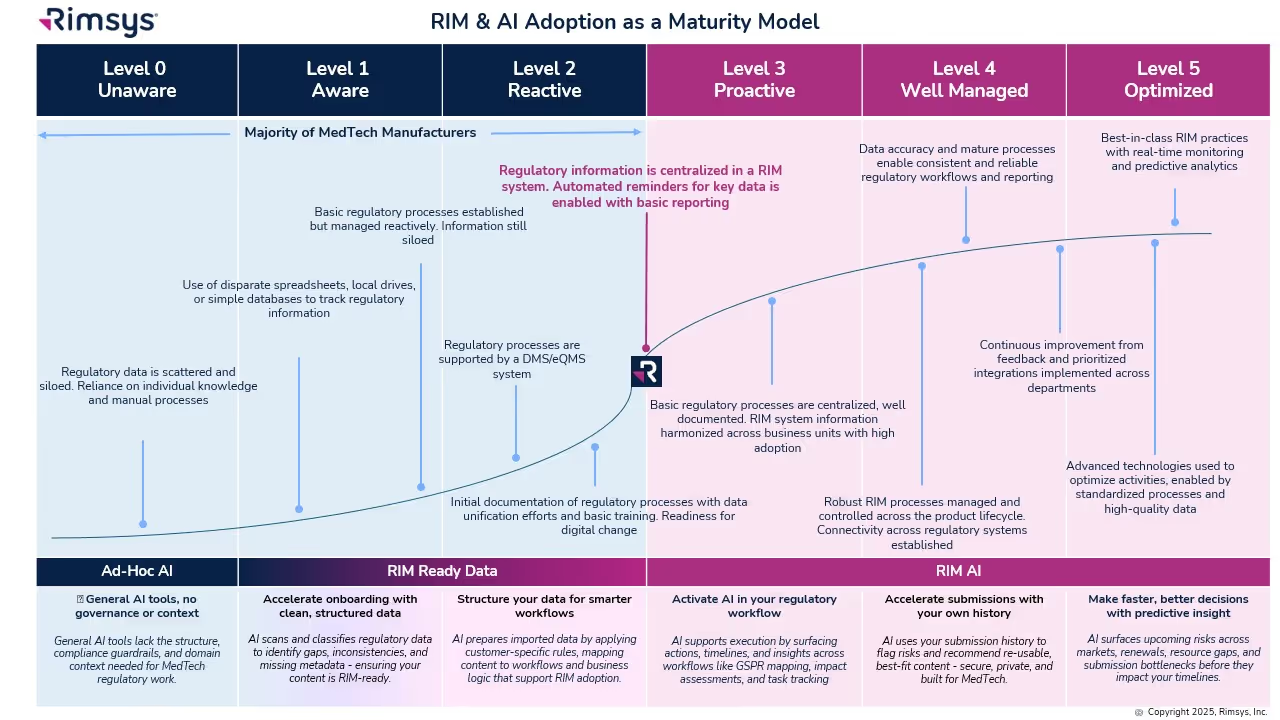
MedTech regulatory affairs teams are at a pivotal moment. The regulatory landscape is evolving quickly, with new regulations emerging and existing requirements changing, which increases complexity. Many teams are digitizing their processes and applying AI to manage regulatory information more effectively. These efforts help reduce compliance risks, improve efficiency, and create sustainable advantages in a competitive environment. However, realizing these benefits requires an understanding of RIM maturity. Teams must first know where their information resides and how it is managed. This executive guide introduces the RIM & AI Adoption Maturity Model, outlines key attributes, and shows how teams can progress.
Get the ebook
Here's some additional content you may find helpful

Nonconformance reporting for medical device manufacturers
Read more

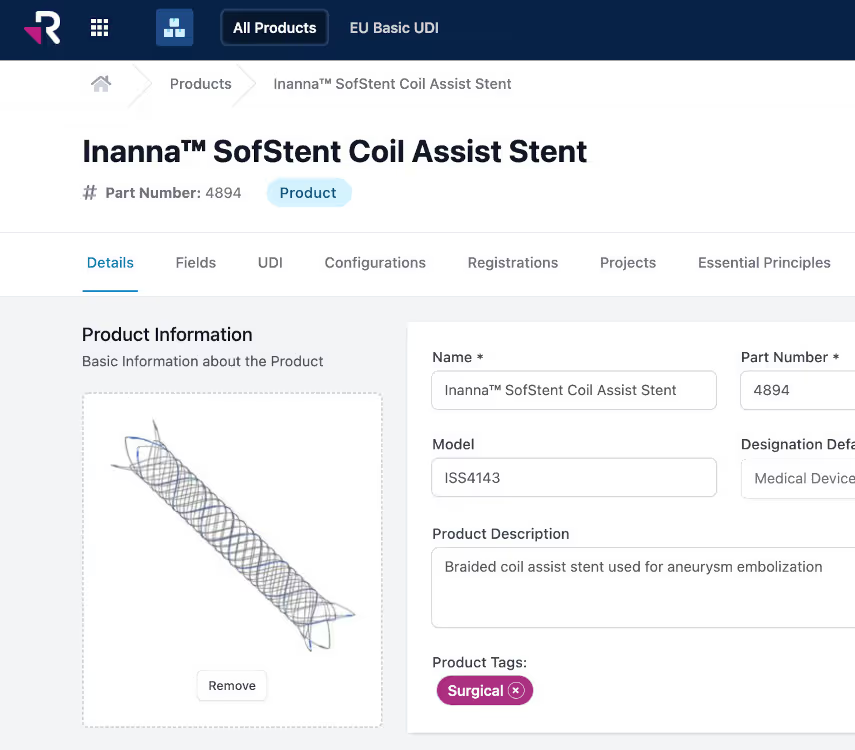
Why we developed Rimsys from the ground up
Read more


Your eSTAR submission questions answered by FDA experts
Read more


Ask us Anything ... about UDI!
Read more


Why should you invest in your regulatory team? Easy Medical Device podcast interview
Read


What is the FDA eSTAR program?
Read more


What's up with the duck?
Read more

%2520(1755%2520x%2520550%2520px)%2520(855%2520x%2520268%2520px).avif)
Why a RIM System is Critical to Successfully Support MedTech M&A Activities
Read more


The five guiding principles for machine learning-enabled medical devices using PCCPs
Read more


The ultimate guide to the EU MDR/IVDR UDI
Read more

%2520(1).avif)
Using EUDAMED as the Foundation for a Global UDI Strategy
Read


The ultimate guide to the EU MDR and IVDR general safety and performance requirements (GSPR)
Read more


Top 6 benefits of a regulatory information management (RIM) system for medical devices
Read more


To build or to buy: evaluating options for Regulatory Information Management
Read more


The RegUP rundown
Read more


The state of regulatory performance in 2023
Read more

The Future of MedTech Compliance: How AI Is Transforming Regulatory Affairs
Read more


The 510(k) application: if content is king, then communication is queen
Read more


Taking SaMDs to market in the US: How is the FDA regulating adaptive machine learning algorithms?

Read


Tackling regulatory continuity: How regulatory affairs teams can use technology to reduce the impact of employee turnover
Read more


Key steps to help you streamline regulatory process management
Read More


Software as a medical device (SAMD) - classification overview
Read more


Selecting a RIM system for your medical device company
Read more


STED is dead
Read more


Selecting and working with medtech regulatory approval consultants
Read more


SaaS 101 for medtech regulatory professionals
Read more


The role of regulatory affairs teams throughout the product lifecycle
Read more

Rimsys secures $1.5 million to accelerate growth of its leading medtech regulatory information management platform
Read more

.avif)
Rimsys secures $5 million in growth financing round
Read


Rimsys releases new automated unique device identification (UDI) module for its holistic medtech RIM platform
Read more


Rimsys ranks number 156 on the 2023 Inc. 5000
Read more


Rimsys POV: Updated EUDAMED timeline
Read more


Rimsys raises $16M Series A to bring regulatory order to the medtech industry
Read more


Rimsys joins MedTech Europe
Read more


Rimsys named 2020 top technology company finalist by Tech 50 awards
Read more

.avif)
Rimsys Enters Strategic Alliance Relationship with KPMG
Read More

.avif)
Rimsys Becomes the Trusted Regulatory Partner for 6 of the Top 12 Global MedTech Manufacturers
Read more

%2520(855%2520x%2520268%2520px).avif)
Rimsys Announces Bulk UDI Submission and Rimsys Connect™ to Empower MedTech Regulatory Teams
Read more


Rimsys announces new brand identity for world-leading regulatory information management (RIM) software
Read more


Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams

Read


Announcing the release of Rimsys 3.0: Rimsys Insight
Read more


RIM vs PLM software for medical device manufacturers
Read more


RIM vs ERP software for medical device companies
Read more


RIM vs eQMS software for medical device manufacturers
Read more


Regulatory information management (RIM) systems and organizational change management
Read more


RIM Readiness: What your medtech company needs before implementing a regulatory information management system
Read more


RIM 101: what is regulatory information management?
Read more


RIM for medtech vs. RIM for pharma
Read more


RIM - Master data management for RA teams
Read more


Your regulatory team needs dedicated regulatory software
Read more


RAPS wrap-up
Read more


Regulatory strategy as a competitive advantage
Read more


Regulatory should be a revenue function
Read more


Learn why UDI is relevant to regulatory affairs and how Rimsys can help medtech RA teams manage the growing complexities associated with UDI data.
Read more


Quick reference guide - global medical device UDI requirements and timelines
Read more


PSUR: Periodic safety update reports for medical device and in vitro diagnostic products
Read more


FDA PMA submission process: a beginner's guide
Read more


Post-market surveillance for medical devices in the European Union
Read more


Podcast – Streamlining the MDSAP device marketing authorization and facility registration process
Read more


The importance of PLM, eQMS, and RIM systems for medical device manufacturers
Read more


An overview of the UK Medicines and Healthcare Products Regulatory Agency (MHRA)
Read more

%2520(1755%2520x%2520550%2520px)%2520(855%2520x%2520268%2520px)%2520(1).avif)
Planning Your Enterprise’s UDI Strategy for EUDAMED and Beyond
Read More


On a mission to improve global health
Read more


An overview of 21 CFR Part 812
Read more


Oh No! How to recover lost medical device certificates
Read more


MedWatch: The FDA safety information and adverse event reporting program
Read more


Medical Devices: Comparing standards, regulations, directives, guidance, and laws
Read more


Medical device audits - preparation and responses
Read more


MDSAP - the ultimate guide to the medical device single audit program
Read more


Is a medical device accessory a medical device?
Read more


MDR transition example under the extension
Read more


MDSAP device marketing authorization and facility registration
Read more


ISO 14971: risk management for medical device manufacturers
Read more


Making the case for a RIM system
Read more


IVDR: In Vitro Diagnostic Regulation within the European Union
Read more


What Sets Rimsys Apart
View UDI demo


ISO 10993: Standards for the biologic evaluation of medical devices
Read more


Introducing impact surveys
Read more


Introducing project management and essential principle templates
Read more

.avif)
Introducing Rimsys Intel: A Free, Centralized Global Regulatory Intelligence Hub for Medtech
Read more

_thumbnail.avif)
Insights from the Gens and Associates Executive Podcast with James Gianoutsos
Read


Introducing government submission templates (i.e. 510k, STED, CSDT) and more!
Read more


GUDID: An overview of the FDA's Global Unique Device Identification Database
Read more


IMDRF: International Medical Device Regulatory Forum
Read more


IEC 62304: Standard for medical device software
Read more


Global strategy for Unique Device Identifier (UDI) data
Read more


FDA’s Final Rule on LDTs: What manufacturers need to know
Read


FDA transition plans for Covid-19-related medical devices
Read more


FDA 510(k) - a beginner's guide
Read more


FDA predicate devices
Read more


FDA medical device registration process - getting new products to market in the U.S.
Read more


FDA listed, cleared, approved, granted - what IS the difference?
Read more


FDA Class II medical devices
Read more

Oops! Something went wrong while submitting the form



.avif)



