

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

Regulatory Strategy as a Competitive Advantage
This article is an excerpt from the Regulatory strategy as a competitive advantage ebook.
Table of Contents
- The regulatory revenue opportunity
- Regulatory responsibilities
- Limitations of the "cost-center" approach to regulatory affairs
- Regulatory as a revenue function
- Competitive advantage #1: Faster time to market
- Competitive advantage #2: Cost avoidance
- Competitive advantage #3: Out-pacing competitors
- Why invest in regulatory/revenue alignment?
- Getting started - 3 steps to move towards a revenue-aligned RA team
It is well known that medical technology (medtech) companies are highly regulated, given the potential risks their products present. Understanding and complying with the complex regulations in each country is, therefore, a necessary part of marketing and selling medical devices. To realize any revenue from a medical device, it must not only demonstrate compliance with all applicable regulations, it must also receive and maintain market clearance from each country in which it is to be sold. No market clearance means no revenue. Given the key role regulatory compliance plays in revenue attainment, regulatory teams, tools, and processes present a significant opportunity for differentiation for organizations willing to invest in them.
For the majority of medtech companies, however, regulatory departments have traditionally been treated as operational cost centers, with departmental improvements focused on cost reduction and efficiency improvements. Limited investment in people and tools, and limited interest in digital transformation, have left regulatory teams across the medtech industry underfunded and under-resourced.
This has led to great resourcefulness within the RA community, where most members can point to heroes within their team who worked long hours to meet a submission deadline, headed off a disaster by uncovering a pending expiration, created ad-hoc systems to organize information and streamline communication between the RA and QA teams for smoother audits, or have otherwise gone above and beyond their typical responsibilities.
Regulatory teams, however, have the potential to be a revenue-driving competitive weapon for companies that are willing to look at them a little differently and invest in regulatory performance above regulatory cost-effectiveness. Well-supported regulatory teams can provide a true competitive advantage by providing the resources and direction to:
- Capture market share by being first to market with novel devices.
- Avoid lost revenue by effectively tracking and planning for registration renewals/updates.
- Out-pace competitors and grow market share by adapting to regulatory changes more quickly and taking advantage of competitors’ non-compliance or inability to enter a new market.
We believe we are entering a new era for regulatory affairs within the medtech industry. One in which RA teams have a seat at the table when go-to-market, competitive positioning, and strategic decisions are being made.
In the medtech industry, regulatory affairs (RA) teams have a broad range of responsibilities across the product lifecycle:
Premarket regulatory strategy
Obtaining market clearance for a new medical device is the primary activity typically attributed to RA teams. It is not unusual for a regulatory team to be given market entrance projects with little warning, but ideally, the RA team would be brought in as early as possible to contribute to go-to-market discussions.
Premarket regulatory strategy, at a minimum, involves:
- Determining the most appropriate pathway to market approval. For example, a 510(k) or PMA submission in the U.S.
- Working with quality, product, and other teams to gather information needed for market submission.
- Establishing communication with applicable regulatory bodies and third-party approved auditors.
- Compiling and submitting required documentation for market approval. This includes managing follow-up activities, questions, and requests for additional information throughout the approval process.
Forward-thinking organizations often look to bring in RA teams even earlier in the process. As regulatory experts, RA professionals can provide unique insight into product development plans. In consultation with R&D teams, can help to refine product strategies, and steer development in areas that will reduce regulatory hurdles when new products are ready to be commercialized.
Maintaining regulatory compliance for existing products
While the primary focus of regulatory teams is often considered to be new market submissions, the majority of their time is actually spent on maintaining compliance for products that are already in-market. Even in situations where market registrations do not expire, constant vigilance is required to ensure that devices remain compliant with current regulations. These efforts take a considerable time for a typical RA team because information is often spread across disparate systems, where it can be difficult to find and confirm.
Maintaining regulatory compliance for approved devices includes:
- Staying on top of changing standards and making changes as required to existing technical files and other documentation.
- Submitting appropriate documentation updates when there is a change made that could potentially affect the efficacy or safety of the product, such as a material switch or facility change.
- Understanding pending regulatory changes and proactively addressing any that have an impact on devices currently in-market.
- Tracking registration expirations and preparing for timely re-submissions to ensure there is no lapse in market clearance.
Post-market activity
Post-market surveillance and vigilance activities are required by most countries and should involve the cooperation of the quality and regulatory teams. Ensuring that changing post-market reporting requirements are understood and complied with is an important regulatory responsibility.
Regulatory teams typically play a role in:
- Post-market surveillance of adverse events, complaints, and any issues associated with a device in the field.
- Assembling and submitting any required periodic safety reports to country/regional health authorities.
- Post-market vigilance and reporting of serious events to the appropriate regulatory agencies.
- Any required communication with regulatory authorities regarding adverse events or concerning trends in product quality.
Ask any RA professional, and they are likely to tell you that they work long hours and are often scrambling to meet looming deadlines...
To continue reading this ebook, download the full version.

2023 Regulatory performance report
Today at Rimsys, we unveiled the 2023 MedTech Regulatory Performance Report, a new set of insights into the state of medtech regulatory affairs. Compiled based on interviews with 200 regulatory professionals and executives, the study provides a detailed look into how regulatory teams are staffed, their processes, the tools they use, and ultimately how they perform.
Why did we create this study? There were two driving factors behind the research. The first was a common theme that we heard from a number of our customers: Regulatory leaders don’t have clear data and benchmarks. They don’t necessarily know how long a new market submission should take, and how to plan for or assess the work of their teams. While other studies look at the medtech industry broadly or the state of the regulatory profession, this study tries to build a comprehensive resource for regulatory (and company) leaders.
The second factor was really for ourselves and the team at Rimsys. As a company building solutions specifically for medtech regulatory affairs, we wanted more insight into where companies were successful, where they struggled, and where we can add value.
What did we find? Regulatory teams perform a lot of hero work and rate themselves highly for their accomplishments. At the same time there is a lot of opportunity for process improvements, and companies that invest in digital transformation for regulatory affairs see better performance.
Regulatory professionals are superheroes
Regulatory teams are generally pretty small. Most companies have less than 10 full-time regulatory professionals. These small teams complete an enormous amount of work. Last year on average, RA teams completed 50 license renewals, 50 license updates, and 10 new market submissions. This is impressive output.
Digging a bit under the covers, we found that this output relied heavily on the support of external consultants. 90% of companies use consultants to keep pace with their regulatory workload. Front-line employees also struggle with burnout. They were much more likely to report feeling under-resourced than regulatory leaders.
But process problems persist
A lot of regulatory work remains extremely manual. 70% of regulatory teams spend half their time or more on repetitive administrative tasks. All of this manual work increases the frequency of errors and required rework. 61% of companies reported a major non-compliance incident in the past 2 years.
Manual work also makes it difficult to complete regulatory projects in a timely fashion. Teams completed a lot of projects, but each took a long time. Over half of all companies spend 4 months or more on license renewals, license updates, and new market submissions.
Moving regulatory affairs forward
As regulatory requirements become more complex, there’s a natural question about how teams will work moving forward. MDR & IVDR in Europe have significantly increased the regulatory workload required to bring and keep products on the market. Will organizations be able to keep pace with the same resources, tools, and processes?
No, and the performance report shows that medtech companies are investing to improve their regulatory capabilities. The majority of companies are planning to increase the sizes of their RA teams in 2023, and 40% expect to increase their investments in regulatory software. Companies are increasingly adopting specialized software to better support regulatory processes.
Dig into the survey results
The full survey results provide insights into more aspects of regulatory performance. They show that companies need to take a deeper look into their processes and how regulatory resources are allocated. There are two ways to learn more:
- Visit the survey page to see the full results (the survey whitepaper can be downloaded at no cost)
- Watch the recording of our webinar with PA Consulting. We discuss the survey results in more detail and share our regulatory predictions for 2023

A primer on medical device classification
While it may seem simple, medical device classification can be a challenging task for many medical device and IVD manufacturers. Device classes for specific regions and countries have a number of small variations, and each of those variations can impact the process by which a device obtains market clearance. Getting it wrong can lead to delays in getting to market. This article explores the classification systems for three major markets, and their associated regulations.
An important component of achieving regulatory approval is the appropriate classification of a medical device or in vitro diagnostic device, according to the specific regulations within a country or region. Product classifications are related to the intended use of the product and the perceived risk that it poses to a patient using the device. While this general approach is pretty standard across all regions, there are many small variations in different country classification systems that can impact how a device is regulated. It would be much easier if there was one global classification system that everyone followed.
However, since there are different guidelines to classifying a medical device (per country), we’ll dig into the most popular classification systems—Canada, the European Union and the United States. These three are globally perceived to have strong, thorough, and trusted quality and regulatory systems. Their approaches are often mirrored or used as proxies for market clearance in other countries.
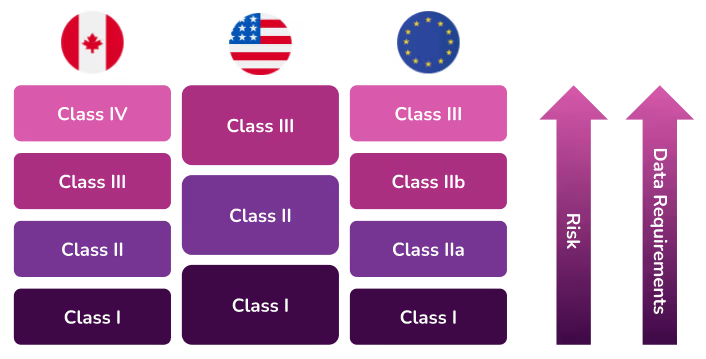
Canada
The Canadian Medical Devices Regulations include guidelines that classify devices into four risk classes. If a medical device can be classified into more than one class, the class representing the higher risk always applies.
- Class I devices do not require a medical device licence to be sold in Canada, but manufacturers, distributors and importers of these devices are required to obtain an establishment licence.
- Class II requires a medical device licence
- Class III requires a medical device licence
- Class IV requires a medical device licence
Some examples of different classes of devices in Canada include:
European Union
One of the main changes introduced with the new MDR/IVDR regulations are new classification rules for medical devices and in vitro diagnostic devices. If you have gone through the process of getting your medical device in the European market before, you might find it more difficult with the new EU MDR rules. For example, a new medical device you want to bring to market might now fall into a higher classification than it would have previously (under the MDD), and therefore require more testing, updates to documents, quality approvals, etc.
The new EU MDR brings the classification of medical devices in Europe more in line with international regulations, specifically the United States. These updated rules are listed in MDR 2017/745 for devices and IVDR 2017/746 for in vitro diagnostic products. As with Canada, if a medical device can be classified into more than one class, the class representing the higher risk always applies.
The EU has recently released a new guidance document MCDG 2021-24 to assist device manufacturers with device classification questions.
Class I – this classification is for the lowest risk device. Most medical devices in this category do not require a conformity assessment from a Notified Body so instead, they can be self-assessed. However, manufacturers must still complete a Technical File as part of the approval process.According to MDCG 2019-15, there are three subclasses under Class I. Unlike most Class I devices, these will require the involvement of a Notified Body.
- Class Im: a product with a measuring function
- Class Is: a product that is sterile
- Class Ir: a product that is a reusable, surgical instrument
Class IIa – this classification is for a medium risk device. A conformity assessment by a Notified Body is required for this classification.
Class IIb – this classification is for medium-to-high risk devices. A conformity assessment by a Notified Body is required for this classification.
Class III – this classification is for the highest risk devices. A conformity assessment by a Notified Body is required for this classification.
Examples of different classes of devices in the European Union include:
In Vitro Diagnostic Devices:
Class A – this classification is for the lowest risk in vitro diagnostic devices. Most IVD devices in this category do not require a conformity assessment. Instead, they can be self-assessed.
Class B – this classification is for medium risk in vitro diagnostic devices. A conformity assessment by a Notified Body is required for this classification.
Class C – this classification is for medium-to-high risk in vitro diagnostic devices. A conformity assessment by a Notified Body is required for this classification.
Class D – this classification is for the highest risk in vitro diagnostic devices. A conformity assessment by a Notified Body is required for this classification.
Examples of the different in vitro diagnostic device classes in the European Union include:
United States
In the United States, the Food and Drug Administration (FDA) is responsible for overseeing the safety of medical devices. The FDA has established classifications for approximately 1,700 different types of devices and grouped them into 16 medical specialties referred to as panels. All three classes of devices are subject to General Controls which are the baseline requirements of the Food, Drug and Cosmetic (FD&C) Act.
- Class I – General Controls (with or without exemptions)
- Class II – General Controls and Special Controls (with or without exemptions)
- Class III – General Controls, Special Controls and Premarket Approval
You are permitted to classify your own medical device based upon the FDA guidance documents and set regulations. However, if you wish for the FDA to assist with establishing your classification you can submit a 513(g) Request for Information. Note that there is a user fee associated with a 513(g) Request.
The device class determines which type of premarketing submission/application is required for market clearance.
In some instances, you do have the opportunity to reclassify your product after it’s been released to the market. The regulatory class of a device type, as defined by the Federal Food, Drug and Cosmetic Act (FD&C Act), may be changed through petition to the FDA. This process is only applied to a device type though, not to an individual device.
Examples of medical device classification in the US include:
Getting classification correct
Medical device classification is simple in that each country and region generally follows the same classification approach, and complex in that minor differences can change how a device is classified across markets. Understanding how a device is classified is one of the critical first steps regulatory affairs teams need to take when entering a new market, as medical device class often determines the pathway to market.
For example, in the EU classification can mean the difference between self-certification and a required conformity assessment from a Notified Body. In the US, classification can mean the difference between a 510(k) or PMA process for market clearance. Getting classification correct can ensure a smoother and faster route to market.
To learn more about market clearance processes for medical devices in the US, check out the Beginner’s Guide to the 510(k).

MDSAP - the ultimate guide to the medical device single audit program
This article is an excerpt from The ultimate guide to the medical device single audit program (MDSAP) ebook.
Table of contents
- What is MDSAP?
- History of MDSAP
- Who is responsible for the MDSAP?
- How does an MDSAP audit work?
- Audit sequence
- You got a nonconformity – now what?
- What does an MDSAP audit cost?
- Why choose the MDSAP certification process?
- Potential disadvantages of the MDSAP
- Ready to participate? – Here’s how to get started
- Completing a successful MDSAP audit
The Medical Device Single Audit Program (MDSAP) was designed and developed to allow a single audit of a medical device manufacturer to be applied to all country markets whose regulatory authorities are members of the program. The MDSAP provides efficient and thorough coverage of the standard requirements for medical device manufacturer quality management systems, and requirements for regulatory purposes (ISO 13485:2016). In addition, there are specific requirements of each medical device regulatory authority participating in the MDSAP that must be met:
- Conformity Assessment Procedures of the Australian Therapeutic Goods (Medical Devices) Regulations (TG(MD)R Sch3)
- Brazilian Good Manufacturing Practices (RDC ANVISA 16)
- Medical Device Regulations of Health Canada (ISO 13485:2003)
- Japan Ordinance on Standards for Manufacturing Control and Quality Control of Medical Devices and In Vitro Diagnostic Reagents (MHLW Ministerial Ordinance No 169)
- Quality System Regulation (21 CFR Part 820), and specific requirements of medical device regulatory authorities participating in the MDSAP program.
This means that a report from a single MDSAP audit of a medical device manufacturer would be accepted as a substitute for routine inspections by all the member Regulatory Authorities (RAs) across the world. There are currently five participating Regulatory Authorities (RA) representing the following countries: Australia, Brazil, Canada, Japan and the USA.

In April, 2021, the RAs released an “Audit Approach” document (MDSAP AU P0002.006) that combines the formerly separate MDSAP Audit Model and Process Companion documents into a single guidance document. It includes guidance for assessing the conformity of each process and includes an audit sequence, instructions for auditing each specific process, and identifies links that highlight the interactions between the processes.
In March 2012 the US FDA announced that they had approved a final pilot guidance document “Guidance for Industry, Third Parties and Food and Drug Administration Staff: Medical Device ISO 13485:2003 Voluntary Audit Report Submission Pilot Program.” This allowed the owner or operator of a medical device manufacturing facility to be removed from FDA’s routine inspection work plan for 1 year upon completing a ISO 13485:2003 audit. This guidance document went into effect in June 2012, and was intended as an interim measure while a single audit program was being developed.
This pilot program was not very successful and few companies signed up because they did not see any advantage in participating. The manufacturer had to pay for a third party to inspect their facilities, generate a report, and share the inspection results back to the FDA. Many companies were reluctant to contract “someone else” to perform their inspection when they could easily wait for the FDA to conduct an inspection for free.
During its inaugural meeting in Singapore in 2012, the International Medical Device Regulators Forum (IMDRF) appointed a working group to develop a set of documents for a harmonized third-party auditor system. Hence, the “Medical Device Single Audit Program” (MDSAP) was formed. The concept was similar to the FDA’s original idea of creating a third-party auditor to help reduce their workload of performing regulatory audits of medical device manufacturers’ quality management systems. This new approach would consist of a single audit that would review regulatory QMS compliance, conducted by a third-party, who would later be called an Auditing Organization (AO).
From January 2014 to December 2016, five countries participated in a Medical Device Single Audit Program Pilot. In June 2017, a report was generated summarizing the outcomes of prospective “proof- of-concept” criteria established to confirm the success of the program. The outcomes are documented in the final MDSAP Pilot Report and recommended that the program become fully active and open to any manufacturer who requested this type of audit.
The governing body of the MDSAP is the Regulatory Authority Council (RAC), which is composed of two senior managers (and a few other staff members) from each participating RA. They are responsible for executive planning, strategic priorities, setting policy, and making decisions on behalf of the MDSAP International Consortium. The RAC also reviews and approves documents, procedures, work instructions, and more. The mission of the MDSAP International Consortium is to jointly leverage regulatory resources to manage an efficient, effective, and sustainable single audit program focused on the oversight of medical device manufacturers on a global scale.
Other international partners that are involved in the MDSAP include:
MDSAP Observers:
- European Union (EU)
- United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA)
- The World Health Organization (WHO) Prequalification of In Vitro Diagnostics (IVDs) Program
MDSAP Affiliate Members:
- Argentina’s National Administration of Drugs, Foods and Medical Devices (ANMAT)
- Republic of Korea’s Ministry of Food and Drug Safety
- Singapore’s Health Sciences Authority (HSA)
The observers and affiliate members are not the same as the participating member RA’s. The observers simply observe and/or contribute to RAC activities. Affiliate members, on the other hand, are interested in engaging in the MDSAP program and are subject to certain rules. They are only given access to a certain level of information about the manufacturers, audit dates, and information in audit reports.
They are also invited to attend sessions that are open to members, observers, and affiliates only.
Audits can also be conducted by MDSAP participating RAs at any time and for various reasons including:
- "For Cause" due to information obtained by the regulatory authority
- as a follow up to findings from a previous audit
- to confirm the effective implementation of the MDSAP requirements
The purpose of audits conducted by the RAs is to ensure appropriate oversight of the AOs MDSAP auditing activities. The AOs are appointed by the RAs and a list of the currently approved AO’s is published on the FDA website. Most AOs offer a broad range of management system certification services, beyond just medical devices. Manufacturers should verify that prospective AOs are clearly trained and perform MDSAP audits of medical devices.
AOs have the final word as to whether a manufacturer has met the requirements for the MDSAP during the execution of the audit and generation of the associated reports summarizing the results. MSDAP RAC participating RAs have the final decision regarding all development, implementation, maintenance, and expansion activities associated with the program.
Although an unannounced visit by an AO is rare, it can happen in circumstances where high-grade nonconformities have been detected.
To continue reading this eBook including a detailed look at the MDSAP audit process and grading, pros and cons of the approach, and how to get started please register to download the full version.

Rimsys joins MedTech Europe
This week Rimsys became an associate member of MedTech Europe, the European trade association representing the medtech industry, alongside some of the world’s largest medical technology companies. As a member of the organization, we’ll have visibility into regulatory changes across the region as they’re being formed, and the opportunity to shape how those regulations are implemented. This access will help us to ensure that the digitization and automation of regulatory processes remains in reach for all medtech companies, and that our software will be best positioned to help them.
What is MedTech Europe?
MedTech Europe is a trade association focused on making innovative medical technology available to more people, and making healthcare systems more sustainable. Members of the organization include medical technology companies—device and in vitro diagnostic manufacturers, industry service and technology providers, and country-level associations within the European region.
The organization serves as a trusted resource of medical technology information for the public, providing data and highlighting the value of new technologies, and for policy-makers and other key stakeholders. It publishes industry research and statistics, contributes to health-related policy and legislation, and works to expand patient and provider access to new, lifesaving technologies.
Why did we join?
At Rimsys, we’re passionate about the medical technology industry, and focused on providing technology solutions to help medtech companies strengthen regulatory compliance, and bring new products to market more quickly. The European region is a focal point for medical technology regulation, and the MDR/IVDR regulations that began to take effect this year will inform the approaches of health authorities around the world.
Regulatory digitization and automation happens both at the manufacturer level and the health authority level. MDR/IVDR brings unique device identification (UDI) requirements, and a centralized technology system for product and post market surveillance information (EUDAMED) to the market. Both of these elements are digital by nature, and can serve as end-points of automated processes run within the Rimsys Platform.
Earlier this year, we introduced a new approach to UDI, one that ties together market requirements, product information, registrations, and selling status in an integrated regulatory lifecycle. Rather than managing UDI data separately from other regulatory information and activities, companies can auto-populate compliant UDI information directly, and will soon be able to submit that information electronically to the EUDAMED database. As a MedTech Europe member, we can participate in working groups around these systems, and ensure that we have the insights and access that we need to develop and deliver features like this for our customers.
How does this benefit our customers?
For Rimsys customers, this is all about maximizing the value they can get from our holistic Regulatory Information Management (RIM) platform. Our platform is designed to support a wide range of regulatory processes, and many of these processes are shaped by the regulations themselves. As members of MedTech Europe, we’ll be involved in policy discussions as they happen, and can ensure that the updates we make and features that we add are in alignment with the latest regulations. Understanding how regional policy is evolving also allows us to share that intelligence in-product—alerting customers to relevant changes, and updating market entrance requirements.
The second key benefit is access. The automation of regulatory processes is most effective when it can be done end to end. This therefore includes interactions with health authorities. Government agencies, including the European Commission, are establishing digital channels for product registration, UDI, and clinical and post market surveillance data. Our goal at Rimsys is to provide as many direct integrations with these systems as possible. As MedTech Europe members, we’ll be part of the groups that advise policy-makers, and help to develop these systems. As a result, we can help to ensure that our integrations are as functional and effective as possible.
Learn more about automation and digitization for regulatory affairs
Whether you distribute medical products in the European Union or in other markets, there can be significant benefits associated with digitizing and automating regulatory processes including better information access, more cross-company collaboration, less manual administrative work, and faster time to market for new products. To learn some more, see the Top 6 Benefits of a RIM system, or contact us to request a custom demo.

FDA 510(k) - a beginner's guide
This article is an excerpt from The beginner's guide to the 510(k) ebook.
Table of Contents
- Introduction
- 510(k) basics
- Contents of a Traditional 510(k)
- 510(k) submission and timelines
- Other 510(k) forms
Congratulations! You have successfully developed a new medical device. Now you need to take it to market. In the United States, this often means submitting a 510(k). A 510(k) is a structured package of information about your device and its performance and safety that you submit to the Food and Drug Administration (FDA) for “clearance” before you can sell your device in the U.S. In order to receive clearance from the FDA, your 510(k) will need to demonstrate that your medical device is substantially equivalent to another legally marketed device (called a predicate device). The substantial equivalence approval process is a simple equation that looks something like this:

The 510(k) is generally the most efficient route to market clearance in the U.S. because you show your device is safe and effective based on this substantial equivalence standard, instead of needing to present more extensive clinical trial data.
There are three types of 510(k): Traditional, Abbreviated, and Special. This eBook will begin with a general overview of the 510(k) process, including its purpose and benefits. Next, we will explore the Traditional 510(k) and the sections and components required in depth. Finally, we will look at the Special and Abbreviated 510(k).
FDA: background and device oversight
Before we explain what a 510(k) is let’s first talk generally about the FDA and device oversight. The FDA is the U.S. governmental agency responsible for overseeing medical devices, drugs, food, and tobacco products. When it comes to medical devices, the FDA’s mission is to “protect the public health by ensuring the safety, efficacy, and security of…medical devices.” At the same time, the FDA also has an interest in “advancing public health by helping to speed innovations.” In other words, the FDA’s goal is to make sure devices are safe and effective for public use, while also ensuring that devices have a quick and efficient path to market.
In order to achieve this balance of safety and efficiency, the FDA has three different levels of oversight depending on the risk level of the device: (1) exempt from premarket submission, (2) Premarket Notification, also known as 510(k), and (3) Premarket Approval (PMA).
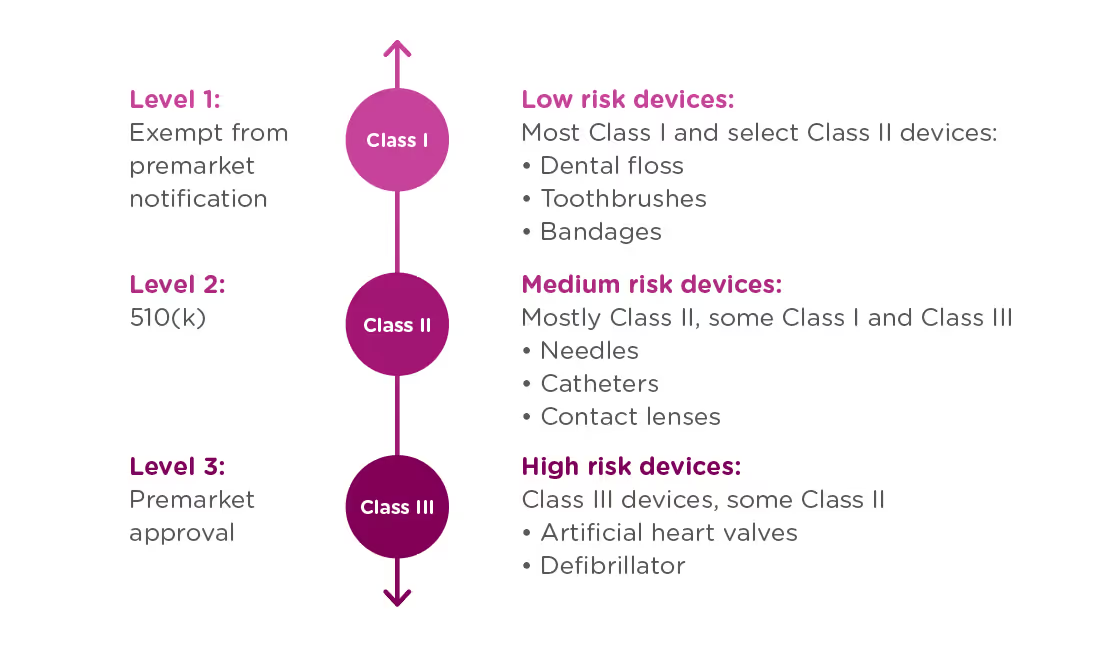
When is a 510(k) required?
A 510(k) is required for medium risk devices that have a predicate on the market which can be used to demonstrate the safety and effectiveness of the new device. Meanwhile, a PMA is required for high-risk or novel devices which require a higher level of scrutiny to be confirmed safe and effective.
A 510(k) is not only required for new devices, but also for devices that have been modified in a way that could impact safety or effectiveness. This could include changes to the:
- Design
- Components
- Materials
- Chemical composition
- Energy source
- Manufacturing process
- Intended use
You must submit your 510(k) at least 90 days before marketing the device.
What Exactly is Substantial Equivalence?
Now that we know what a 510(k) is, let’s talk about the substantial equivalence standard. You’ll recall from the introduction that your 510(k) must show that the new (or modified) device is substantially equivalent to at least one other legally marketed device, called a predicate device. Substantial equivalence looks at the intended use and the technological characteristics of the two devices.
More specifically, you must show:
- that the new device has the same intended use as the predicate, and
- the differences between the two devices do not raise questions about the safety and effectiveness of the new device.
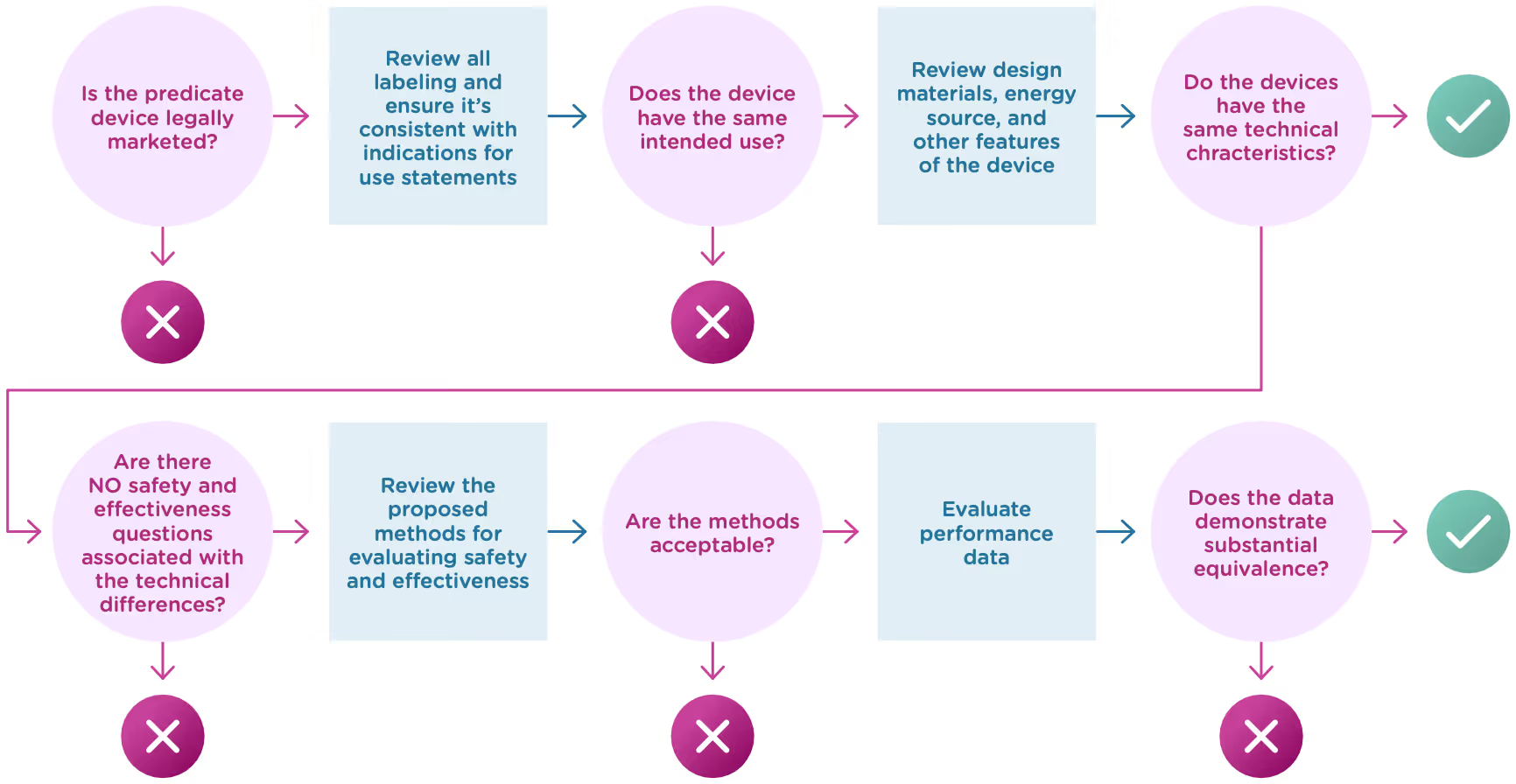
Now let’s take a closer look at intended use and technological characteristics.
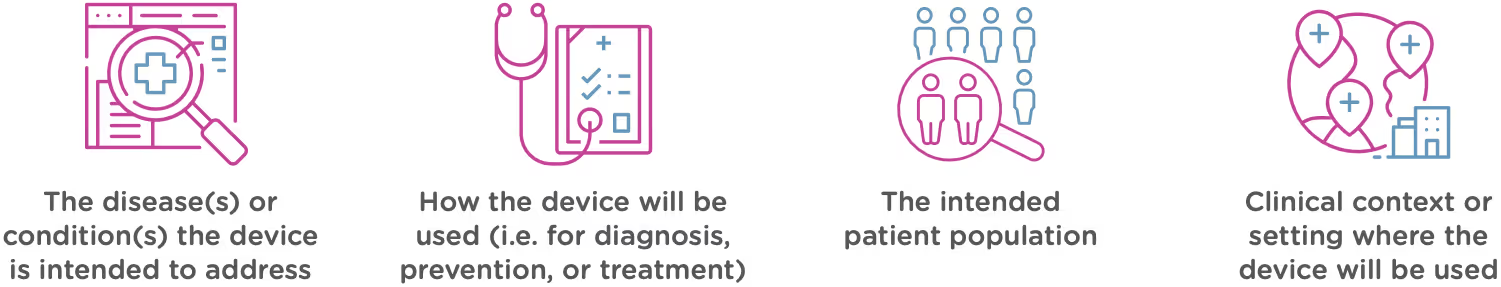
Intended use
Intended use means the general purpose or function of the device. The FDA will look at your proposed labelling and your Indications of Use section of the 510(k) to determine the intended use of your device (this is covered in Chapter 2). Intended use includes:
Technological characteristics
Once the FDA has determined that a predicate device exists and that the new device and the predicate device have the same intended use, it will move on to compare the technological characteristics. Technological characteristics include:
- Materials
- Design
- Energy source
- Other device features
The two devices do not have to be identical, and in fact they almost never are. The key here is to demonstrate that any differences do not have a significant impact on safety or effectiveness. Here’s what to cover when you compare your device’s technological characteristics with that of the predicate device:
Overall description of the device design
- Engineering drawings or diagrams to explain the device and component parts.
- List of component parts and explanation of how each component contributes to the overall use and function of the device.
- Physical specifications: dimensions, weight, temperature, tolerances, etc.
Materials
- Detailed chemical formulation used in all materials of constructions (especially those that come into contact with a patient).
- Any additives, coatings, paint, or surface modifications.
- How materials have been processed and what state they’re in.
Energy Sources
- Use of batteries, electricity, etc.
Other technological features
- Software/hardware
- Features
- Density
- Porosity
- Degradation characteristics
- Nature of reagents
- Principle of the assay method
In deciding whether the differences in technological characteristics impact safety or effectiveness, the FDA will typically rely on descriptive information about the technological characteristics as well as non-clinical and clinical performance data.
Let’s look at an example: A manufacturer submits a 510(k) for a new type of contact lens. Both the new device and the predicate device are indicated for daily wear for the treatment of astigmatism. The predicate device is only available in a clear lens, but the new device comes in a line of colors, including purple tinted lenses.

Who is responsible for submitting a 510(k)?
The following four types of organizations may be responsible for submitting a 510(k):
Manufacturers
- End-of-line device manufacturers who will be placing a device on the U.S. market.
- Note: Does not apply to component part manufacturers unless components will be marketed independently.
Specification developers
- Companies that develop the specifications for a finished device which has been manufactured elsewhere
Repackers or relabelers
- Required to submit a 510(k) if they significantly alter the labeling or condition of the device, including modification of manuals, changing the intended use, deleting or adding warnings, contraindications, sterilization status.
- Note: This is rare. The manufacturer, not the repackager or labeler, is typically responsible for the 510(k) submission.
Importers
- Importers that introduce a new device to the U.S. market may need to submit a 510(k), if it hasn’t already been submitted by the manufacturer.
Now that we’ve covered the basics, let’s explore what actually goes into your 510(k).
A Traditional 510(k) should contain all the following components in the list below. In some cases, a particular section may not apply to your device. When that happens, it’s a good idea to include the section anyway and just state “This section does not apply” or “N/A” under that heading.
To continue reading this eBook including a detailed walk-through of all the Traditional 510(k) components, submission requirements and timelines, and an overview of the other 510(k) forms including the Abbreviated 510(k) and the Special 510(k), please register to download the full version

EUDAMED and UDI in Europe: an overview
What is EUDAMED?
The European Databank on Medical Devices (EUDAMED) is an IT system developed by the European Commission to increase transparency and improve surveillance of medical devices (and in vitro diagnostics) that are sold within the European Union. It serves a key functional role in the unique device identification (UDI) requirements included in the MDR and IVDR regulations.
This article excerpts from The Ultimate Guide to the EU/IVDR UDI ebook to highlight the specific role and associated requirements that the EUDAMED database plays in the new regulations.
Element 4: The UDI database
The fourth component of the UDI system is entry of UDI and device information into the UDI database through EUDAMED. The MDR and IVDR require manufacturers to register all devices and submit specific information to the database before placing a device on the market. The core data elements provided through the UDI database will be accessible to the public free of charge. The list below outlines all of the required information for device registration.
Annex VI, Part A of 2017/745, states that the UDI database will contain all information about devices presently on the market and discontinued products. The database is designed to allow for linking across all packaging levels of the device. For all devices currently on the market, manufacturers are required to periodically verify the accuracy of the information in the database. If any changes are made to a device that do not require a new UDI-DI, manufacturers must still update the database within 30 days.
Information to be submitted with the device and economic operator registration must include
- Economic operator information (2017/745 Annex VI, Part A(1)):
- Information relating to the device
- Manufacturing information
The tables below outline all of the specific information that must be submitted to EUDAMED:
In addition to this information, the European Commission recently released the UDI HelpDesk. The HelpDesk will provide support to economic operators in the implementation of the obligations and requirements introduced by the new UDI system.
To learn more about EUDAMED and the European UDI system including a UDI overview, formats and issuing entities, implementation timelines, and key differences between the EU and US UDI systems, please register to download the full ebook.

Tackling regulatory continuity: How regulatory affairs teams can use technology to reduce the impact of employee turnover
Regulatory affairs is without a doubt a challenging function for all life sciences companies. In a recent survey of industry executives, 72% of them reported that regulatory affairs is one of their top 3 challenges. The pain is even more acute in the medical technology space. In the past few years medical device and in vitro diagnostic companies have had to contend with a host of new, more complex regulations for the European market (the new EU MDR/IVDR regime), as well as nascent medical device regulations in new markets around the world.
A significant additional challenge that medtech companies have to manage is that of continuity. Perhaps due to the challenges of the job, turnover among regulatory affairs (RA) professionals is surprisingly high. According to data from Zippia, RA professionals at all levels average less than 3 years in their jobs. This is echoed by the RAPS Global Compensation and Scope of Practice Report which showed that 63% of RA professionals have more than 7 years of regulatory experience, but 60% have been in their current jobs 3 years or less.
The limitations of outsourcing
One of the most common ways that medtech companies attempt to address continuity challenges is by employing external consultants. Consulting firms provide needed expertise, and especially bandwidth to resource-constrained RA teams. A recent survey by Grand View Research found that large medtech companies regularly outsource 50% or more of their regulatory activities.
Consultants are undoubtedly helpful, but the general approach to consulting engagements makes them less than ideal for a broad set of regulatory activities. Consulting engagements are inherently project-based. They generally have a specific scope, and a set of associated deliverables. However, medtech regulatory affairs is inherently product-based. Activities revolve around the product’s lifecycle—from market introduction to maintenance to withdrawal. A consulting firm may help to complete market applications for a new device in Latvia, Japan, and Brazil, but the company still needs to maintain a full post market surveillance regime, track standards in associated essential principles tables, and keep UDI information up to date as packaging permutations change.
At some point there’s a transition from the consultants back to the internal RA team, and the same continuity problem re-asserts itself. How to capture and disseminate the information, applications, and country certificates associated with a consulting project so that the internal team can move forward while maintaining compliance and market clearance?
"Poor-fit" tooling creates information silos
The lack of technology solutions designed specifically for medtech regulatory affairs is another driver of continuity issues. Regulatory activities necessitate the organization and management of large amounts of information. Detailed product specifications, performance and safety data, regulatory requirements, and relevant standards are just a few of the key pieces of information associated with each individual product and countries in which it’s sold. This is why RA professionals spend a huge amount of their time (up to 50%) simply looking for information.
Typically organizations manage regulatory information using spreadsheets or other general purpose tools. These tools, used to try and help manage information continuity, can actually create information silos. This happens for two reasons. One, the information that RA teams need for their activities is often broadly dispersed across the organization. Regulatory documents may be in a document management system (or just as often on individual employees’ computers), product and quality information is stored in PLM/QMS systems, and information about countries and selling status is contained within ERP systems. Spreadsheets don’t actually integrate with these sources, they just link. As information changes or is updated, it’s incumbent on employees to make manual updates to the spreadsheet.
Which brings us to the second problem. While there have been significant improvements in collaborative editing, documents like spreadsheets aren’t designed for the level of auditing and information management that regulatory teams need. Knowing whether an individual line item showing market status to be active, or a link to testing information in the QMS systems is up to date, still becomes “tribal knowledge” known only to the individual employees that make the update. If an employee leaves the company, all of that information goes with them.
How regulatory information management systems can improve continuity
The emergence of new technologies designed specifically for regulatory affairs activities has provided some new ways that medtech companies can tackle the continuity issue. Regulatory information management (RIM) systems provide a couple key capabilities that can increase the productivity of RA team members, and ensure that their work continues uninterrupted as team members come and go.
The first advantage of a RIM system is data association and integration. Rather than simply noting information, RIM systems can actively integrate with information sources, and automatically update as information changes. All of the information can be organized around individual products—the way that feels most intuitive to RA professionals. For any given product, an RA team member can quickly find country selling status, registrations, certificates, and expiration dates.
RIM systems can also eliminate a lot of the manual administrative work associated with regulatory activities. They can help streamline registrations with digitized authoring and publishing of regulatory submissions, and the creation and maintenance of digital essential principles tables. These capabilities are wrapped in a full set of project management tools, allowing teams to create workflows, approvals, timelines, etc. that match their internal processes and organizational goals. Individual records can be automatically updated such as the auto-population of UDI data when product details change, or bulk updates of essential principles tables when a standard is updated. These capabilities increase bandwidth across the RA team, and ensure that even new members can easily operate in accordance with the company’s processes.
RIM systems function as a “single source of truth” for RA teams. Because they work with all regulatory activities, they not only store information, but cross-link it in ways that make it instantly usable. RA team members can quickly pull regulatory information about products, product families, countries, regions, individual projects, eliminating the time spent searching for information. With enterprise integration capabilities, information can be pulled and shared with PLM, QMS, and ERP systems. This ensures that information is automatically updated, and key dependencies on regulatory activities are automatically linked. For example, clearance to enter a new market could automatically trigger a change in selling status in the ERP system. Having an organized, central source of regulatory data prevents information silos from forming, and ensures that nothing is lost when RA team members move on to other roles.
Finding the right RIM solution
Medtech regulatory complexity isn’t likely to abate any time soon, but there are steps that companies can take to better manage RA team bandwidth and continuity. Traditional approaches like outsourcing, or general productivity/information tools like spreadsheets are helpful, but they have limitations that can exacerbate continuity challenges. Modernizing the regulatory affairs toolset can be a good starting point. RIM systems with the right capabilities have the potential to increase RA team productivity, and eliminate information silos.
Key RIM features include support for a broad set of regulatory activities. Some tools focus on one aspect of RA like product registrations. As a result, they’re unlikely to effectively centralize and organize a broad set of regulatory information. While helpful from a productivity perspective, they can still lead to information silos for activities that aren’t digitized within the tool. Other RIM systems are primarily designed for the pharmaceutical industry. While they appear similar, regulations, and processes for medical technologies are different across the board. Tools that aren’t designed for medtech won’t feel as intuitive for employees, and likely won’t do as good a job at automating regulatory activities.
Rimsys provides a full-featured RIM system that is designed specifically for medtech companies, and supports a full range of regulatory activities. To see if the Rimsys Platform can improve your RA continuity, request a custom demo today.




.avif)
