

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

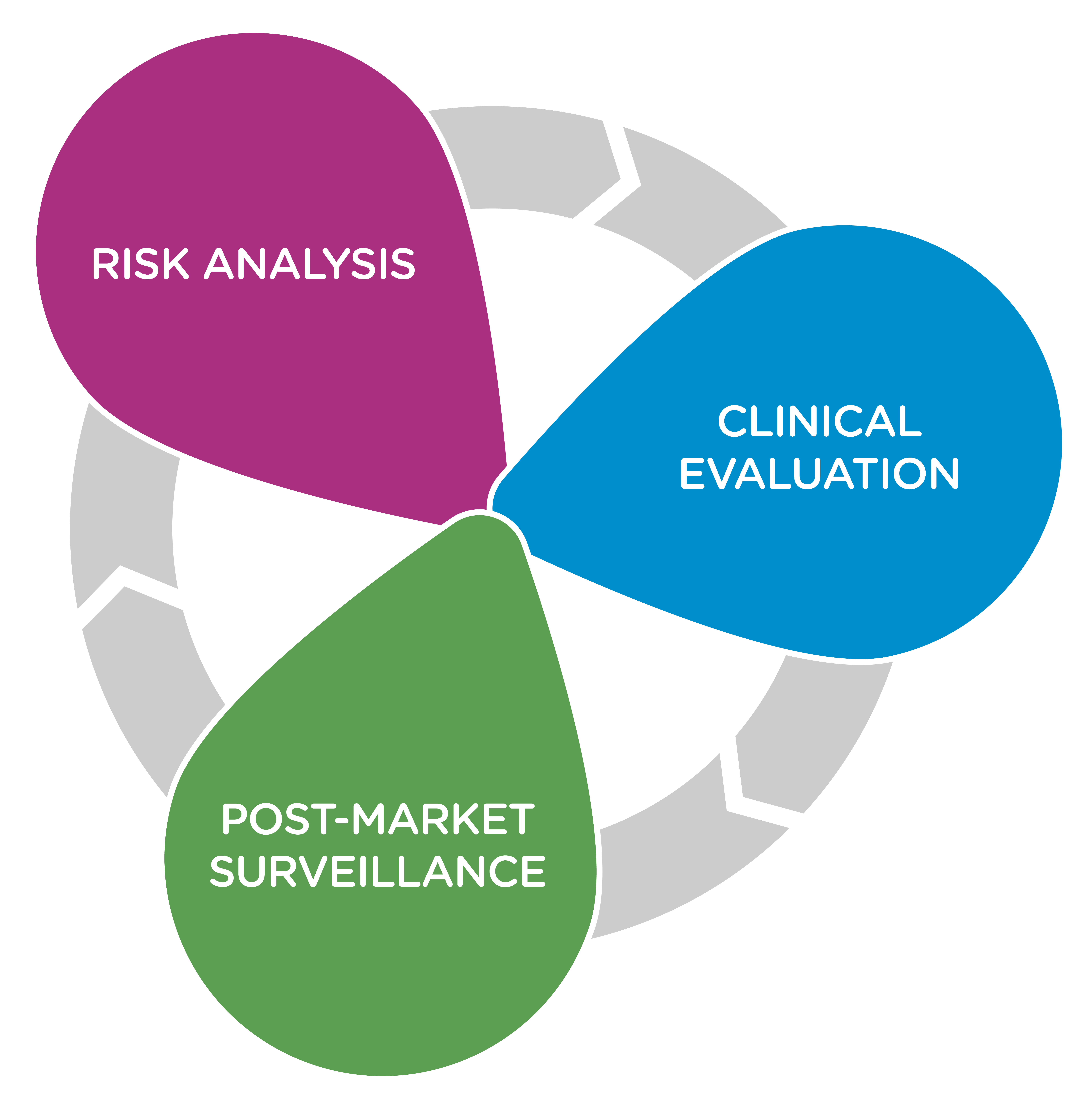
Post-market surveillance for medical devices in the European Union
This article is an excerpt from Post-market surveillance for medical device in the European Union.
Table of Contents
- What is post-market surveillance?
- What classes of medical devices require post-market surveillance?
- Components of a successful post-market surveillance plan
- PMS data requirements
- Post-market surveillance system goals
- Required post-market surveillance reporting
- Embracing post-market surveillance as an integral part of your quality program
- Getting started with post-market surveillance
Post-market surveillance (PMS) is designed to monitor the performance of a marketed medical device by collecting and analyzing field use data. Article 10 of the EU MDR and IVDR requires all device manufacturers to have a post-market surveillance system in place. The main elements of the PMS are laid out in Article 83, and additional details for lower-risk and higher-risk devices are covered in articles 84 and85, respectively.
In general, a PMS system consists of both proactive activities and reactive, or vigilance, activities. While post-market surveillance and vigilance are sometimes used interchangeably, vigilance consists of separate activities that feed post-market surveillance programs.
Post-market surveillance systems are used to collect and analyze data not only about the manufacturer’s device but also about related competitors’ devices that are on the market. Data collected through PMS procedures is then used to identify trends that may lead to, among other things, quality improvements, updates to user training and instructions for use, and identification of manufacturing issues.
Note that “market surveillance” encompasses activities performed by a Competent Authority to verify MDR compliance, and should not be confused with the topic of this ebook,“post-market surveillance," which is performed by the manufacturer.
All medical devices marketed in the EU require some level of post-market surveillance, and all medical device manufacturers must implement a post-market surveillance system (PMS). The requirements of the PMS, however, vary and should be “proportionate to the risk class and appropriate for the type of device” (MDR Chapter VII). In particular, the type and frequency of reporting vary based on a device’s risk class.
A post-market surveillance plan (PMS) is an integral part of a manufacturer’s quality management system and provides a system for compiling and analyzing data that is relevant to product quality, performance, and safety throughout the entire lifetime of a device. The PMS should also provide methods for determining the need for and implementing any preventative and corrective actions. A PMS system should include and define:
Surveillance data sources
With the increased focus on proactive risk identification in the MDR, it is important to design post-market surveillance systems that actively acquire knowledge and detect potential risks. It is not sufficient to rely solely on spontaneous reporting by healthcare providers, patients, and other stakeholders.
In addition to information coming from Clinical Evaluation Reports and complaint and adverse event reporting, typical sources of surveillance data include:
• Social media networks: Because many of your stakeholders may be communicating on social media networks, it is important to employ social listening techniques and/or tools to identify issues and concerning trends as they develop.
• Industry and academic literature: Any studies, academic papers, and other literature that addresses similar devices or the specific use cases for which your device is designed should be evaluated. In particular, risk factors and adverse events identified with similar devices should be closely examined. It is also important to identify newer technologies that may affect the benefit-risk ratio and establish a new definition of “state of the art” for the device type.
• EUDAMED: While the European Database on Medical Devices (EUDAMED) is not yet fully functional, it is intended to provide a living picture of the lifecycle of all medical devices marketed in the EU. Manufacturers should take special care to consider information for similar devices made available through the EUDAMED system in the future.
• Registries: Patient, disease, and device registries can provide information that informs the clinical evaluation process which provides input into the post-market surveillance system.
Data analysis methodology
A well-defined data analysis methodology will accurately identify trends and lead to defendable decisions in the application of post-market experience. Once the necessary information has been identified and collected, and potentially cleaned of incomplete or otherwise unusable data, the data needs to be analyzed.
The goal is to identify meaningful trends, correlations, variations, and patterns that can lead to improvements in the safety and efficacy of the device. There are many data analysis tools available that can assist with:
• Regression analysis that will identify correlations between data (e.g. the device location/geography correlates to battery life).
• Data visualization that can be useful in spotting trends in the data.
• Predictive analytics, which can be particularly useful with large data sets, to identify future trends based on historical data.
• Data mining, which is also normally used with large datasets, to organize data and identify data groups for further analysis.
Benefit-risk indicators and thresholds
The MDR requires that medical device manufacturers not only demonstrate the clinical benefit of their device but also quantify the benefit-risk ratio. The benefit of a device must be shown to clearly outweigh the risk for it to gain market approval. Article 2 (24) of the MDR defines the benefit-risk determination as “the analysis of all assessments of benefit and risk of possible relevance for the use of the device for the intended purpose when used in accordance with the intended purpose given by the manufacturer.”
A PMS system should clearly define benefit-risk calculations and the data used to support them. Post-market surveillance activities are critical in order to re-evaluate and maintain the benefit-risk calculations and determinations of a device throughout its life. Information that is gained through a PMS system can lead to:
• Identification of new risk factors.
• Adjustments to risk frequency and/or severity values based on actual use data.
• Adjustments to established risk calculations based on new “state of the art” technologies becoming available.
• Adjustments to established benefit calculations based on actual use data.
While complaint handling and other feedback tracking are more often described as part of post-market vigilance systems, they play a role in the more proactive post-market surveillance processes as well. A PMS system should define ...
To continue reading this ebook, download the full version.

An overview of 21 CFR Part 820 - quality systems for medical device manufacturers
What is 21 CFR Part 820?
21 CFR 820 is the FDA federal regulation that pertains to quality systems for medical device manufacturers, and it is part of the agency’s set of Current Good Manufacturing Practices (CGMP) for industry. Also referred to as the FDA’s quality system regulation (QSR), the regulation defines design controls and quality processes at all stages of device development in order to ensure that all medical devices marketed in the United States are safe and effective.
21 CFR 820 consists of 15 subparts, which define quality system requirements for each stage and function within the medical device manufacturing process. We define each subpart below.
Federal regulations are organized as Title → Chapter → Subchapter → Part, which means that 21 CFR 820 is short-hand for:

21 CFR 820 vs ISO 13485
ISO 13485 is the de facto international quality system standard for medical device manufacturers, but this is not currently the standard in the United States. While Part 820 and ISO 13485 are structured differently, they have no conflicting requirements. Therefore, companies that are marketing medical devices in the U.S. and in other markets will need to comply with both ISO 13485 and the FDA’s QSR, as defined in 21 CFR 820.
However, the FDA is moving towards harmonizing these standards, and on February 23, 2022 issued a proposed rule to amend the QSR to align more closely with the international consensus standard for Quality Management Systems, primarily by incorporating reference to the ISO 13485 standard. The FDA has published FAQ’s about the proposed rule.
21 CFR Part 820 Requirements
Part 820: General Controls (subpart A)
The General Controls subpart contains three sections providing general information about the regulation, including the scope and applicability along with key definitions.
Scope
The regulation defines current good manufacturing practice (CGMP) requirements governing the methods, facilities, and controls used for the “design, manufacture, packaging, labeling, storage, installation, and servicing of all finished devices intended for human use." Specifically, this subpart defines:
- Applicability:
The requirements of this regulation are intended to ensure the safety and efficacy of all finished medical devices intended for human use that are manufactured in or imported into the United States. Manufacturers that are involved in some, but not all, manufacturing operations should comply with those requirements that are applicable to the functions they are performing.
Exceptions:
- This regulation does not apply to manufacturers of medical device components, but such manufacturers are encouraged to use this regulation as guidance.
- Class I medical devices are exempt from the Design Controls defined in this regulation, except for those listed in § 820.30(a)(2).
- Manufacturers of blood and blood components are not subject to this regulation but are subject to Biologics good manufacturing practices as defined in Subchapter F, Part 606 of the regulation.
Definitions
This section of the regulation contains definitions for a number of terms used throughout the document. The following are the major definitions related to quality records:
- Design history file (DHF): A compilation of records that describes the design history of a finished device.
- Design input: The physical and performance requirements of a device that are used as a basis for device design.
- Design output: The results of a design effort at each design phase and at the end of the total design effort. The finished design output is the basis for the device master record. The total finished design output consists of the device, its packaging and labeling, and the device master record.
- Device history record (DHR): A compilation of records containing the production history of a finished device.
- Device master record (DMR): A compilation of records containing the procedures and specifications for a finished device.
Quality System
The section of the regulation sets the basic requirement for a quality system by stating that “Each manufacturer shall establish and maintain a quality system that is appropriate for the specific medical device(s) designed or manufactured, and that meets the requirements of this part.”
The term “appropriate” is used throughout this regulation and can be open to interpretation. A manufacturer, however, should assume that all requirements are appropriate and applicable except in cases where non-implementation of the requirement can be shown to have no effect on the product's specified requirements or ability to carry out necessary corrective actions.
Quality system requirements (subpart B)
This section of the regulation defines the overall responsibilities and the resources required for the management of the quality system.
Management responsibilities
Executive management is responsible for establishing a quality policy and ensuring adequate resources to effectively maintain and manage the quality system. In addition, management is responsible for establishing a specific quality plan, consisting of relevant practices, resources, activities, and procedures.
Quality audit
Periodic audits of the quality system are required to be conducted by personnel not directly responsible for the activities being audited. The dates and results of each audit need to be documented, along with the results of the audit. It is expected that corrective actions and, when necessary, reaudits, be performed for any identified noncompliances.
Personnel
Manufacturers are responsible for assigning sufficient personnel with appropriate experience and training to perform all tasks required by the quality system plan.
Design controls (subpart C)
Manufacturers of all class II and class III medical devices, along with the specific class I devices listed in paragraph (a)(2) of this regulation, are required to establish design control procedures that ensure design requirements are met as specified.
Design controls shall define:
- Design and development planning - Plans that describe the design and development activities, and responsibilities for these activities and their implementation.
- Design input - Procedures that ensure design requirements are appropriate and address the intended use of the device.
- Design output - Procedures that document design output, including acceptance criteria, so that conformance to design input requirements can be adequately evaluated.
- Design review - Formal and documented reviews of the ensign results that include participation from representatives of all.
- Design verification - Procedures for verifying the device design that confirm that the design output meets the design input requirements.
- Design validation - Procedures for validating the device design, ensuring that devices conform to defined user needs and intended uses, and including testing of production units under actual or simulated conditions.
- Design transfer - Procedures to ensure that the device design is correctly translated into production specification.
- Design changes - Procedures for identifying, documenting, validating, and managing the verification and approval process of all design changes before they are implemented.
- Design history file - A design history file (DHF) is required for each type of device and should include or reference the records necessary to demonstrate that the design was developed in accordance with the approved design plan and device requirements.
Document controls (subpart D)
Medical device manufacturers are required to put in place document controls for all documents required in this regulation.
Document approval and distribution
One or more people must be assigned to review and approve documents prior to issuance. The approval must be documented, include a date and the signature of the approver, and be made available at all locations where applicable. Procedures must also be in place to ensure that obsolete documents are removed and/or prevented from being used.
Document changes
Similar to document approval procedures, changes to documents must be approved, reviewed, and documented. Records of all changes must be maintained.
Purchasing controls (subpart E)
To continue reading this Regulatory Brief, including a definition of the remaining subparts and a comparison of 21 CFR 820 to ISO 13485, please download the full brief.

Ask Rimsys!
At Rimsys, we pride ourselves on delivering tools that empower medical device regulatory affairs professionals to simplify and automate daily workflows, react quickly to changing regulations, and track product registrations around the globe. In order to deliver software that empowers RA teams to exceed regulatory campaign expectations, we not only need to understand current and changing global regulations; but we also work hard to understand what the community is talking about, what issues they are trying to solve, and what they would like to explore in more depth.
So - we are asking for your input!
In order to address the topics that are of most interest to you, we’re launching our “Ask Rimsys” campaign. As part of this initiative, we invite you to submit your medical device regulatory questions to us - and once a week we’ll choose a question to answer either via direct social media response, a blog post, or even a short video response. Preference will be given to the following:
- Questions with interest to the broader regulatory community (we will not be able to provide individual or company-specific consultation).
- Questions which touch on regulatory workflows and best practices.
- Questions dealing with the use of technology in optimizing regulatory activities.
If your question is selected, we’ll send you some Rimsys gifts - typically one of our highly requested “Regulatory Ducks in a Row” t-shirts featuring our too-cool for regulatory problems mascot, Reggie the Regulatory Duck. Look for random giveaways during the week too!
We’ll also be holding periodic “As us Anything” webinars with our regulatory team and other experts. These webinars will most often cover a broad topic, such as UDI, and we will take questions ahead of, and during, the webinars. Check out our events page for a list of upcoming webinars and recordings of past webinars.
As a company built by regulatory professionals, we understand the constant pressure on regulatory affairs professionals to stay on top of the changing regulatory landscape and improve your game. That’s why we’re committed to being a reliable friend and a source of relevant information - as well as a provider of world-class RIM software and services. So, help us focus on the issues most important to you by submitting your regulatory questions on Twitter by using #AskRimsys (and be sure to follow us) or answering our short 3-question survey.
And feel free to respond to any of questions or our responses with any regulatory insights you have as well. We look forward to hearing from you, and remember, #AskRimsys!

Introduction to Notified Bodies
The medical device industry is vast, diverse, and heavily regulated, with different countries and regions having varying regulatory requirements and processes. The European Union (EU) is one of the largest markets in the world for medical devices, and if you intend to put a medical device on the market in the EU, there is a good chance you’ll have to work with a Notified Body.
So, what is a Notified Body? In this brief blog post, we’ll discuss what Notified Bodies are, what they do, and how to work with one.
What is a Notified Body?
Notified Bodies play the role of gatekeepers to the EU market for a majority of medical devices/in vitro diagnostics. Notified Bodies for medical devices are independent organizations appointed by EU member states to assess the conformity of certain products before they are placed on the market. Conformity assessments ensure that products are technically safe, compliant, correctly documented, and manufactured under the correct conditions as outlined by applicable legislation, primarily the EU MDR and IVDR in the case of medical devices.
It’s important to note that not all Notified Bodies have the capability to perform assessments for all types of products. For instance, some Notified Bodies have been designated for MDR, meaning they can perform conformity assessments according to EU MDR 2017/745, Article 35, and others can provide conformity assessments for in vitro diagnostics (IVDs) in accordance with EU IVDR 2017/746, Article 32. It’s essential to make sure you work with the right Notified Body to get your medical device to the EU marketplace.
What types of medical devices require a Notified Body?
Medical devices regulated under the MDR as Class IIa, Class IIb, or Class III devices require that a conformity assessment be performed by an accredited Notified Body before receiving a CE marking and being placed in the market. Some Class I devices also require an assessment by a Notified Body, including devices that are sterilized, have a measuring function, or are reusable surgical instruments. Other Class I devices can be self-assessed by the manufacturer.
In vitro diagnostic (IVD) devices designated as Class B, Class C, or Class D also require a conformity assessment by a Notified Body. Only Class A IVDs can be self-assessed by the manufacturer.
What is the role of Notified Bodies under the EU MDR/IVDR?
All medical devices must have CE marking before they can enter the European market. One of the main roles of Notified Bodies is to provide conformity assessment on medical devices before granting them a CE marking, which is also known as a “European Passport,” as it allows products to move relatively freely throughout the EU.
Notified Bodies provide conformity assessment during both the design and production phases of a product, ensuring that medical device manufacturers live up to the requirements of the MDR/IVDR. They audit manufacturing processes and conditions, quality management systems (QMS), and product specifications to make sure that they conform with EU regulations before being placed on the market. It’s also the role of the Notified Body to make sure the manufacturer has the infrastructure in place to market and provide post-market surveillance in the EU.
Notified Bodies provide conformity assessment in support of the EU MDR/IVDR General Safety and Performance Requirements (GSPR) but ultimately, it’s the manufacturer’s responsibility to ensure all requirements have been fulfilled. This includes choosing the right Notified Body to work with to get their medical device on the EU market.
How do you choose the right Notified Body to work with?
It might surprise you to know that not all Notified Bodies are designated for both MDR and IVDR, meaning that it is critical that you do your due diligence to connect with the right Notified Body for your product. Notified Bodies are designated by the competent authority of EU member states to perform certain types of conformity assessments for certain types of medical devices and manufacturing processes/conditions. This means that the Notified Body you work with is largely dependent on the type and classification of your medical device as well as the type of conformity assessment process you want the Notified Body to perform. A list of accredited Notified Bodies is available in the EU NANDO database, which can be searched and filtered by country, specific legislation, and other technical competencies.
A lot rides on selecting the right Notified Body, so it’s imperative that you find the organization that best fits your company’s needs. Learn what you can about their processes, how they communicate with their clients, and even their submission templates and processes. Choose an organization whose standards and processes align with your company’s goals.
According to Rimsys regulatory expert, Bruce McKean, when it comes to working with a Notified Body, “You can benefit from building a relationship with your Notified Body and collaborating with them on various topics. Although MDR and IVDR have forced more stringent requirements that may hinder a consulting relationship, they’re usually willing to offer a certain level of guidance to help you.”
Choosing and working with an Notified Body is a critical step towards getting your product on the EU market, especially for new medical devices. Be sure to do your research before selecting an Notified Body, and communicate with them throughout the product realization phases to ensure your medical device is in compliance with MDR/IVDR requirements. Working successfully with a Notified Body is the best way to fast-track your medical device into one of the largest markets in the world.
Read more about the general safety and performance requirements (GSPR) that Notified Bodies use as the basis for conformity assessments.

MedWatch: The FDA safety information and adverse event reporting program
What is MedWatch?
What should you do if you encounter a serious side effect from a prescription drug you've taken? Or what if you were using a medical device and it malfunctioned and caused you serious harm? Do you contact the manufacturer, your doctor, or your local government? The quick answer is to contact the FDA, and it’s not as complex as you may think.
In the early 1990s, the FDA knew it needed a reliable way to help physicians, health care workers, and all those who work in the medical field to report adverse events related to pharmaceuticals and medical devices. An adverse event is any undesirable experience associated with the use of a medical product or pharmaceutical drug. They wanted a system that could be used to track these events and give them the insight they needed to review and track the reported issues.
In 1993 the FDA introduced the MedWatch program designed to collect voluntary reports of adverse reactions and quality problems of drugs and medical devices, along with all other FDA-regulated products such as dietary supplements, cosmetics, pet food, toys, tobacco, infant formulas, etc. The Stakeholder Engagement Staff within the office of the FDA commissioner is ultimately responsible for this program. They work closely with all FDA centers and offices across the entire agency and serve as a liaison between stakeholder organizations and the general public. In addition, it is their responsibility to report on medical product safety alerts, recalls, statements, and periodic safety labeling change summaries.
MedWatch is not only for medical professionals to report adverse events, but the public is also encouraged to report adverse events as well. And reporting an event to the FDA may be easier than you may think.
MedWatch Reporting
When the FDA approves a medical drug or product, the agency has determined that the benefits of the product outweigh the risks. But, sometimes there are unforeseen risks that are discovered once a product or drug gets to market and people start using it. For example, a product may be used by a larger and more diverse group of people than was represented in the clinical testing. They may differ in the complexity of their health problems or other medications that they use. Because of these unanticipated events, the FDA encourages people to report problems they feel that they’ve experienced.
To report a voluntary event online, you can use the MedWatch Online Voluntary Reporting Form 3500B, which is located on the FDA’s website. Form 3500B is a consumer-friendly reporting form that contains less technical language than the online FDA Form 3500, which is to be used by healthcare professionals. The form will step you through the process and should take about 15-20 minutes to complete. You can submit your contact information (in case they have questions or need clarification) or you can request that your information be kept confidential.
What happens after a report is made to MedWatch?
- FDA staff enter the report into a database so that it is available for review and comparison to other reports.
- An FDA safety evaluator, often a pharmacist, doctor, or nurse, reviews the report and examines the database for similar reports.
- The FDA monitors the data for trends and investigates, if appropriate.
- The FDA takes necessary action to protect public health.
Examples of FDA actions that could result from a report include:
- issuing safety alerts with recommendations to monitor a product's use, adjust the way it is used, or stop using it
- updating the product labeling to reflect new warnings
- inspecting the manufacturer
- requesting a change in the product’s design, manufacturing process, packaging, or distribution
- requesting that a company recall a product
MedWatch is a safety tool for everyone to use. Every MedWatch report is important and is recorded in an FDA database for review and comparison to similar previous reports. Taken together, these reports can identify trends and signal safety problems, leading to an FDA action to protect the public. Remember, reporting can help you, a family member, or someone else avoid harm, serious illness, or even death.
For more information, refer to the FAQ’s on the FDA website.
Join the MedWatch e-mail list here

An overview of 21 CFR Part 820
What is 21 CFR Part 820?
21 CFR 820 is the FDA federal regulation that pertains to quality systems for medical device manufacturers, and it is part of the agency’s set of Current Good Manufacturing Practices (CGMP) for industry. Also referred to as the FDA’s quality system regulation (QSR), the regulation defines design controls and quality processes at all stages of device development in order to ensure that all medical devices marketed in the United States are safe and effective.
21 CFR 820 consists of 15 subparts, which define quality system requirements for each stage and function within the medical device manufacturing process. We define each subpart below.
Federal regulations are organized as Title → Chapter → Subchapter → Part, which means that 21 CFR 820 is short-hand for:

21 CFR 820 vs ISO 13485
ISO 13485 is the de facto international quality system standard for medical device manufacturers, but this is not currently the standard in the United States. While Part 820 and ISO 13485 are structured differently, they have no conflicting requirements. Therefore, companies that are marketing medical devices in the U.S. and in other markets will need to comply with both ISO 13485 and the FDA’s QSR, as defined in 21 CFR 820.
However, the FDA is moving towards harmonizing these standards, and on February 23, 2022 issued a proposed rule to amend the QSR to align more closely with the international consensus standard for Quality Management Systems, primarily by incorporating reference to the ISO 13485 standard. The FDA has published FAQ’s about the proposed rule.
21 CFR Part 820 Requirements
Part 820: General Controls (subpart A)
The General Controls subpart contains three sections providing general information about the regulation, including the scope and applicability along with key definitions.
Scope
The regulation defines current good manufacturing practice (CGMP) requirements governing the methods, facilities, and controls used for the “design, manufacture, packaging, labeling, storage, installation, and servicing of all finished devices intended for human use." Specifically, this subpart defines:
- Applicability:
The requirements of this regulation are intended to ensure the safety and efficacy of all finished medical devices intended for human use that are manufactured in or imported into the United States. Manufacturers that are involved in some, but not all, manufacturing operations should comply with those requirements that are applicable to the functions they are performing.
Exceptions:
- This regulation does not apply to manufacturers of medical device components, but such manufacturers are encouraged to use this regulation as guidance.
- Class I medical devices are exempt from the Design Controls defined in this regulation, except for those listed in § 820.30(a)(2).
- Manufacturers of blood and blood components are not subject to this regulation but are subject to Biologics good manufacturing practices as defined in Subchapter F, Part 606 of the regulation.
Definitions
This section of the regulation contains definitions for a number of terms used throughout the document. The following are the major definitions related to quality records:
- Design history file (DHF): A compilation of records that describes the design history of a finished device.
- Design input: The physical and performance requirements of a device that are used as a basis for device design.
- Design output: The results of a design effort at each design phase and at the end of the total design effort. The finished design output is the basis for the device master record. The total finished design output consists of the device, its packaging and labeling, and the device master record.
- Device history record (DHR): A compilation of records containing the production history of a finished device.
- Device master record (DMR): A compilation of records containing the procedures and specifications for a finished device.
Quality System
The section of the regulation sets the basic requirement for a quality system by stating that “Each manufacturer shall establish and maintain a quality system that is appropriate for the specific medical device(s) designed or manufactured, and that meets the requirements of this part.”
The term “appropriate” is used throughout this regulation and can be open to interpretation. A manufacturer, however, should assume that all requirements are appropriate and applicable except in cases where non-implementation of the requirement can be shown to have no effect on the product's specified requirements or ability to carry out necessary corrective actions.
Quality system requirements (subpart B)
This section of the regulation defines the overall responsibilities and the resources required for the management of the quality system.
Management responsibilities
Executive management is responsible for establishing a quality policy and ensuring adequate resources to effectively maintain and manage the quality system. In addition, management is responsible for establishing a specific quality plan, consisting of relevant practices, resources, activities, and procedures.
Quality audit
Periodic audits of the quality system are required to be conducted by personnel not directly responsible for the activities being audited. The dates and results of each audit need to be documented, along with the results of the audit. It is expected that corrective actions and, when necessary, reaudits, be performed for any identified noncompliances.
Personnel
Manufacturers are responsible for assigning sufficient personnel with appropriate experience and training to perform all tasks required by the quality system plan.
Design controls (subpart C)
Manufacturers of all class II and class III medical devices, along with the specific class I devices listed in paragraph (a)(2) of this regulation, are required to establish design control procedures that ensure design requirements are met as specified.
Design controls shall define:
- Design and development planning - Plans that describe the design and development activities, and responsibilities for these activities and their implementation.
- Design input - Procedures that ensure design requirements are appropriate and address the intended use of the device.
- Design output - Procedures that document design output, including acceptance criteria, so that conformance to design input requirements can be adequately evaluated.
- Design review - Formal and documented reviews of the ensign results that include participation from representatives of all.
- Design verification - Procedures for verifying the device design that confirm that the design output meets the design input requirements.
- Design validation - Procedures for validating the device design, ensuring that devices conform to defined user needs and intended uses, and including testing of production units under actual or simulated conditions.
- Design transfer - Procedures to ensure that the device design is correctly translated into production specification.
- Design changes - Procedures for identifying, documenting, validating, and managing the verification and approval process of all design changes before they are implemented.
- Design history file - A design history file (DHF) is required for each type of device and should include or reference the records necessary to demonstrate that the design was developed in accordance with the approved design plan and device requirements.
Document controls (subpart D)
Medical device manufacturers are required to put in place document controls for all documents required in this regulation.
Document approval and distribution
One or more people must be assigned to review and approve documents prior to issuance. The approval must be documented, include a date and the signature of the approver, and be made available at all locations where applicable. Procedures must also be in place to ensure that obsolete documents are removed and/or prevented from being used.
Document changes
Similar to document approval procedures, changes to documents must be approved, reviewed, and documented. Records of all changes must be maintained.
Purchasing controls (subpart E)
To continue reading this Regulatory Brief, including a definition of the remaining subparts and a comparison of 21 CFR 820 to ISO 13485, please download the full brief.

What is the FDA eSTAR program?
History
In September of 2018, the FDA took their first step in their commitment to improve the process of a 510(k) electronic submission. This commitment was established as part of the Food and Drug Administration Safety and Innovation Act (FDASIA) that was enacted on July 9, 2012, amending the FD&C Act by adding section 745A.
Based on the FDA’s experience with the original eSubmitter process, the Center for Biologics Evaluation and Research (CBER) group piloted a slightly different program and called it the Electronic Submission Template and Resource Pilot Program, thus creating eSTAR. This second attempt at electronic submissions consists of a collection of questions, text, and prompts within a PDF template that guides a user to a ‘complete’ 510(k) electronic submission.
The FDA’s goal of this process is to enhance the quality of submissions by helping to ensure consistent, quality, comprehensive data for the Center for Disease and Radiological Health (CDRH) premarket review. With a standardized format in place, submitters of a 510(k) or De Novo can ensure that their submissions are complete and premarket reviews will be more efficient.
eSTAR timeline
.jpeg)
eSTAR Requirements
Currently, eSTAR is a voluntary process that will become mandatory starting October 1, 2023. It is free to use by medical device applicants wishing to submit a 510(k) or a De Novo to the CDRH, but is not to be used for combination products. Although it’s currently free to use, the standard submission fees for 510(k) and De Novo still apply.
A Refuse to Accept (RTA) review (a preliminary review used to ensure the submission is complete) will not be conducted on submitted eSTAR templates as the eSTAR template replaces this checklist and will state if the template is complete or incomplete on the first page. If the eSTAR does not have a completed status, it will not be reviewed by the FDA.
Guidance Information
The FDA has provided final guidance documentation in “Providing Regulatory Submissions for Medical Devices in Electronic Format — Submissions Under Section 745A(b) of the Federal Food, Drug, and Cosmetic Act” (referred to as the “745A(b) device parent guidance”). This guidance document provides a process for the development of templates to facilitate the preparation, submission, and review of regulatory submissions for medical devices solely in electronic format.
The FDA then provided final guidance in “Electronic Submission Template for Medical Device 510(k) Submissions.” There are exemptions listed in the guidance document that may allow users in certain situations to be exempted from eSTAR.
eSTAR Templates
The two templates provided by the FDA are PDF forms that can be filled out and saved, with attachments. The image below shows the device description page of the non-IVDR form. When using the form, red bars will appear in front of any required questions that have not yet been answered. Bars will turn green when all associated questions in a section have been completed. Gray bars indicate an optional question.

As questions are answered, additional dropdowns may appear that can require users to add additional information. For example, in the Pre-Submission Correspondence and Previous Regulatory Interaction Section within the non-IVD template, if you Select “Yes” the PDF will then add a section where you will need to add the Submission, Submission Number, and copies of the correspondence.

Pop-ups with useful information may also appear. For example, within the non-IVD template, in the General Device Characteristics Section, if a user selects that a device is a single use device, non-sterile or packaged as sterile, a Java Script window will appear with FDA guidance.

A note on embedded attachments: Any embedded attachments may only be used once per eSTAR PDF. You may need to “break up” documentation to fit the eSTAR template requirements if your testing reports cover multiple tests in one document.
What’s involved in a submission?
There are currently two processes for submission:
- The Customer Collaboration Portal
- Mailing the eCopy (CD, DVD or USB drive) containing the eSTAR PDF
First, effective July, 2022, The Customer Collaboration Portal was made available to existing users of the portal if they are the official correspondent of an organization and have also received the FDA email letting them know that they’re eligible.
The second method consists of a printed cover letter with an accompanying eCopy (CD, DVD or USB drive) containing the eSTAR PDF. Note that if the PDF submission exceeds 1 GB in size, it could delay the process, so high-resolution videos and images should only be included when necessary.
- Download the specific eSTAR PDF template for an In Vitro or Non-In Vitro device (see "Current eSTAR versions" section on this linked page).
- Fill out the template accordingly but note that it is only used for constructing - not submitting - your submission.
- The eSTAR does not need to comply with the FDA’s eCopy Guidance document, however, any additional files that are provided with the eSTAR PDF submission will need to comply with it. The FDA strongly recommends not providing additional files with the eSTAR PDF, though, as you already will be embedding your documents within the eSTAR template itself.
- You do not need to provide an Indications for Use page, the Premarket Review Submission Cover Sheet, or a Declaration of Conformity (if applicable) with your submission since they are already built into the eSTAR PDF. An additional option is to also build the 510(k) summary within the template itself instead of writing one.
An eSTAR submission will contain a larger number of files (as they will be embedded in the PDF eSTAR template) than traditional submissions, which are usually combined into one PDF file. We recommend preparing your entire submission before filling out the eSTAR template to minimize the need for reworking the submission.
A note on 510(k) product change submissions: The eSTAR template requires that all subsections be completed, so if you are submitting a 510(k) to address a product change, you will want to include a justification for not including original information in required sections that did not change. Otherwise, all submitted information will be reviewed again, even if it does not differ from the original submission.
Review Timeline
The review timeline will be similar to the review timeline for a non-electronic submission for a 510(k). The guidance document “The 501(k) Program: Evaluating Substantial Equivalence in Premarket Notification [510(k)]” can offer more details. The same would apply for the De Novo in that you would need to reference the guidance document “De Novo Classification Process (Evaluation of Automatic Class III Designation)” for review timelines.
Tracking the Submission
The FDA secures the information about each submission’s progress so only its Official Correspondent (your company’s submission representative) can view it. You will be able to track the progress of your submission by using the Customer Collaboration Portal (CCP).
- If this is your Official Correspondent’s first time tracking a submission online, the FDA will automatically email a link to create a login password soon after the FDA starts its review.
- The FDA currently displays progress online for Traditional, Abbreviated, and Special 510(k) submissions.
- The FDA will formally notify you of your submission’s status by emailing your Official Correspondent with official actions and requests.
The future of eSTAR
These efforts are part of the FDA’s ongoing commitment to work with the industry and to improve the efficiency of the medical device review process. The final guidance for the 510(k) eSTAR was published this month and will become effective October 1, 2023, for 510(k) and De Novo submissions. In the long term if MUDUFA V is passed with the current requests from the FDA, the process will become a required standard for additional submission types such a PMA and will replace the need to mail eCopies containing eSTAR files through the FDA Collaboration Portal.

RAPS wrap-up
What a great conference! This year’s RAPS Convergence conference in Phoenix was outstanding by many measures. Not only was this the first time in quite a while that our community has had a chance to network in person, but our team also reported that the quality of conversations and presentations far surpassed their expectations. Kudos to the RAPS conference team for putting together a valuable and memorable experience.
We asked our team to share their thoughts about the conference, along with information they learned while attending sessions or in conversation with other RA professionals. We’ve compiled their thoughts below.
General thoughts on the conference
The consensus from our team was that this conference attracted a really good group of knowledgeable regulatory professionals and that the majority of sessions were incredibly informative. Everyone referenced great conversations they had throughout the conference.
.jpeg)
There were a lot of discussions around the challenges that RA teams face today, and what can be done to ease the burden of regulatory professionals in the face of ever-changing requirements across the globe.
I was impressed by the consistently high level of quality in the conversations I had. Every person I spoke with had an intimate understanding of the challenges currently faced by industry and expressed a willingness to engage in conversation about how to improve things. While there are clearly significant challenges to overcome, I am bullish about the future.
James Trotter
And regulatory teams are looking for ways to be less reactive and find time for strategic planning to prepare for future challenges.
One of my biggest takeaways is the true shift in thinking that is happening within the industry in regard to the need to think strategically about supporting regulatory teams. In a number of conversations, it was mentioned that RA folks wished that they had more time to take a step back and have future-facing conversations, rather than just “keeping up” with their day-to-day activities.
Alex Tallentire
We noticed how well-attended the sessions were and heard comments from a number of people that they were having trouble deciding which sessions to attend. RAPS has made presentation slide decks available to attendees through the conference website.
.jpeg)
Reggie, our regulatory duck, was also a big hit! Did you get one of our duck t-shirts?
Key session takeaways
CDRH Forum
The conversation was animated during the CDRH Forum, which ended with FDA stating “Communication is key. We take all feedback seriously and we are consistently making improvements.” Discussions centered largely around MDUFA V and the use of eSTAR templates. FDA is focusing on addressing the rising volumes of premarket submissions. This includes hiring additional reviewers, better support of 3rd party reviews to eliminate the need for FDA re-review, improved deficiency letter communication, and the expansion of electronic templates for submissions (including use with PMAs). They see these methods plus others as helping the agency meet the performance requirements on MDUFA V.
EU Regulations: PMCF and SSCP
Industry frustration - both by Notified Bodies and manufacturers - clearly continues over the burdensome requirements around PMCF and SSCP. As a subset of PMS, a PMCF plan is critical to MDR compliance and mandatory for Class III and implantable devices. Having a good plan is key, and industry is struggling to figure out what to do with the data once it is collected.
Because the audience for SSCP documents is healthcare professionals and patients, manufacturers will have to conduct readability assessments for each Summary of Safety and Clinical Performance (SSCP) through computer-based software programs or layperson assessments in all languages used in the EU market.
EUDAMED UDI
While the EUDAMED UDI module isn’t expected to be mandatory until the second quarter of 2026, there are triggering events that will require a manufacturer to enter data into the UDI module earlier. For example, mandatory use of the EUDAMED Vigilance module is Q4, 2024. If you have to report a product in the Vigilance module, that product must also be entered into the UDI module at that time. Bruce McKean, Rimsys Director of Regulatory, says that “Manufacturers cannot wait, they must be proactive rather than reactive when it comes to UDI.”
eSTAR
During the Solution Circle on “How to efficiently prepare your eSTAR submission,” attendees learned that FDA plans to expand the program to include more submission types. eSTAR submissions require a larger number of files attached to the PDF checklist, compared to the number of files submitted under traditional submission programs. One recommendation was to prepare your submission completely before filling out the eSTAR template. It seems apparent that the eSTAR program is designed to make the review process simpler for agency reviewers, but does not simplify the creation of the submission by the manufacturers.
If you are submitting a 510(k) to address a product change, the eSTAR template requires that all subsections be completed, even if they are not applicable to the change. This means that previously submitted and cleared information goes under review again. To avoid this, the recommendation was made that submitters include justification for not resubmitting data in these sections, rather than including the original data.
It is believed that the current guidance will be made final by the end of September 2022 with a one-year transition period for 510(k) and De Novo submissions.
What’s next?

RAPS has announced that next year’s conference will be in Montreal, October 3-5. We look forward to seeing everyone there (and we are working on next year's t-shirt design already). After this year’s great conference, we can’t wait to see next year’s!





