

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

The ultimate guide to the EU MDR and IVDR general safety and performance requirements (GSPR)
This article is an excerpt from The ultimate guide to the EU MDR and IVDR general safety and performance requirements (GSPR) ebook.
Table of contents
- Overview
- Terminology
- EU MDR/IVDR Annex I
- EU MDR/IVDR Annex II
- Proactive Monitoring & Maintenance
- Comparison Table: EU MDR/IVDR Annex I GSPRs vs EU MDD/IVDD Annex I Essential Principles
With the initial rollout of the European Medical Device Regulation (MDR) complete, medical device companies are shifting focus to the sister In Vitro Diagnostic Regulation (IVDR) which has rolling effective dates starting in May 2022. Like the MDR, the IVDR also includes new General Safety and Performance Requirements (GSPR). The expanded 2nd edition of this ebook includes a detailed summary of the IVDR GSPR regulations in addition to those of the MDR. It provides you with practical guidance on how to meet the GSPR requirements for all types of medical technology products. This ebook, however, should not take the place of reviewing the actual regulations and consulting regulatory experts when needed
Timeline
The EU MDR submission became mandatory from the previous MDD directive on May 26, 2021, and the EU IVDR effective date is quickly approaching. In fact, all submissions for new devices under the new EU IVDR must be implemented no later than May 25, 2022. Below is a high-level overview of key dates for both regulations.
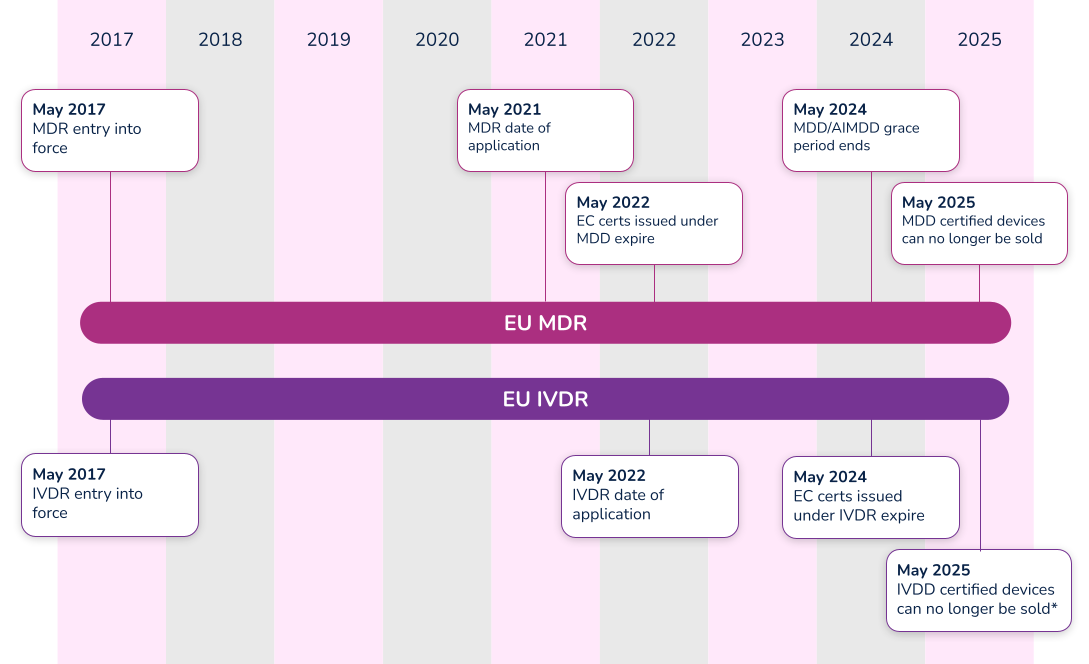
*Note that the timeline for compliance was extended in 2021. Class D (high-risk) devices have until 2025 to comply with IVDR, while Class C devices have until 2026. Class B and Class A sterile devices have until 2027 to comply with IVDR.
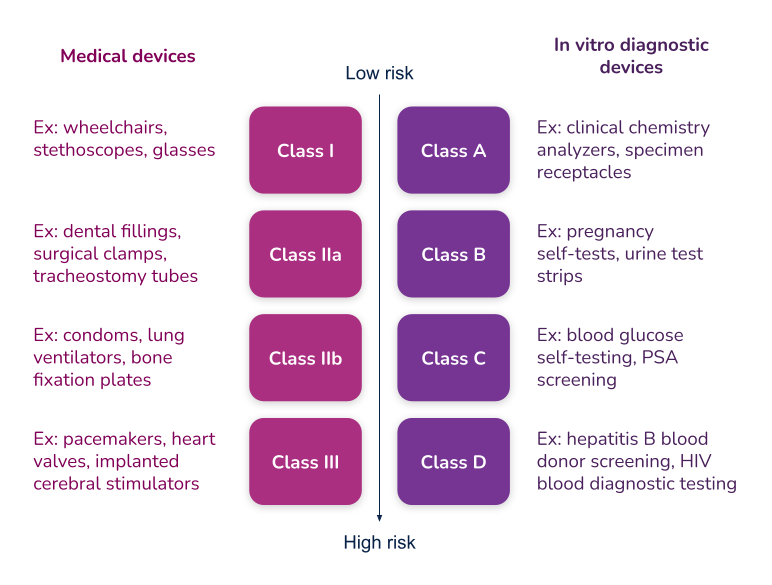
What’s the difference between Essential Requirements, General Safety and Performance Requirements (GSPR), and Essential Principles. In order to have a meaningful dialogue, let’s first discuss the three (3) main terms used in the industry.
#1 Essential requirements
The ‘Essential Requirements’ is the backbone for establishing conformity with the Medical Device Directive (MDD 93/42/EEC) and the Active Implantable Medical Device Directive (AIMDD 90/385/EEC). Detailed within Annex I of the MDD and AIMDD, the ‘Essential Requirements’ laid out the requirements that devices must meet in order to state compliance to the directives. With the implementation of the new EU Medical Device Regulation (MDR 2017/745), the ‘Essential Requirements’ will become superseded by the new EU MDR General Safety and Performance Requirements (GSPRs).
#2 Essential principles
The IMDRF laid out Essential Principles requirements in a document entitled Essential Principles of Safety and Performance of Medical Devices and IVD Medical Devices. From a high-level perspective, three basic tenets make up these ‘Essential Principles’:
- A device must be designed to be safe and perform effectively throughout its lifecycle.
- Device manufacturers must maintain all design characteristics.
- Devices must be used in a way that is consistent with how it was designed.
Many countries use the term ‘Essential Principles’ when compiling the documentation required to determine compliance to the law. For instance, the Australian Therapeutic Goods Administration (TGA) uses the term ‘Essential Principles Checklist’. Regardless of the term used, Essential Principles are of similar nature and overlap many of the Essential Requirements and new GSPRs.
#3 General safety and performance requirements (GSPR)
As of May 26, 2021, medical device manufacturers must start to comply with Annex I – General Safety and Performance Requirements (GSPRs) of the new EU Medical Device Regulation (MDR 2017/745). GSPRs are specific to the European MDR and IVDR. If you hear any other term (i.e. Essential Principles), it most likely means it is not referencing the European market.
Annex I of the EU MDR and IVDR details the specific requirements of the General Safety and Performance Requirements (GSPRs). The GSPRs are broken down into three (3) chapters in Annex I, MDR 2017/745 and IVDR 2017/746:
- Chapter 1 - General requirements
- Chapter 2 - Requirements regarding design and manufacture
- Chapter 3 - Requirements regarding the information supplied with the device
Chapter 1 - General requirements
Both the EU MDR and the EU IVDR outline General Safety and Performance Requirements (GSPRs) in great detail for medical device designers and manufacturers. The general requirements for each are almost identical and consist of the following:
- Devices must perform in a way that aligns with the intended design.
- They must not compromise the health or safety of a patient, user, or any other person associated with the device.
- Risks must be reduced as much as possible, but not so much that they negatively affect the risk-benefit ratio.
- Device manufacturers must implement and maintain a thorough, well-documented, and evaluative risk management system that continues to be updated throughout the life cycle of a device.
- Manufacturers and designers must include any necessary measures for protecting users in cases where risks cannot be completely eliminated.
- Manufacturers must provide users with information about any potential risks that remain. This information must be clear, easy to understand, and considerate of the users’ technical knowledge level, use environment, and any applicable medical conditions.
- Devices must withstand the stresses of normal use for the duration of their lifecycle. Devices must be designed, manufactured, and packaged in a way that protects them from damage during transport and storage.
- When it comes to risks and negative side effects that are known and foreseeable, designers and manufacturers must make every effort to minimize negative outcomes. They must also ensure that potential risks are acceptable when compared to the potential benefits of a device to its users.
Chapter 2 - Requirements regarding design and manufacture
The GSPRs also provide key details regarding specific information about the performance, design and manufacture of medical devices. As it relates to design inputs, the MDR and IVDR GSPRs provide highly detailed requirements relating to a device’s technical information. Further detail can be found in the comparison tables in Appendix A and Appendix B, where we have compared MDR to MDD and IVDR to IVDD.
Chapter 3 - Requirements regarding the information supplied with the device
The final key area of governance within the GSPRs relates to specific information a manufacturer must supply with a device. The general requirements for this information states that, “Each device shall be accompanied by the information needed to identify the device and its manufacturer, and by any safety and performance information relevant to the user, or any other person, as appropriate.” The requirements provide further detail as far as location - specific information that must be provided on the following:
- The device label includes its UDI.
- The user instructions.
- The packaging of a device that is intended to maintain its sterile condition.
Medical devices are subject to significant regulations and a full understanding of EU MDR and/or IVDR labeling as defined in Annex 1 Chapter 3.
In addition to the specific requirements identified within Annex I of the EU MDR and IVDR, Annex II, Technical Documentation, identifies additional requirements. Specifically, in both EU MDR and IVDR’s Section 4 – General Safety and Performance Requirements it states:
“the documentation shall contain information for the demonstration of conformity with the general safety and performance requirements set out in Annex I that are applicable to the device taking into account its intended purpose, and shall include a justification, validation and verification of the solutions adopted to meet those requirements. The demonstration of conformity shall include:
(a) the general safety and performance requirements that apply to the device and an explanation as to why others do not apply;
(b) the method or methods used to demonstrate conformity with each applicable general safety and performance requirement;
(c) the harmonised standards, CS or other solutions applied; and
(d) the precise identity of the controlled documents offering evidence of conformity with each harmonised standard, CS or other method applied to demonstrate conformity with the general safety and performance requirements. The information referred to under this point shall incorporate a cross reference to the location of such evidence within the full technical documentation and, if applicable, the summary technical documentation.”
Let’s break this down into each part.
Requirement
(a) the general safety and performance requirements that apply to the device and an explanation as to why others do not apply;
What needs to be documented for the requirements that apply or the requirements that do not apply?
Each and every section of the EU MDR GSPR or EU IVDR should be assessed in its own right as it pertains to your medical device. When a requirement applies, a simple statement may be made that this requirement applies to the device. In practice this is often achieved using a checklist or table, with a column for applicability and a Yes/No answer against each requirement. When a requirement applies, you can move on to the other parts of demonstrating conformity regarding methods used and standards applied.
When a requirement is not applicable, a statement must be made to that effect, i.e. a ‘No’ in the applicability column. Additionally, it must be fully and properly justified. Such a justification may be something like ‘The device is not powered and is therefore not an active device. This requirement does not apply.' The justification should clearly state why the requirement has been deemed not to apply so that your notified body can understand your reasoning
Requirement
(b) the method or methods used to demonstrate conformity with each applicable general safety and performance requirement;
What is meant by “method or methods used”?
This relates to the way you complied with that GSPR requirement, historically it would be listed as a standard or other documentation reference that you have applied to demonstrate compliance, however, the question of ‘method or methods used’ is new to the MDR and it is expected that a verbal description be provided such as:
i. Risk analysis weighed against clinical evaluation benefit
ii. Performance intended demonstrated by design requirements, verification and validation
Requirement
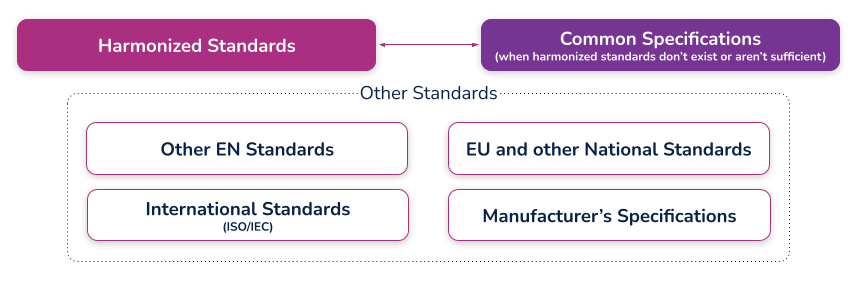
(c) the harmonized standards, common standards (CS) or other solutions applied;
What are harmonized standards, common specifications (CS), and “other solutions”?
Harmonized standards
These are standards that have been specifically developed and assessed for compliance to a regulation or directive. They are published in the Official Journal of the European Union (sometimes just referred to as ‘the OJ’) and if you comply with these standards then there is a ‘presumption of conformity’ with that directive or regulation to which they have been harmonized. These harmonized standards can only be created by a recognized European Standard Organization (such as CEN or CENELEC). When a standard is harmonized, an annex is added that describes how the standard conforms to the directive or regulation. When using harmonized standards, you should make sure that you understand how the standard conforms so that you do not claim compliance when the standard either does not meet that requirement or only partially meets that requirement.
If a standard does not meet a certain requirement of the directive or regulation, or indeed only partially meets it, then you must employ additional mechanisms for compliance. If a harmonized standard meets part of a directive or regulation, then by complying with that standard you also fully meet the corresponding requirement(s) The list of harmonized standards continues to grow - refer to the “Healthcare Engineering” section of the European Commission’s Harmonized Standards page for current information. In this case, using an MDD harmonized standard and documenting a justification for doing so (i.e. how you believe the standard demonstrates compliance with the GSPRs), should provide sufficient evidence
Common specifications
Common Specifications (CS) are a new concept in the MDR. They allow the European Union to add additional requirements that must be met in order to claim compliance where harmonized standards do not exist or where relevant standards are considered insufficient. The definition of a Common Specification is:
‘A set of technical and/or clinical requirements, other than a standard, that provides a means of complying with the legal obligations applicable to a device, process or system.’
Requirement
(d) the precise identity of the controlled documents offering evidence of conformity with each harmonized standard, CS or other method applied to demonstrate conformity with the general safety and performance requirements. The information referred to under this point shall incorporate a cross- reference to the location of such evidence within the full technical documentation and, if applicable, the summary technical documentation;
What is the expectation for incorporating a "cross-reference to the location of such evidence within the full technical documentation"?
This means that someone looking at the document should be able to identify exactly where in the technical documentation that the compliance evidence can be found. For example, this may refer to test reports and their exact location, or it could even reference locations within a large document, depending on the GSPR and your particular documentation. (i.e. if you have included usability risks as part of a larger risk assessment, you may need to say ‘See Technical File XXX, Section XX, Doc RMF001 rev 3 lines 65-78’). In other cases it could just mean the whole document reference, i.e. Have you done risk management? – then yes, it is RMF001 rev 3. What the specific reference actually is depends on how you have managed your technical documentation and how defined it is (i.e. separate reports or one big one). There should be no ambiguity as to where the document is located
An example of a completed GSPR checklist could look something like this (applicable and nonapplicable examples are shown):
Specification developers and manufacturers must continually maintain their technical documentation to stay compliant. Part of this process is to ensure that they take into account the "generally acknowledged state of the art".
Proactive monitoring
'State of the art'
There is no formal definition of ‘state of the art’ within the EU MDR or IVDR, although it is mentioned many times. ‘State of the art’ is an ongoing debate; however, it generally means that it embodies what is currently and generally accepted as good practice in the medtech industry. The ‘state of the art’ does not necessarily imply the most technologically advanced solution.
One consensus on state of the art is being up to date and compliant with the current and in effect standards that are applicable to your device. This means that if a standard is updated that your medical device is compliant with, you must evaluate that update to ensure that it would meet the EU MDR or EU IVDR ‘state of the art’ requirement. This is not a new requirement from the EU MDD but it is spelled out more clearly in the EU MDR.
The specification developer or manufacturer is ultimately responsible for determining if the updated standard applies or does not apply to their device(s). Either way, the justification should be documented within a gap analysis.
Monitoring for changes
Of course, 'state of the art' only applies if you actually know if something changed. This is why you need to develop a process for monitoring the standards that compliance is claimed. Every single standard that is associated with your technical documentation must be actively monitored, reviewed, and reported on.
If you have a product on the market and need a better way to monitor and maintain your General Safety and Performance Requirements (GSPR) or Essential Principles, Rimsys can help. Rimsys digitizes and automates GSPR and Essential Requirements so you can dynamically update and proactively monitor changing standards and evidence files.
When a standard or evidence file changes, you will automatically be notified and can update one GSPR or all of your GSPRs as applicable with a single click of a button. If additional information is needed, such as testing, it’s also invaluable to ensure that all devices are identified. What used to take weeks of manual, error-prone administrative tasks is now done in seconds within a fully validated, secure, maintenance-free, cloud-based solution
Maintenance
Maintaining and updating your technical documentation is generally the hardest part of staying compliant. Robust processes must be established to ensure nothing slips through the cracks and show up as nonconformances during regulatory audits.
Gap analysis
In addition to meeting the ‘state of the art’ requirements and the continuous proactive monitoring of standards, once a change has been detected that affects the technical documentation, a proper and thorough gap analysis must be completed.
The gap analysis between the old versions and the new versions, or an evaluation of a brand new standard, must occur and be properly documented. The gap analysis should detail what is applicable and what is not applicable, with your supporting justification.
If something within the new or revised standard was applicable to your device, additional engineering testing, documentation, justification, and, in some instances design changes, may be needed to ensure compliance
GSPR updates
Once the gap analysis has been properly documented, specification developers and manufacturers must update their GSPRs.
These updates include finding the withdrawn or superseded standard or evidence file throughout each row within your GSPR table, for every single device on the market on which this change is applicable. This could be one table or dozens of tables depending on the complexity of the products and your product mix.
Without a holistic RIM system to help you, this is an error-prone process as is it tedious, administrative, and extremely easy to miss an inappropriate referenced standard or evidence file.
Extreme diligence on the regulatory or engineering team must occur to ensure these critical updates to the GSPRs are not missed and a gap analysis must be properly referenced throughout. Any justification for including or excluding a new standard or evidence file will be scrutinized by regulatory auditors, and without proper maintenance, may lead to additional review time.
To continue reading this eBook including Comparison Table of the EU MDR Annex I GSPR vs. the EU MDD Annex I Essential Requirements, please register to download the full version.

The beginner's guide to the FDA PMA submission process
This article is an excerpt from The beginner's guide to the FDA PMA submission process ebook.
Table of Contents
- Introduction
- PMA basics
- FDA interactions
- Contents of a traditional PMA submission
- PMA supplements and amendments
- PMA Quality Management System (QMS)
- Review process and timeline
If your organization is planning to market a new medical device in the United States, you first need to determine which regulatory class the device falls under. The vast majority of medical devices regulated by the FDA are either Class I or Class II medical devices, requiring a 510(k) premarket notification or a simple registration if exempt from 510(k) requirements. However, if your device sustains or supports life, is implanted, or presents a “potential unreasonable risk of illness or injury,” your device is likely a Class III device which will require Premarket Approval (PMA) from the FDA before it can be marketed in the United States. Novel devices, for which there are no existing substantially equivalent devices, are automatically classified as Class III as well. Novel devices with a lower risk profile, however, may qualify for the De Novo process instead of the PMA. Just 10% of devices regulated by the FDA are Class III devices.
This ebook provides an overview of the PMA process and its requirements, but it is not designed to be the only resource used in compiling a PMA submission. The FDA provides significant documentation on this process, starting with the regulation governing premarket approval that is located in Title 21 Code of Federal Regulations (CFR) Part 814.
FDA: Background and device oversight
Before we explain what a PMA is, let’s first talk generally about the Food and Drug Administration (FDA) and device oversight. The FDA is the U.S. governmental agency responsible for overseeing medical devices, drugs, food, and tobacco products. When it comes to medical devices, the FDA’s mission is to “protect the public health by ensuring the safety, efficacy, and security of...medical devices.” At the same time, the FDA also has an interest in “advancing public health by helping to speed innovations.” In other words, the FDA’s goal is to make sure devices are safe and effective for public use, while also ensuring that devices have a quick and efficient path to market.
In order to achieve this balance of safety and efficiency, the FDA has three different levels of oversight depending on the risk level of the device: (1) exempt from premarket notification, (2) Premarket Notification, also known as 510(k), and (3) Premarket Approval (PMA).
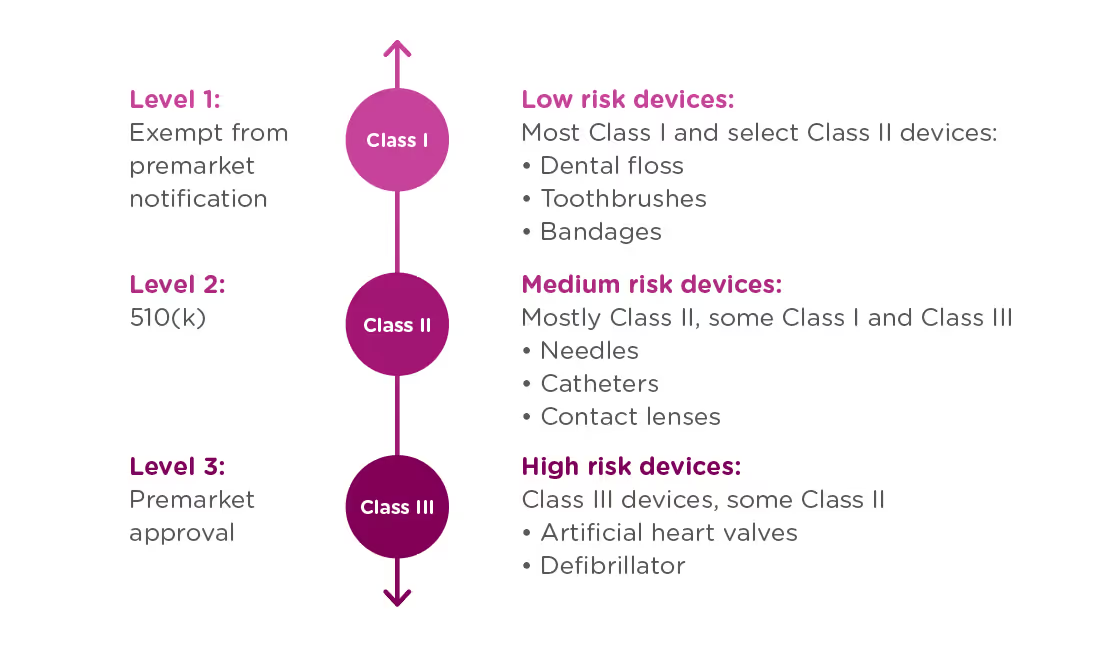
When is a PMA required?
The PMA process is the most stringent regulatory process for medical device approval under the FDA and applies to almost all Class III devices. To determine whether your device requires a PMA, you must first Classify your device by searching the Product Classification Database. The database will provide you with similar devices; their name, classification, and link to the Code of Federal Regulations (CFR) if applicable.
- If a substantial equivalent is found in the Product Classification Database with a submission type of 510(k), you should submit a 510(k), not a PMA.
- If the product classification database identifies your device as Class III and/or requiring a PMA - you should submit a PMA.
- If your device involves a new concept and does not have a classification regulation in the CFR, the database will list only the device type name and product code. In this case, the three-letter product code can be used to search the PMA database and the 510(k).
- If your device cannot be found in the product classification database because it is a new type of device and should be classified as a Class III device because of the level of risk it presents*.
Class III devices support or sustain human life, are of substantial importance in preventing impairment of human health, or present a potential and unreasonable risk of illness or injury.
Note that if your device is a new concept without a substantial equivalent, but does not present the level of risk of a class III device, it may be eligible for the De Novo process as a class I or class II device.
PMA vs 510(k)
Not only are PMA and 510(k) processes applicable to different types of devices, they have different purposes.
510(k): A 510(k) is intended to demonstrate that the device for which approval is being sought is as safe and effective as a currently marketed device that does not require a PMA.
PMA: A PMA is intended to prove that a new device is safe and effective for the end user. A PMA is much more detailed and in-depth than a 510(k). Device manufacturers are typically required to present human clinical trial data, in addition to laboratory testing data.
The difference in complexity between a PMA and 510(k) also affects the time needed to process the submissions. The FDA typically accepts or rejects a 510(k) submission within 30-90 days, at which point the device is posted to the FDA’s 510(k) database. A PMA submission can take up to 180 days to be processed, at which point the FDA can approve or deny the application. The FDA may also issue an “approvable” or “not approvable” letter, which the applicant can choose to respond to, thereby adding time to the submission process.
PMA application methods
There are a number of types of PMA application methods. While most devices which require a PMA will follow the traditional process, be sure to verify that you are using the correct application process to maximize your chances for success and avoid unnecessary delays:
Traditional PMA
The most common method for attaining FDA clearance for Class III devices, the traditional PMA is the appropriate option for most devices that have completed clinical testing.
Modular PMA
The modular PMA is the appropriate application method for devices that have not yet completed clinical testing. Applicants complete individual “modules,” with final confirmation granted once all sections are completed. For additional information on specific requirements of a modular PMA, read the FDA’s Premarket Approval Application Modular Review.
Product Development Protocol
Use the Product Development Protocol (PDP) with medical devices that are based on well-established technology. The PDP process for gaining market approval merges the clinical evaluation and development of information, and involves an agreement between the manufacturer and the FDA. The process provides the advantage of early predictability for the manufacturer and allows early interaction that can identifyFDA concerns as soon as possible in the development process. Because the PDP identifies the agreed upon design and development details, a completed PDP is considered to have an approved PMA. For additional information, read more about the FDA’s PMA Application Methods.
Humanitarian Device Exemption
A Humanitarian Use Device (HUD) is specifically defined as a device intended to benefit patients that are affected by a disease or condition that affects less than 8,000 individuals in the U.S. per year. TheHumanitarian Device Exemption (HDE) approval process is designed to encourage clinical activity around rare conditions, and does have certain restrictions, including:
- After receiving HDE approval, a HUD is eligible to be sold for profit only if the device is intended to address a disease or condition that occurs primarily in pediatric patients, or occurs in pediatric patients in small numbers.
- If an HDE is approved to be sold for profit, the FDA will determine an annual distribution number(ADN). Any devices sold beyond the ADN limit are required to be sold for no profit.
For more information see the FDA’s explanation of the Humanitarian Device Exemption.
CBER Submissions
There are two centers within the FDA responsible for evaluating medical devices. While the majority of devices will go through the Center for Devices and Radiological Health (CDRH), some will be managed by The Center for Biologics Evaluation and Research (CBER). CBER regulates medical devices related to blood and cellular products, including blood collection and processing procedures as well as cellular therapies. This ebook focuses on submissions made through the CDRH, but you can view CBER Regulatory Submissions – Electronic and Paper for more information on the CBER process.
To continue reading this eBook, including a walk through of the different types of required and optional FDA meetings and communications, a detailed list of the contents of a traditional PMA submission, and an overview of quality management system requirements, please register to download the full version.

An overview of 21 CFR Part 11 regulations for medical device companies
What is 21 CFR Part 11?
21 CFR Part 11 refers to the federal regulation that address electronic records and electronic signatures associated with FDA requirements. This single, relatively small, part of the Code of Federal Regulations is extremely significant for companies with FDA-regulated products because it impacts every document signature, electronic file, and FDA submission. Codified in 1997, interpretations of this FDA-issued regulation continue to be debated and re-evaluated as the technology supporting electronic records and signatures changes. In this article, we’ll discuss the regulation and generally accepted interpretations.
Note that discussions and statements in this document are our observations only and should not be taken as fact. You can refer directly to the regulation here.
Part 11: General Provisions
The General Provisions section of 21CFR11 addresses the scope of the regulation, when and how it should be implemented, and defines some of the key terms used. It states that the purpose of Part 11 is to define the criteria under which electronic records, electronic signatures, and handwritten signatures attached to electronic records are equivalent to, and as reliable as, handwritten signatures on paper documents.
Fundamentally, any record that is maintained, used, or submitted under any FDA records regulation is subject to Part 11, and the FDA will accept electronic records in lieu of paper records if an organization can prove that their records and systems meet the Part 11 requirements.
The General Provisions subpart also sets forth a number of definitions, and we’ve listed the ones that are most significant to our discussion here:
- Closed System: A computer system or software whose access is controlled by the same people who are responsible for the information stored in the system. Because the opposite of a closed system, and “open system,” is subject to additional scrutiny be sure that you are able to thoroughly explain and provide documentation for a decision to classify your system as a “closed system.”
- Open System: A computer system or software whose access is not controlled by the same people who are responsible for the information stored in the system.
- Digital Signature: An electronic signature created in a manner that can be verified, ensures the identity of the signer, and maintains the integrity of the document and signature. This often involves the use of cryptography and/or biometric data.
- Electronic Signature: Symbols that represent a legally binding equivalent to an individual’s handwritten signature (as adopted and authorized by the signer).
Part 11: Electronic Records
The Electronic Records section sets forth the requirements for administration of closed and open electronic record-keeping systems, then discusses signature manifestations and requirements for establishing a link between signatures and records.
Part 11 defines a “closed system” as any computer system in which the users controlling access to the system are the same people who are responsible for the data in the system. Today, most systems can be classified as closed systems, but take special care to document control procedures around software that is hosted offsite or classified as a SaaS solution.
This section of the regulation deals with the controls that need to be in place for all applicable electronic record systems by defining:
- Procedures to ensure that all electronic records are authentic, have integrity, and can ensure confidentiality (where that is appropriate).
- Validation requirements for systems that maintain electronic records to ensure that all records are accurate, reliable, and that the system performs consistently according to regulatory requirements.
- Audit trail requirements for all regulated records to ensure a complete history of all changes to records are maintained.
- Controls around system access and document signatures.
Part 11: Electronic Signatures
The Electronic Signatures section defines the components of electronic signatures and the required controls and procedures necessary for using them.
In general, an organization must be able to demonstrate that electronic signatures:
- Are unique to each individual, and that the individual assigned an electronic signature has had their identity and level of authorization verified.
- Must be based either on biometric data (such as fingerprints) or made up of two distinct pieces (ie: a User ID and password)
- Require appropriate controls to ensure that they are verified periodically, cannot be used by someone other than the intended user, and are immediately deactivated if compromised in any way.
Practical application of 21CFR Part 11 for regulatory affairs professionals
21 CFR Part 11 is a critical regulation, and one that can be open to interpretation. Below, we cover some of the key areas that should be of concern for RA professionals. This is an overview of key areas only, and should not be taken as complete instruction or guidance for 21CFR part 11 compliance.
System compliance and validation
Any system that you are using to store electronic records that fall under FDA regulations needs to be compliant with Part 11. This includes everything from spreadsheets to full-featured RIM and document management systems.
Software vendors will often document how their systems are developed to be compliant, and may even support system validation during implementation - but it is ultimately the responsibility of the user organization to ensure that their systems and processes are compliant with Part 11. System validation is the process of documenting that your system meets all of the Part 11 requirements. Software vendors can support this process by ensuring that their systems are built on a highly secured infrastructure that can be demonstrated and proven.
The Rimsys system was built from the ground up to meet the stringent requirements of not only 21 CFR Part 11, but other industry standards and good practices guidelines (GxP). We have put in place a rigorous validation program, built by industry experts and supported by a secure and well-documented infrastructure. For more information, visit the Rimsys Security and Privacy page.
Audit trails
Audit trails are the required system logs that track the who, when, and what of every change made to data that falls under Part 11. Audit trails should be generated and time-stamped by the system, with no ability for users to change that information. Audit trails serve two purposes under 21 CFR Part 11:
- To demonstrate that documented policies and procedures are being followed, including that only users with the appropriate authority are managing data.
- To prove that data retention policies are being adhered to (see below).
At any time, you should be able to view the history of any record, from a Design History File to a submission document, in order to determine what changes have been made, when they were made, and by whom.
Record retention
21 CFR Part 11 specifies that electronic records must be protected and readily available throughout the defined record retention period. Additionally, 21 CFR Part 820 specifies that records related to the quality, manufacturer, regulatory submissions, or any other data that falls under FDA regulation, should be maintained for the life of the medical device and for a minimum of two years from the date of first commercial distribution. This is often referred to as “cradle to grave” tracking.
This means that regulatory professionals need to not only be aware of their company’s record retention policy, but need to ensure that any system being used to track regulatory submissions or other data subject to audit meets Part 11 and Part 820 requirements. Note that record retention requirements apply also to paper records where they are the source document.
Electronic and digital signatures
An important piece of 21 CFR Part 11 is its definition of electronic and digital signatures. “Electronic signature” is used to define any set of symbols that are used in place of a handwritten signature, whereas a “digital signature” is an electronic signature based on methods that ensure the identity of the signer where the integrity of the data can be verified. A digital signature can be based on biometric data (such as fingerprints) or secure user IDs and passwords that are controlled to ensure only one authorized user can use the signature.
As a regulatory affairs professional, you should ensure that:
- Everyone on your team who needs to sign documents has their own unique digital signature and understands the importance of protecting it. Sharing of electronic credentials is a common FDA audit observation. Also ensure that users who are not required to sign documents have appropriate access to data to discourage other users from sharing login credentials with them.
- You are following your company’s policies concerning electronic signature audits so that passwords remain updated and strong and signatures are revoked when a user leaves or changes positions.
- You immediately report any possible loss, theft, or sharing of user credentials or devices that generate identification codes.
While 21 CFR Part 11 is usually considered more of a “quality regulation,” it is important that regulatory teams within medical device organizations fully understand this regulation and its compliance implications. To learn more about the regulations, click below to read our regulatory brief.

Is a medical device accessory a medical device?
Rimsys’ own James Gianoutsos recently contributed an article on www.meddeviceonline.com discussing FDA’s guidance document describing accessories and classification pathways.
On Dec. 20, 2017, the FDA issued Medical Device Accessories – Describing Accessories and Classification Pathways: Guidance for Industry and Food and Drug Administration Staff, which applies to the Center for Devices and Radiological Health (CDRH) and Center for Biologics Evaluation and Research (CBER) for combination products.
The guidance document offers welcomed clarity on the role of an “accessory” and its regulatory relationship to its parent device. As always, guidance documents are not legally enforceable; rather, they describe the Agency’s current thinking on a topic and should be viewed only as recommendations, unless specific regulatory or statutory requirements are cited.
The guidance explains which devices FDA generally considers “accessories” and describes the processes under Section 513(f)(6) of the Federal Food, Drug, and Cosmetic Act (FD&C Act) to allow requests for risk- and regulatory control-based classification of accessories. In other words, it specifically details what is and is not an accessory, as well as the regulatory routes to classification.
The updated guidance was derived from an August 2017 amendment to section 513(f) of the FD&C Act (FDA Reauthorization Act of 2017 (Pub. L. 115-52)) to state that “the Secretary shall … classify an accessory under [section 513] based on the risks of the accessory when used as intended and the level of regulatory controls necessary to provide a reasonable assurance of safety and effectiveness of the accessory, notwithstanding the classification of any other device with which such accessory is intended to be used.”
The amendment allows for some accessories to have a lower risk profile than that of their parent device and, therefore, may warrant being regulated in a lower class. As classifications for accessories are now risk-based, it provides manufacturers with regulatory flexibility to loosen some of the regulatory burdens on accessories that may not have the same risk profile as their parent devices.
For example, an accessory to a class III parent device may pose lower risk that could be mitigated through general controls, or a combination of general and special controls, and thus could be regulated as class I or class II. A common example of this would be a ventilation system (parent device) with a face mask (accessory).
Additionally, the guidance details the applicable definitions within Section IV: Definitions:
- Accessory — “A finished device that is intended to support, supplement, and/or augment the performance of one or more parent devices.”
- Component (21 CFR 820.3(c)) — “[A]ny raw material, substance, piece, part, software, firmware, labeling, or assembly which is intended to be included as part of the finished, packaged, and labeled device.”
- Finished Device (21 CFR 820.3(l)) — “[A]ny device or accessory to any device that is suitable for use or capable of functioning, whether or not it is packaged, labeled, or sterilized.”
- Parent Device — “A finished device whose performance is supported, supplemented, and/or augmented by one or more accessories.”
Accessory classification policy
The risks of an accessory are the risks it presents when used with the corresponding parent device as intended. To classify an accessory, FDA addresses the following two questions:
- Is the article an accessory? This can be answered by determining the intended use of the accessory. Is it intended for use with one or more parent devices, and does it support, supplement, and/or augment the performance of one or more parent devices?
- What is the risk of the accessory when used as intended with the parent device(s), and what regulatory controls are necessary to provide a reasonable assurance of its safety and effectiveness? This can be answered by providing a detailed risk assessment, outlining the potential hazards and reasonable regulatory and quality controls necessary to assure the accessory’s safety and effectiveness.
Individual accessories may be classified pursuant to the same regulation as a corresponding parent device, when appropriate, or be regulated independently.
Once an accessory has been classified, there is another consideration manufacturers need to decide: the Unique Device Identifier (UDI) rule. Not only does your parent device need to have a UDI, but any and all accessories each need to be assigned a UDI.
As 21 CFR 801 Labeling, Subpart B, Section 801.20(a) states: “(1) The label of every medical device shall bear a unique device identifier (UDI) that meets the requirements of this subpart and part 830 of this chapter,” and “(2) Every device package shall bear a UDI that meets the requirements of this subpart and part 830 of this chapter.”
Further, 21 CFR 830 UDI, Subpart A, Section 830.3 defines “finished device” and “device package” as follows:
- Finished device* means any device or accessory to any device that is suitable for use or capable of functioning.
- Device package means a package that contains a fixed quantity of a particular version or model of a device.
*Note that, although “medical device” and “finished device” are not consistent terminology used within the chapters, the terms are one and the same.
In short, any sellable finished device must bear a UDI, either on the device itself, on the device package, or both. Components to the finished device (i.e., service components and spare parts kits) are not considered accessories, and therefore are not required to bear a UDI.
What does this ultimately mean for manufacturers?
If there was any confusion as to whether a specific accessory is classified as a medical device, it has now been clarified, or at least partially clarified, depending on your specific situation. If there is still confusion among your engineering and regulatory teams, FDA recommends contacting them, via the accessory classification process outlined in the guidance, to classify the accessory appropriately. FDA will treat each accessory classification request as a Q-Submission. Requests may be for a new accessory type (new classification), an existing accessory type (reclassification), or classification of a new accessory type through the de novo process.
A gap analysis should be performed to identify a thorough and complete list of your current and future accessories to determine applicability to the guidance document. Justification also should be documented, should an accessory not apply to the guidance document. Additionally, internal procedures and the process associated with assigning UDIs may need to be updated to ensure there are no compliance gaps.

Podcast – Streamlining the MDSAP device marketing authorization and facility registration process
I had the opportunity to sit down with Jon Speer, Founder & VP of QA/RA at Greenlight Guru to record a podcast to discuss streamlining the MDSAP Marketing Authorization & Facility Registration Process.
If you are not familiar with this topic, you need to hear this.
We discuss:
- Why your regulatory team may be perceived as a bottleneck and why it is important everyone needs to be on the same page about when the product can be released.
- The connection between marketing authorization and facility registration required for various countries – United States, European Union, or elsewhere.
- The need to get organized, get better systems in place, and stay compliant when an auditor comes through your door or when you plan to sell into markets.
- Why small and large companies need to get organized now (i.e. small companies have too much information to maintain, organize, and track while large companies may have resources but suffer from miscommunication and disjointed processes.)
- The need to not be complacent and not be afraid to change systems. Rimsys was created to help regulatory professionals successfully and efficiently handle documentation.
You can hear the podcast below or at the Greenlight Guru blog.




