

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

The beginner's guide to the FDA De Novo classification process
This article is an excerpt from The beginner's guide to the FDA De Novo classification process ebook.
Contents
- Introduction
- Chapter 1: What is an FDA De Novo request?
- Chapter 2: Contents of a De Novo request
- Chapter 3: Submitting a De Novo request
- Appendix A: Acceptance review checklist
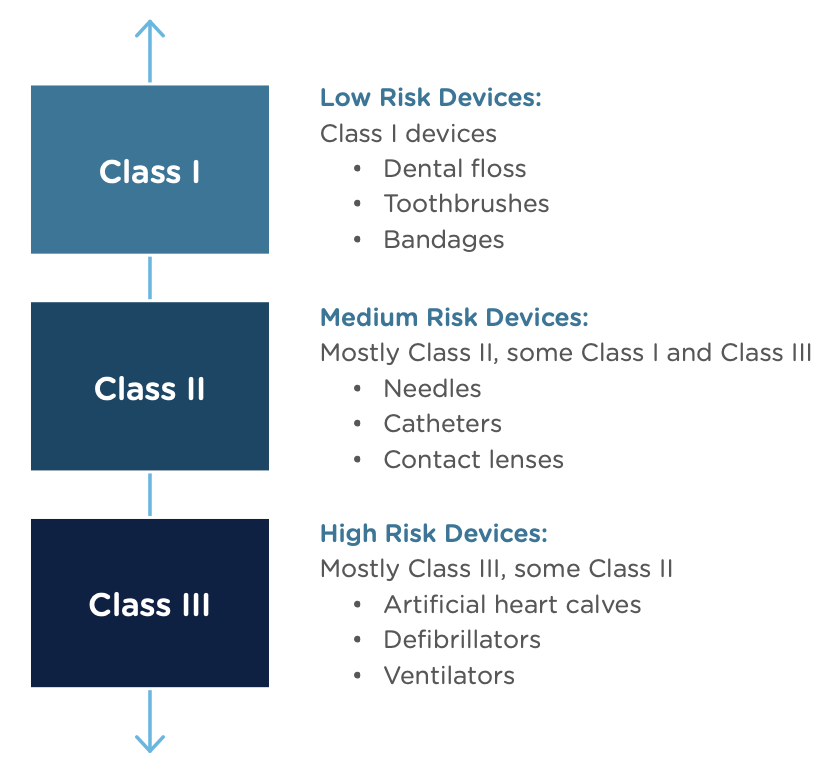
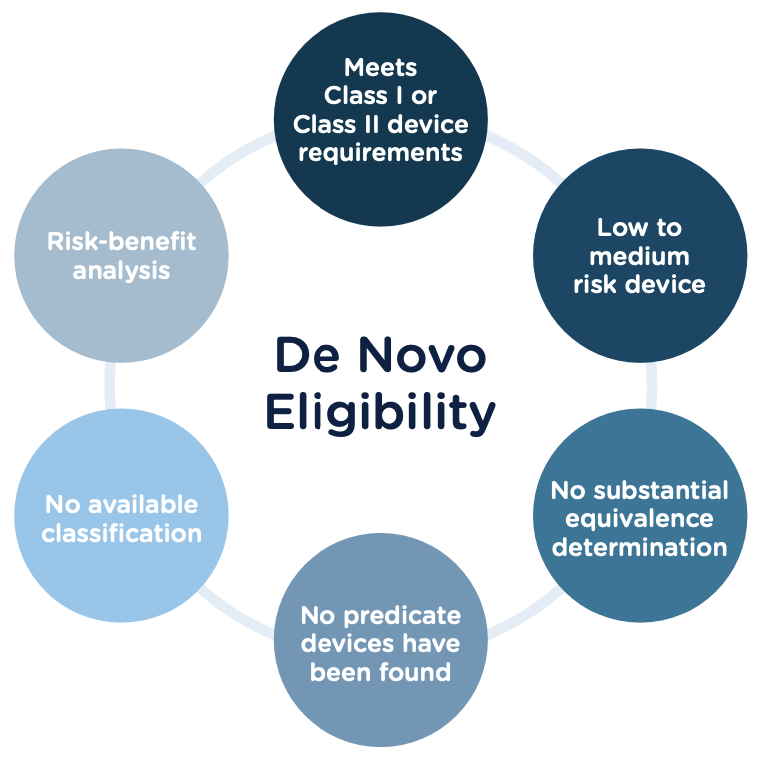
Congratulations, you have successfully developed a new medical device! Now you need to take it to market. Normally in the United States this would mean completing a 510(k) submission. However, the 510(k) relies on “substantial equivalence”—a comparison to a similar device already on the market (also called a predicate device) to assess the risk profile of the new device. What if your device is totally new, and there isn’t a similar device to compare it to? Enter the FDA De Novo process. The De Novo process provides a pathway to market for novel devices with a low to medium risk profile.
What does De Novo mean?
According to the Merriman-Webster dictionary, de novo is a Latin word meaning “as if for the first time; or anew.” Perfectly fitting that the FDA uses this term “De Novo” to describe market approval requests for new medical devices or technology where there is no comparable predicate device on the market.
The Food and Drug Administration Modernization Act of 1996 provided the FDA with the authority to create the De Novo Classification Process. It's a process that uses a risk-based strategy for a new, novel kind of medical device, in vitro diagnostic, or medical software solution whose type has previously not been identified and/or classified. It’s a process by which a novel medical device can be classified as a Class I or Class II device, instead of being automatically classified as Class III, which may not be appropriate. Before the implementation of the De Novo process in 1997, all the “not substantially equivalent” (NSE) products were required to be initially classified as a Class III device. But for a lot of devices, this risk class didn’t really make sense. The De Novo process provides a pathway for more accurate classifications of novel, lower-risk devices.
October, 2021, the FDA released a final guidance document "De Novo Classification Process (Evaluation of Automatic Class III Designation)" to provide guidance to the requester (also known as the manufacturer) and the FDA on the process for the submission and review of a De Novo Classification Request under section 513(f)(2) of the Federal Food, Drug, and Cosmetic Act (the FD&C Act). This process provides a pathway to an initial Class I or Class II risk classification for medical devices for which general controls or general and special controls, provide a reasonable assurance of safety and effectiveness, but for which there is no legally marketed predicate device. This guidance document replaced the "New Section 513(f)(2) – Evaluation of Automatic Class III Designation, Guidance for Industry and CDRH Staff" document, dated February 19, 1998.
Consistent with the final rule, the FDA updated the guidance documents below to provide recommendations for submitting De Novo requests, as well as criteria and procedures for accepting, withdrawing, reviewing, and making decisions on De Novo requests, effective January 3, 2022.
- User Fees and Refunds for De Novo Classification Requests
- FDA and Industry Actions on De Novo Classification Requests: Effect on FDA Review clock and Goals
- Acceptance Review for De Novo Classification Requests
The 510(k) and the De Novo processes are similar in that they are both pathways to market for medical devices with low to moderate risk, which is Class I and Class II. The biggest difference between the two is that the 510(k) heavily relies on the concept of "substantial equivalence" to an existing medical device. You must prove this to get the clearance of your 510(k) submission. In the De Novo process, there isn’t a product currently on the market that is “substantially equivalent” to yours, so it’s like starting with a clean slate. For more on the 510(k) process, see our Beginner’s Guide to the 510(k) ebook.
A result of the De Novo process to be aware of is that a successful submission will lead to a new predicate device type that someone else can reference to bring their product to market through the 510(k) process. You’ve done all the work, so now it’s available for anyone to use to provide "substantial equivalence".
De Novo history/timeline
Preparing a De Novo request
1. Do your research! Be sure to complete all the necessary research prior to your submission. You want to be sure that your device is not substantially equivalent to an existing device. Resources to review include:
- The Center for Devices and Radiological Health (CDRH)
- U.S. FDA Device Classification Database
- Device Classification Under Section 513(f)(2)(De Novo)
2. A De Novo request can be submitted with or without a preceding 510(k). There are two options for when you can submit a De Novo request:
Option A: After receiving a not substantially equivalent (NSE) determination (that is, no predicate, new intended use, or different technological characteristics that raise different questions of safety and effectiveness) in response to a 510(k) submission.
Option B: If you’ve determined, after extensive research, that there is no legally marketed device on which to base a determination of substantial equivalence.
3. Be sure all fees are paid to the FDA in advance of submitting a De Novo request. The FDA’s fiscal year begins in October and runs through the following September. Fees have increased each year since they were introduced, but the FDA’s percentage of reviews completed within the 150-day window has increased as well.
A business that is qualified and certified as a “small business” is eligible for a substantial reduction in most of the FDA user fees, including De Novo. The CDRH is responsible for the Small Business Program that determines whether a business is qualified.
Medical Device User Fee Amendments (MDUFA) guidance documents can provide more detailed information about all FDA user fees.
4. The initial request process serves only to determine if the De Novo request is administratively acceptable based upon the Acceptance Checklist. The initial acceptance is followed by substantive review which will determine the final risk classification of your device.
5. A Pre-Submission (Pre-Sub) is a formal written request for feedback from the FDA that is provided in formal written form, and then followed by a meeting. Although a Pre-Sub is not required prior to a De Novo request, it can be extremely helpful to receive early feedback, especially for devices that have not previously been reviewed under a 510(k). If you think you would like to submit a pre-sub first, there are suggested guidelines for submission you should consider:
- Describe your rationale for a Class I or Class II classification for your device.
- Provide the search results of FDA public databases and other resources used to determine that no legally marketed device and no classification for the same device type exists.
- Provide a list of regulations and/or product codes that may be relevant.
- Provide a rationale for why the subject device does not fit within and/or is different from any identified classification regulations, based on available information.
- Identify each health risk associated with the device and the reason for each risk.
- Briefly describe any ongoing and/or planned protocols/studies that need to be completed in order to collect the necessary data to establish the device’s risk profile.
- Provide information regarding the safety and effectiveness of the device. Cite the types of valid scientific evidence you anticipate providing in your De Novo request, including types of data/studies relating to the device’s safety and effectiveness.
- Briefly describe any ongoing and/or planned protocols/studies that need to be completed to collect the necessary safety and effectiveness data.
- Provide protocols for non-clinical and clinical studies (if applicable), including how they will address the risks you anticipate and targeted performance levels that will demonstrate that general controls or general and special controls are sufficient to provide reasonable assurance of safety and effectiveness.
- Share any proposed mitigation measure(s)/control(s) for each risk, based on the best available information at the time of the submission. Highlight which mitigations are general controls and which are special controls and provide details on each.
- Include any other risks that may be applicable, in addition to those identified in the Pre-Sub, given the indications for use for the device.
- If applicable, provide any controls that should be considered to provide a reasonable assurance of safety and effectiveness for the device.
- Provide any non-clinical study protocols that are sufficient to allow the collection of data from which conclusions about device safety and/or effectiveness can be drawn. These protocols should address whether the identified level of concern is the appropriate level of concern for the device software, and if any additional biocompatibility and/or sterility testing is required.
- If clinical data is needed, provide information to show that the proposed study design and selected control groups are appropriate?
6. The FDA will attempt to review the De Novo request submission within 15 calendar days of receipt of the request to make a determination that the submission is declined or accepted for review. If they are unable to complete the review within the 15 days, your submission will automatically move to “accepted for review” status. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/de-novo-classification-process-evaluation-automatic-class-iii-designation
7. There are times when the FDA will refund your application fee. They have created a guidance document “User Fees and Refunds for De Novo Classification Requests” for the purpose of identifying:
- the types of De Novo requests subject to user fees
- exceptions to user fees
- the actions that may result in refunds of user fees that have been paid
When is a De Novo request subject to a user fee?
When will the FDA refund a De Novo user fee?
What fee must be paid for a new device submission following a De Novo “decline” determination?
To continue reading this eBook including a detailed walk-through of all the Traditional 510(k) components, submission requirements and timelines, and an overview of the other 510(k) forms including the Abbreviated 510(k) and the Special 510(k), please register to download the full version.

The ultimate guide to the medical device single audit program (MDSAP)
This article is an excerpt from The ultimate guide to the medical device single audit program (MDSAP) ebook.
Table of contents
- What is MDSAP?
- History of MDSAP
- Who is responsible for the MDSAP?
- How does an MDSAP audit work?
- Audit sequence
- You got a nonconformity – now what?
- What does an MDSAP audit cost?
- Why choose the MDSAP certification process?
- Potential disadvantages of the MDSAP
- Ready to participate? – Here’s how to get started
- Completing a successful MDSAP audit
The Medical Device Single Audit Program (MDSAP) was designed and developed to allow a single audit of a medical device manufacturer to be applied to all country markets whose regulatory authorities are members of the program. The MDSAP provides efficient and thorough coverage of the standard requirements for medical device manufacturer quality management systems, and requirements for regulatory purposes (ISO 13485:2016). In addition, there are specific requirements of each medical device regulatory authority participating in the MDSAP that must be met:
- Conformity Assessment Procedures of the Australian Therapeutic Goods (Medical Devices) Regulations (TG(MD)R Sch3)
- Brazilian Good Manufacturing Practices (RDC ANVISA 16)
- Medical Device Regulations of Health Canada (ISO 13485:2003)
- Japan Ordinance on Standards for Manufacturing Control and Quality Control of Medical Devices and In Vitro Diagnostic Reagents (MHLW Ministerial Ordinance No 169)
- Quality System Regulation (21 CFR Part 820), and specific requirements of medical device regulatory authorities participating in the MDSAP program.
This means that a report from a single MDSAP audit of a medical device manufacturer would be accepted as a substitute for routine inspections by all the member Regulatory Authorities (RAs) across the world. There are currently five participating Regulatory Authorities (RA) representing the following countries: Australia, Brazil, Canada, Japan and the USA.
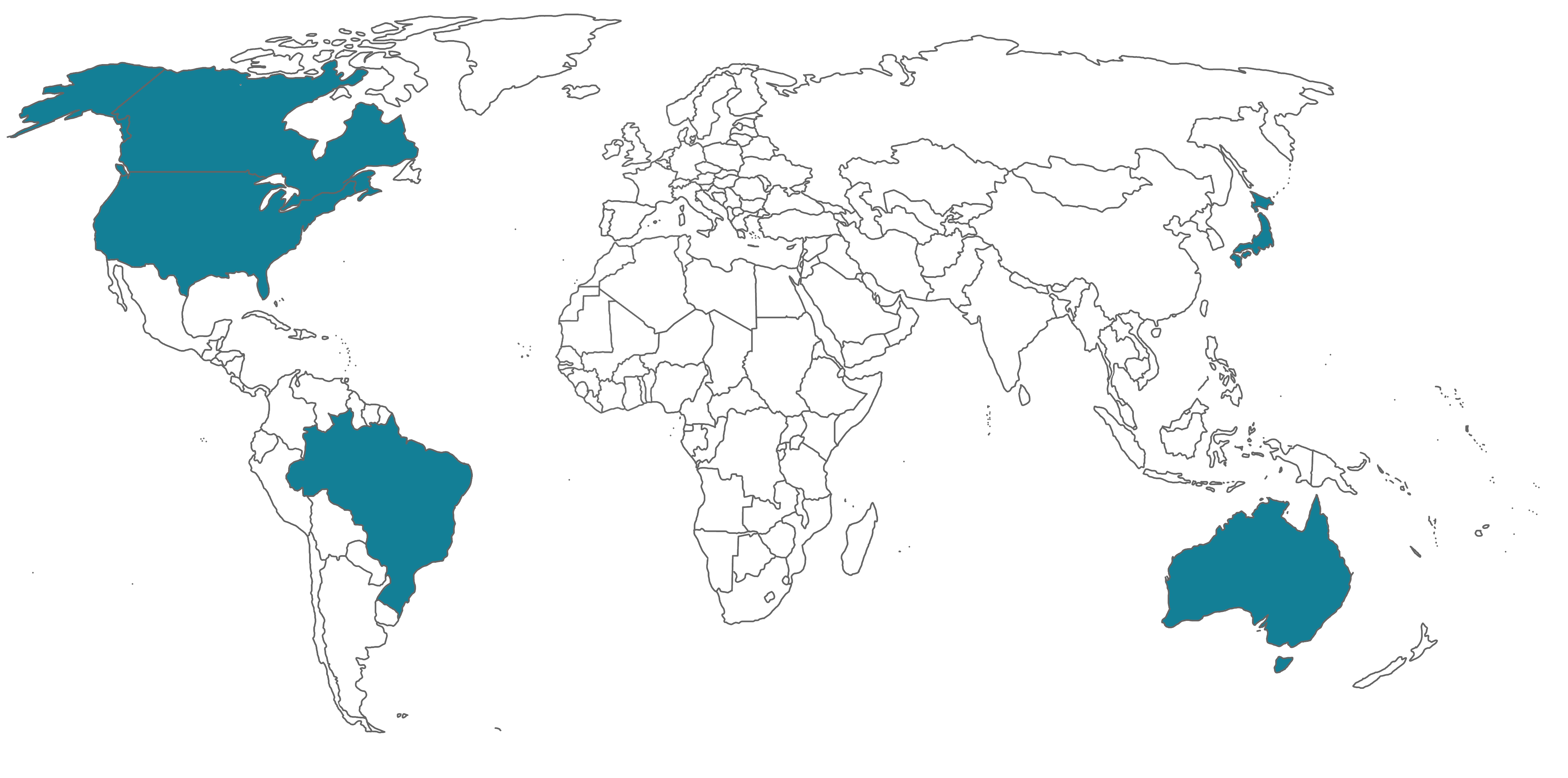
In April, 2021, the RAs released an “Audit Approach” document (MDSAP AU P0002.006) that combines the formerly separate MDSAP Audit Model and Process Companion documents into a single guidance document. It includes guidance for assessing the conformity of each process and includes an audit sequence, instructions for auditing each specific process, and identifies links that highlight the interactions between the processes.
In March 2012 the US FDA announced that they had approved a final pilot guidance document “Guidance for Industry, Third Parties and Food and Drug Administration Staff: Medical Device ISO 13485:2003 Voluntary Audit Report Submission Pilot Program.” This allowed the owner or operator of a medical device manufacturing facility to be removed from FDA’s routine inspection work plan for 1 year upon completing a ISO 13485:2003 audit. This guidance document went into effect in June 2012, and was intended as an interim measure while a single audit program was being developed.
This pilot program was not very successful and few companies signed up because they did not see any advantage in participating. The manufacturer had to pay for a third party to inspect their facilities, generate a report, and share the inspection results back to the FDA. Many companies were reluctant to contract “someone else” to perform their inspection when they could easily wait for the FDA to conduct an inspection for free.
During its inaugural meeting in Singapore in 2012, the International Medical Device Regulators Forum (IMDRF) appointed a working group to develop a set of documents for a harmonized third-party auditor system. Hence, the “Medical Device Single Audit Program” (MDSAP) was formed. The concept was similar to the FDA’s original idea of creating a third-party auditor to help reduce their workload of performing regulatory audits of medical device manufacturers’ quality management systems. This new approach would consist of a single audit that would review regulatory QMS compliance, conducted by a third-party, who would later be called an Auditing Organization (AO).
From January 2014 to December 2016, five countries participated in a Medical Device Single Audit Program Pilot. In June 2017, a report was generated summarizing the outcomes of prospective “proof- of-concept” criteria established to confirm the success of the program. The outcomes are documented in the final MDSAP Pilot Report and recommended that the program become fully active and open to any manufacturer who requested this type of audit.
The governing body of the MDSAP is the Regulatory Authority Council (RAC), which is composed of two senior managers (and a few other staff members) from each participating RA. They are responsible for executive planning, strategic priorities, setting policy, and making decisions on behalf of the MDSAP International Consortium. The RAC also reviews and approves documents, procedures, work instructions, and more. The mission of the MDSAP International Consortium is to jointly leverage regulatory resources to manage an efficient, effective, and sustainable single audit program focused on the oversight of medical device manufacturers on a global scale.
Other international partners that are involved in the MDSAP include:
MDSAP Observers:
- European Union (EU)
- United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA)
- The World Health Organization (WHO) Prequalification of In Vitro Diagnostics (IVDs) Program
MDSAP Affiliate Members:
- Argentina’s National Administration of Drugs, Foods and Medical Devices (ANMAT)
- Republic of Korea’s Ministry of Food and Drug Safety
- Singapore’s Health Sciences Authority (HSA)
The observers and affiliate members are not the same as the participating member RA’s. The observers simply observe and/or contribute to RAC activities. Affiliate members, on the other hand, are interested in engaging in the MDSAP program and are subject to certain rules. They are only given access to a certain level of information about the manufacturers, audit dates, and information in audit reports.
They are also invited to attend sessions that are open to members, observers, and affiliates only.
Audits can also be conducted by MDSAP participating RAs at any time and for various reasons including:
- "For Cause" due to information obtained by the regulatory authority
- as a follow up to findings from a previous audit
- to confirm the effective implementation of the MDSAP requirements
The purpose of audits conducted by the RAs is to ensure appropriate oversight of the AOs MDSAP auditing activities. The AOs are appointed by the RAs and a list of the currently approved AO’s is published on the FDA website. Most AOs offer a broad range of management system certification services, beyond just medical devices. Manufacturers should verify that prospective AOs are clearly trained and perform MDSAP audits of medical devices.
AOs have the final word as to whether a manufacturer has met the requirements for the MDSAP during the execution of the audit and generation of the associated reports summarizing the results. MSDAP RAC participating RAs have the final decision regarding all development, implementation, maintenance, and expansion activities associated with the program.
Although an unannounced visit by an AO is rare, it can happen in circumstances where high-grade nonconformities have been detected.
To continue reading this eBook including a detailed look at the MDSAP audit process and grading, pros and cons of the approach, and how to get started please register to download the full version.

Rimsys Launches the Regulatory Execution Engine for MedTech
Spring 2026 embeds submission authoring, AI-poweredregulatory monitoring, and configurable impact workflows inside a single RIM platform,the first step toward Rimsys’ AI vision for global regulatory operations.
PITTSBURGH, PA, May 5, 2026 –Regulatory Information Management (RIM) software was built to store records.That foundation has served its purpose and reached its limit. Today, Rimsys announces the Spring 2026 release: aplatform designed not to hold regulatory data, but to executeon it.
Submission volumes are growing. Markets are multiplying. Regulatory change is accelerating. Spring 2026 gives regulatory teams the tools to keep pace: embedded authoring, reusable submission content, configurable impact workflows,and AI-powered intelligence, all inside a single platform.
"Our vision for Rimsys is a platform that makes regulatory expertise go further, companies move faster, and products reach more markets than any team could accomplish alone. Spring 2026 is another meaningful step toward that vision. We are embedding the tools and intelligence that allow regulatory affairs professionals to operate at a different level, doing more strategic work, entering markets faster, and staying ahead of regulatory change rather than reacting to it. What we are building next makes this release thestarting line." – James Gianoutsos, CEO
What Spring 2026 Delivers
A brand new website that provides in-depthinformation about the Rimsys offering and the benefits to MedTech manufacturers,including details on these new products:
Intelligence: AI-Powered Regulatory Monitoring
Rimsys Intelligence provides access to regulations, guidance documents, safety alerts, and legislationacross more than 90 countries. AI triage and prioritization surface the updates most relevant to each customer’s specific products and markets, eliminatinghours of manual surveillance and putting the right information in front of theright people.
When a changerequires action, teams can move directly from regulatory signal to impact assessment without a manual handoff. Intelligence represents Rimsys’ firstproduction deployment of context-aware AI operating across a customer’s liveregulatory data, a foundation that will expand significantly in future releases.
Advanced Submissions: A Unified Submission Execution Workflow
Advanced Submissions consolidates everything required to create, manage, and publish a regulatory submission into a single workflow inside Rimsys, eliminating the disconnected tools, manual reformatting, and version fragmentation that have defined submission work for too long. Three capabilities anchor it:
Rimsys Editor
The Rimsys Editor is the cornerstone of Advanced Submissions and the most significant capability in this release. It brings word-compatible authoring and editing natively inside Rimsys, fully compatible with Microsoft Word®, allowing regulatory teams to create, co-author, review, and publish submission content without leaving the platform for the first time.
The Editor supports real-time co-authoring, tracked changes and redlining, rich content including tables and images, document comparison, and PDF publishing with standardized headers, footers, and company branding applied automatically.AI-assisted authoring is available as a configurable option, enabling teams to summarize, refine, expand, and translate content within their workflow. Rimsys AI is human-in-the-loop by design.
Universal Submissions
Universal Submissions enables teams to build from a single universal template (an IMDRF Technical Document) with content automatically mapped into market-specific templates. One master structure, many markets,without rebuilding from scratch.
Reusable Submissions
Reusable Submissions takes a completed submission from one market and uses it as the starting point for a new one. The system automatically maps content into the target market’s template, carrying applicable sections forward reducing the content creation time up to 90% and compressing the time required to enter each additional market.
Configurable Impact Surveys: Governed Change Assessment at Scale
Impact Surveys are now fully configurable. Templates can be defined for specific change event types, tied to countries orregistrations, and triggered automatically from Rimsys Intelligence findings replacing ad hoc assessments with repeatable, governed workflows. This integration creates a direct line from change event toregulatory scope, with results tracked in a single audit-ready trail.
A Platform Built for What’s Next
Spring 2026 establishes more than a set of new capabilities. It establishes the execution infrastructure, structured data model, and embedded AI foundation on which Rimsys’ longer-term vision is being built.
That vision: aworld where regulatory experts are amplified by intelligence, not constrained by information. Where the knowledge required to enter a new market, interpret a regulatory change, or scope a submission is instantly available to every member of the team. Where regulatory operations scale not by spreading experts thin, but by giving them tools that multiply their impact.
Spring is the first production step in that direction. Every submission authored inside the platform, every intelligence signal triaged by AI, and every impact assessment connected to structured regulatory data deepens the foundation. Future releases will build on it directly, expanding AI capabilities, automating more of theregulatory workflow, and ultimately enabling teamsto do work that today requires external expertise to be done inside Rimsys.
Regulatory Execution as a Business Lever
Spring 2026 is built to move metrics that matter: reduced submission cycle time variance,improved approval predictability, lower marginal effort per market, and increased team capacity without proportional headcount growth. For executive leadership, earlier approvals translate directly into faster market access and accelerated revenue recognition.
Availability
Spring 2026 isnow Generally Available. Existing customers on the Organizer product will retain access to their current experience.
To learn more about the Spring 2026 release and how Rimsys can accelerate your regulatory operations, visit rimsys.io or contact your Rimsys representative.
About Rimsys
Rimsys is the heart of regulatory operations for the medical device industry and the platformat the center of an AI-driven transformation in how regulated products reachglobal markets. A living, connected regulatory platform, Rimsys keepsregulatory intelligence, product data, approvals, and change management continuously connected, enabling organizations to expand into global markets with speed, precision, and confidence. Enterprise-ready yet intuitive to use,Rimsys is trusted by 6 of the top 12 global MedTech manufacturers to acceleratetime to market and scale regulatory operations worldwide. To learn more, visit rimsys.io.
Media Contact
letschat@rimsys.io
rimsys.io

The Real Cost of “We’ll Build It Ourselves”
If you are reading this from inside a large MedTech organization, you may be thinking: we have ten times the engineering staff. Why can’t we just build this ourselves?
We-Should-Just-Build-This-Ourse…
It is a fair question.
But software has a well-known paradox. Adding more people to a complex project does not make it go faster. It usually makes it go slower. More coordination. More handoffs. More meetings about meetings. More surface area for misalignment
A large IT organization is optimized for breadth — supporting dozens of systems, managing infrastructure, keeping the lights on across the enterprise
That is valuable work.
But it is fundamentally different from building and sustaining a deep vertical product over a decade.
The people on your team have day jobs. They run devices through regulatory pathways, manage quality systems, support manufacturing, and commercialize products globally
Building a regulated platform is not a side quest.
It is a second company
What the Numbers Actually Look Like
When people compare license fees to internal builds, they stop at the wrong baseline
The real comparison is:
Licensing a specialized platform
versus
Standing up and operating a regulated software company inside your enterprise
Product management.
UX research.
Engineering.
Regulatory SMEs.
Validation and QA.
Security operations.
Compliance programs.
24/7 support.
Infrastructure.
Multi-year modernization
AI makes some of that faster.
It does not make any of it optional
With a specialized vendor, that investment is amortized across an entire customer base.
With an internal build, the full long tail of ownership falls on you
And most of that spend ends up recreating the 80 percent that has already been solved — all because someone decided the remaining 20 percent justified building from scratch
The return on that 20 percent rarely survives honest scrutiny.
The Questions That Should Keep You Honest
It is easy to get excited about how fast something can be built.
The harder exercise is asking what happens in year three, year five, year eight
When your VP of Regulatory Affairs leaves, who maintains validation documentation?
When regulations change across jurisdictions simultaneously, who redesigns workflows and pushes a validated release before the deadline?
When an auditor asks for change control history and disaster recovery test results, who is accountable?
Internal initiatives often stumble not because engineers cannot prototype, but because sustaining them for a decade is brutally hard
Sponsors move on. Budgets change. Teams reorganize.
Regulatory systems do not get to pause.
They must remain inspection-ready through acquisitions, divestitures, and leadership turnover
Systems of record are commitments, not experiments
AI Changed the Tools, Not the Gravity
I am genuinely excited about what AI enables. It will reshape regulatory operations, reduce headcount growth, compress timelines, and raise expectations for every vendor in this space
What it has not done is repeal gravity.
Most of what AI replaces today is busy work. That is enormously valuable. But busy work was never the strategic bottleneck
The hard parts remain.
- Deciding submission strategy.
- Interpreting regulator feedback.
- Designing defensible workflows.
- Staying inspection-ready.
- Running global rollouts
Agents help teams move faster.
They do not decide what is safe, defensible, or durable
In MedTech, software is not just built.
It is designed, governed, operated, and defended
And gravity still applies.

Day Zero Is Easy. Day One Is Where It Gets Hard
There is something I keep coming back to in these conversations.
You can go from idea to prototype incredibly fast right now. That is the day-zero problem, and AI has essentially solved it. You can spit out working code, scaffold an integration, and stand up a proof of concept in a week
But the nuance around an actual business workflow — the day one and beyond activities — those are dramatically harder than day zero ever was
Software engineering done well is craftsmanship.
There is more to it than generating code and turning a prototype into something a regulated enterprise can depend on. It means thinking about edge cases, failure modes, upgrade paths, observability, and long-term operability. It means deleting as much as adding. Simplifying interfaces. Collapsing concepts down to what actually matters
Inside my own teams, I see impressive first versions all the time.
That is not the hard part anymore.
The hard part is everything that comes after
We-Should-Just-Build-This-Ourse…
Faster Engineering Just Pushes Work Somewhere Else
There is a tradeoff that rarely makes it into the first ROI spreadsheet.
AI compresses build cycles. In regulated companies, that speed shows up downstream. More releases mean more validation, more SOP updates, more training, more compliance review, and more audit prep
Engineering gets cheaper.
Governance becomes the constraint
There is also a subtler version of this problem.
Agents make it easy to generate output at scale. More workflows. More automation. More code.
But in regulated environments, every new service or automation path increases surface area. More things to secure. More things to validate. More things to explain to auditors
Speed without discipline creates complexity faster.
For CTOs, that is an architectural concern.
For Regulatory leaders, that is an inspection risk.
Are You Trying to Be a Software Company?
This is the part of these conversations that most often gets skipped.
A MedTech company is not a software shop. Most are largely outsourced IT organizations, and there is nothing wrong with that. The core business is devices, science, R&D, manufacturing quality, clinical programs, and global commercialization
When internal teams talk about building major regulatory platforms, the question is not whether they can spin up a prototype.
It is whether they want to operate a full-time software company inside their enterprise
Building software at scale is a people problem. It is not a technology problem. The constraint is coordination, judgment, institutional knowledge, and sustained focus over years
The people problem does not get fixed by agents and AI.
Regulatory platforms are deeply vertical. They encode jurisdiction-specific rules, regulator expectations, submission templates, QMS integrations, inspection trails, and post-market obligations
That knowledge is earned slowly.
It lives in product decisions, data models, operating procedures, and support playbooks.
AI will reshape how these platforms evolve.
It does not remove the learning curve that created them
AI Agents and the Confidence Shift Inside MedTech IT
In some MedTech IT planning meetings, a new kind of confidence has started to show up.
Not everywhere. Not in every organization. But often enough that it is worth paying attention to.
It is subtle. Casual. The kind that appears when something new begins to feel inevitable
A VP of IT or a CIO sits in a planning meeting. Someone pulls up a demo. An AI agent drafts a regulatory summary, generates a workflow, and scaffolds an integration. It looks impressive. It is impressive
Then someone says it:
Why are we paying for a platform when we could build this ourselves?
I understand the impulse.
SaaS valuations are volatile. Boards are pressing on efficiency. Hiring is under scrutiny everywhere. AI arrives, and suddenly there is a clean story. Automate friction. Avoid headcount growth. Modernize everything
Some of that is real.
I am optimistic about AI. In the right hands, it is a genuine superpower
But hope, cost pressure, aggressive marketing, and very human psychology are colliding right now. That collision is shaping how executives talk about technology strategy
In regulated industries, that matters.
The Confirmation Bias Problem
When leaders already feel pressure to reduce costs or flatten organizations, they naturally gravitate toward stories that validate those instincts. Flashy demos and headlines about agents replacing departments reinforce the belief that a breakthrough must be right around the corner
Once that belief sets in, messy operational details get discounted. Risk gets deferred.
That does not make the technology fake.
It does explain why ambition so often outruns delivery reality
For CTOs and Regulatory leaders, this is the moment to slow the conversation down.
Because prototypes are not platforms.
What AI Actually Changes
Years ago, Harvard Business Review wrote about the “hidden data factory,” the idea that organizations accumulate thousands of small one-off efforts to clean data, reconcile systems, patch workflows, and keep operations moving. No single fix ever justifies a major initiative. In aggregate, it quietly costs millions
That concept maps directly to what AI is good at today.
Inside engineering organizations, we call this work toil.
The repetitive, manual, low-judgment effort that keeps systems running but should not consume the time of highly trained people. Environment setup. Data reconciliation. Migration scripts. Test generation. Documentation drafts. Classification lookups. Compliance artifacts
AI is excellent at eliminating toil. It removes friction, collapses queues, and gives teams back time
In regulated environments, that is meaningful.
But here is the distinction that matters:
Eliminating toil does not eliminate accountability
It does not remove the need for architecture, UX design, validation strategy, regulatory interpretation, or operational ownership.
What it does is allow smaller, more senior teams to focus on the work that actually differentiates platforms.
That is very different than from saying agents replace the platforms themselves

Why MedTech Regulatory Teams Are Delegating EUDAMED to IT
And Why That Creates Bigger Problems Over Time
As EUDAMED implementation accelerates and the UDI/Devices module becomes mandatory in May of 2026, many MedTech companies have made a seemingly practical decision. They hand EUDAMED compliance to IT.
At first glance, the logic feels sound. EUDAMED is a system. It requires integrations, data transmission, and technical connectivity. IT already owns those capabilities, so the project lands there.
But this handoff reveals a deeper misunderstanding of what EUDAMED actually represents. It is a tool that enables manufacturers to meet ongoing regulatory obligations that touch product data, submissions, post-market activities, and lifecycle management. EUDAMED also enables manufacturers’ ACTOR partners like Notified Bodies, Authorized Representatives, Importers, and Distributors to meet their obligations under those EU regulations. Treating it as an isolated, one-time IT project creates risks to EU regulatory compliance that grow and spread across partners over time. MDR/IVDR regulatory compliance cannot be established and maintained with a one-time technical integration.
The first problem with delegating EUDAMED to IT is what it signals internally. It frames the regulation as a single event rather than a continuous program.
EUDAMED is not just about getting data into a database. It requires ongoing updates tied to regulatory changes, product modifications, vigilance activities, certificates, and market status. Every change across the product lifecycle can trigger downstream updates in EUDAMED.
When EUDAMED is positioned as a one-time event, organizations underestimate the scope, effort, and ownership required to maintain compliance over time. That gap does not show up immediately. It appears months later when updates are missed; data falls out of sync, or responsibilities become unclear.
IT teams often take on EUDAMED with the expectation that once the pipes are built, the work is largely done. In reality, the opposite happens.
As regulatory data changes, IT becomes the default escalation point for updates they do not own and cannot validate. They are asked to manage regulatory timelines, interpret data requirements, and support continuous updates that fall outside their core mandate.
This creates friction on both sides. Regulatory teams feel blocked by technical dependencies. IT teams feel burdened by compliance work they were never meant to manage. Over time, updates slow down, workarounds emerge, and risk quietly increases.
The most damaging consequence of delegating EUDAMED to IT is architectural. When EUDAMED operates outside of a centralized Regulatory Information Management system, organizations lose the opportunity to reuse data and reduce burden across the business.
Most of the data required for EUDAMED already exists within product information management and resource planning systems. Product registrations, certificates, submissions, UDI, and post-market data are not new. They are part of the regulatory lifecycle. When EUDAMED is disconnected from RIM, teams are forced to duplicate work, reconcile inconsistencies, and manually manage updates across systems.
Instead of becoming a natural extension of regulatory operations, EUDAMED turns into another silo. One that increases workload rather than streamlining it.
Establishing and maintaining regulatory information in EUDAMED is a regulatory obligation, not a technical one. While IT plays a critical role in enablement and integration, there should be a strong partnership between regulatory and IT (or a third-party submitter), but IT shouldn’t own it completely.
When EUDAMED is managed as part of a centralized RIM approach, organizations gain consistency, traceability, and reuse. Regulatory teams can leverage existing data, control updates at the source, and reduce the ripple effects of change across departments. IT supports the infrastructure, but regulatory owns the process.
This shift also changes how organizations think about compliance. Instead of reacting to EUDAMED as a standalone requirement, they treat it as part of a broader regulatory operating model that supports long-term compliance and growth.
Delegating EUDAMED to IT is rarely a conscious strategy. It is usually a symptom of fragmented regulatory operations and unclear ownership.
As MedTech companies scale globally and regulatory expectations continue to evolve, these handoffs become harder to sustain. EUDAMED exposes the cost of treating regulatory compliance as a series of isolated projects rather than an ongoing operational discipline.
The companies that navigate EUDAMED successfully are not the ones with the most complex integrations. They are the ones that anchor EUDAMED within regulatory operations, supported by centralized RIM systems that establish data consistency and reduce duplication, improve visibility, and spread the burden across the organization in a controlled way.

Agentic AI and the Future of Regulatory Operations
Why Regulatory Operations Is Ready for Agentic AI
Regulatory operations teams are under increasing pressure. Global regulatory complexity is rising, data volumes continue to grow, and teams are expected to move faster, often without additional headcount. At the same time, employee turnover and fragmented systems make it harder to maintain continuity and institutional knowledge.
As outlined in the RIM & AI Maturity in MedTech Executive Guide, many organizations are still operating with scattered regulatory data, reactive processes, and manual workflows. These conditions increase compliance risk and slow growth.
This environment has created the conditions where a more advanced form of AI can deliver meaningful value. That is where agentic AI comes into play, not as a replacement for regulatory expertise, but as a way to strengthen how regulatory operations function day to day.
What Is Agentic AI and Why It Matters
Most AI used in regulatory environments today is assistive. It helps classify documents, extract text, or answer questions when prompted. Agentic AI goes further by operating within defined workflows and processes.
Agentic AI systems can monitor structured regulatory data continuously, identify upcoming risks or deadlines, recommend actions based on rules and historical context, and surface next steps within governed processes. Instead of responding to requests, agentic AI supports execution by working alongside regulatory teams inside their operational systems.
The distinction is important. In regulated environments, value does not come from generative output alone. It comes from intelligence that is embedded, auditable, and aligned with how regulatory work actually gets done.
Moving Regulatory Teams Off the Data Treadmill
The executive guide describes early-stage regulatory teams as being stuck on a back-office data treadmill. Highly skilled professionals spend a disproportionate amount of time searching for information, reconciling spreadsheets, and repeating manual tasks rather than applying their expertise strategically.
Agentic AI helps reduce this burden by continuously organizing and validating regulatory data, identifying missing metadata or inconsistencies early, and reducing reliance on individual memory or tribal knowledge. Over time, this improves not just efficiency, but operational resilience. Teams become less vulnerable to audits, turnover, and last-minute regulatory surprises.
Why Agentic AI Depends on Operational Maturity
One of the most important insights from the paper is that AI value scales with RIM maturity. Advanced AI capabilities are not effective without centralized regulatory information and standardized processes .
At higher maturity levels, AI can surface upcoming risks across markets and renewals, analyze submission history to recommend reusable content, and identify bottlenecks before they impact timelines. At this stage, agentic AI begins to function as an operational partner, helping teams anticipate issues rather than react to them.
This is also where many organizations encounter friction. Skipping foundational steps may create the appearance of progress, but it limits reliability and long-term impact. Agentic AI is only as effective as the data, governance, and workflows it operates within.
From Task Automation to Predictive Compliance
At the most mature stage of regulatory operations, AI becomes fully embedded in daily work. The guide describes this level as one where real-time monitoring, predictive analytics, and continuous improvement are standard practice .
In this environment, agentic AI supports predictive compliance by identifying emerging risks, highlighting resource constraints, and improving visibility across submissions and renewals. These insights allow teams to act earlier and with greater confidence.
The paper is clear on one point. AI enhances regulatory expertise, but it does not replace it. Human judgment remains essential for interpretation, decision-making, and accountability. The real value of agentic AI is that it frees regulatory professionals from low-value work so they can focus on the decisions that matter most .
Regulatory Operations as the Heart of Compliant Growth
The most significant impact of agentic AI is not automation alone. It is the elevation of regulatory operations from a reactive support function to the heart of compliant growth.
Organizations that invest in strong RIM foundations, data governance, and workflow integration are better positioned to apply AI in a way that is safe, scalable, and durable. When implemented thoughtfully, agentic AI helps regulatory operations keep pace with growth, reduce risk, and support faster, more confident decision-making across the business.





