

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

The beginner's guide to the FDA De Novo classification process
This article is an excerpt from The beginner's guide to the FDA De Novo classification process ebook.
Contents
- Introduction
- Chapter 1: What is an FDA De Novo request?
- Chapter 2: Contents of a De Novo request
- Chapter 3: Submitting a De Novo request
- Appendix A: Acceptance review checklist
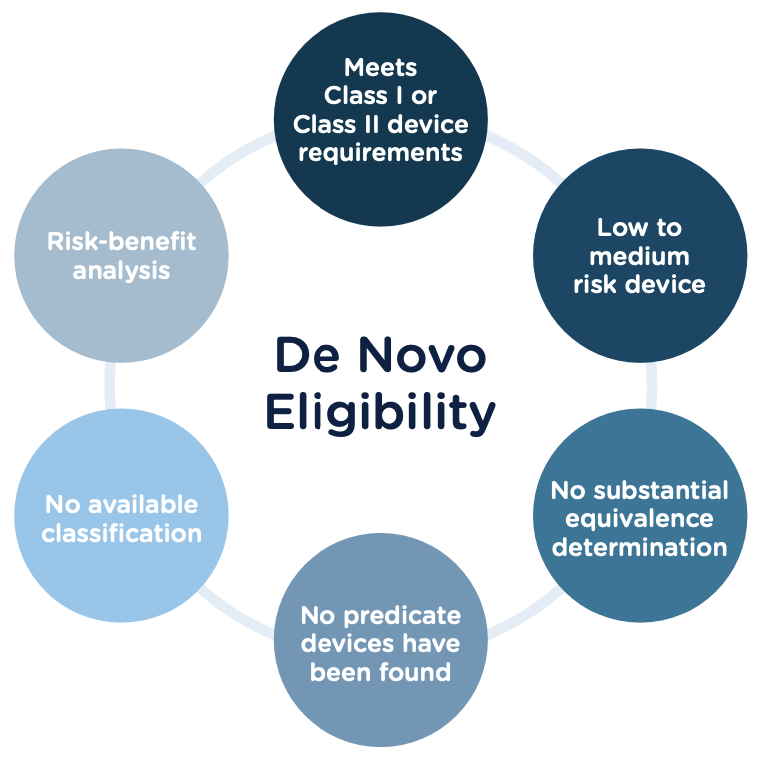
Congratulations, you have successfully developed a new medical device! Now you need to take it to market. Normally in the United States this would mean completing a 510(k) submission. However, the 510(k) relies on “substantial equivalence”—a comparison to a similar device already on the market (also called a predicate device) to assess the risk profile of the new device. What if your device is totally new, and there isn’t a similar device to compare it to? Enter the FDA De Novo process. The De Novo process provides a pathway to market for novel devices with a low to medium risk profile.
What does De Novo mean?
According to the Merriman-Webster dictionary, de novo is a Latin word meaning “as if for the first time; or anew.” Perfectly fitting that the FDA uses this term “De Novo” to describe market approval requests for new medical devices or technology where there is no comparable predicate device on the market.
The Food and Drug Administration Modernization Act of 1996 provided the FDA with the authority to create the De Novo Classification Process. It's a process that uses a risk-based strategy for a new, novel kind of medical device, in vitro diagnostic, or medical software solution whose type has previously not been identified and/or classified. It’s a process by which a novel medical device can be classified as a Class I or Class II device, instead of being automatically classified as Class III, which may not be appropriate. Before the implementation of the De Novo process in 1997, all the “not substantially equivalent” (NSE) products were required to be initially classified as a Class III device. But for a lot of devices, this risk class didn’t really make sense. The De Novo process provides a pathway for more accurate classifications of novel, lower-risk devices.
October, 2021, the FDA released a final guidance document "De Novo Classification Process (Evaluation of Automatic Class III Designation)" to provide guidance to the requester (also known as the manufacturer) and the FDA on the process for the submission and review of a De Novo Classification Request under section 513(f)(2) of the Federal Food, Drug, and Cosmetic Act (the FD&C Act). This process provides a pathway to an initial Class I or Class II risk classification for medical devices for which general controls or general and special controls, provide a reasonable assurance of safety and effectiveness, but for which there is no legally marketed predicate device. This guidance document replaced the "New Section 513(f)(2) – Evaluation of Automatic Class III Designation, Guidance for Industry and CDRH Staff" document, dated February 19, 1998.
Consistent with the final rule, the FDA updated the guidance documents below to provide recommendations for submitting De Novo requests, as well as criteria and procedures for accepting, withdrawing, reviewing, and making decisions on De Novo requests, effective January 3, 2022.
- User Fees and Refunds for De Novo Classification Requests
- FDA and Industry Actions on De Novo Classification Requests: Effect on FDA Review clock and Goals
- Acceptance Review for De Novo Classification Requests
The 510(k) and the De Novo processes are similar in that they are both pathways to market for medical devices with low to moderate risk, which is Class I and Class II. The biggest difference between the two is that the 510(k) heavily relies on the concept of "substantial equivalence" to an existing medical device. You must prove this to get the clearance of your 510(k) submission. In the De Novo process, there isn’t a product currently on the market that is “substantially equivalent” to yours, so it’s like starting with a clean slate. For more on the 510(k) process, see our Beginner’s Guide to the 510(k) ebook.
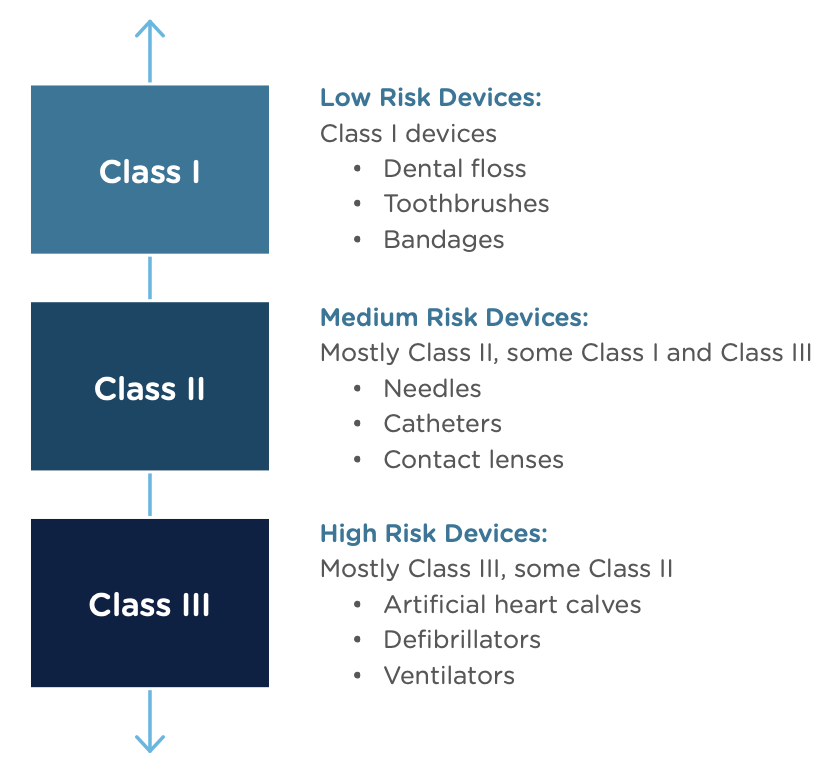
A result of the De Novo process to be aware of is that a successful submission will lead to a new predicate device type that someone else can reference to bring their product to market through the 510(k) process. You’ve done all the work, so now it’s available for anyone to use to provide "substantial equivalence".
De Novo history/timeline
Preparing a De Novo request
1. Do your research! Be sure to complete all the necessary research prior to your submission. You want to be sure that your device is not substantially equivalent to an existing device. Resources to review include:
- The Center for Devices and Radiological Health (CDRH)
- U.S. FDA Device Classification Database
- Device Classification Under Section 513(f)(2)(De Novo)
2. A De Novo request can be submitted with or without a preceding 510(k). There are two options for when you can submit a De Novo request:
Option A: After receiving a not substantially equivalent (NSE) determination (that is, no predicate, new intended use, or different technological characteristics that raise different questions of safety and effectiveness) in response to a 510(k) submission.
Option B: If you’ve determined, after extensive research, that there is no legally marketed device on which to base a determination of substantial equivalence.
3. Be sure all fees are paid to the FDA in advance of submitting a De Novo request. The FDA’s fiscal year begins in October and runs through the following September. Fees have increased each year since they were introduced, but the FDA’s percentage of reviews completed within the 150-day window has increased as well.
A business that is qualified and certified as a “small business” is eligible for a substantial reduction in most of the FDA user fees, including De Novo. The CDRH is responsible for the Small Business Program that determines whether a business is qualified.
Medical Device User Fee Amendments (MDUFA) guidance documents can provide more detailed information about all FDA user fees.
4. The initial request process serves only to determine if the De Novo request is administratively acceptable based upon the Acceptance Checklist. The initial acceptance is followed by substantive review which will determine the final risk classification of your device.
5. A Pre-Submission (Pre-Sub) is a formal written request for feedback from the FDA that is provided in formal written form, and then followed by a meeting. Although a Pre-Sub is not required prior to a De Novo request, it can be extremely helpful to receive early feedback, especially for devices that have not previously been reviewed under a 510(k). If you think you would like to submit a pre-sub first, there are suggested guidelines for submission you should consider:
- Describe your rationale for a Class I or Class II classification for your device.
- Provide the search results of FDA public databases and other resources used to determine that no legally marketed device and no classification for the same device type exists.
- Provide a list of regulations and/or product codes that may be relevant.
- Provide a rationale for why the subject device does not fit within and/or is different from any identified classification regulations, based on available information.
- Identify each health risk associated with the device and the reason for each risk.
- Briefly describe any ongoing and/or planned protocols/studies that need to be completed in order to collect the necessary data to establish the device’s risk profile.
- Provide information regarding the safety and effectiveness of the device. Cite the types of valid scientific evidence you anticipate providing in your De Novo request, including types of data/studies relating to the device’s safety and effectiveness.
- Briefly describe any ongoing and/or planned protocols/studies that need to be completed to collect the necessary safety and effectiveness data.
- Provide protocols for non-clinical and clinical studies (if applicable), including how they will address the risks you anticipate and targeted performance levels that will demonstrate that general controls or general and special controls are sufficient to provide reasonable assurance of safety and effectiveness.
- Share any proposed mitigation measure(s)/control(s) for each risk, based on the best available information at the time of the submission. Highlight which mitigations are general controls and which are special controls and provide details on each.
- Include any other risks that may be applicable, in addition to those identified in the Pre-Sub, given the indications for use for the device.
- If applicable, provide any controls that should be considered to provide a reasonable assurance of safety and effectiveness for the device.
- Provide any non-clinical study protocols that are sufficient to allow the collection of data from which conclusions about device safety and/or effectiveness can be drawn. These protocols should address whether the identified level of concern is the appropriate level of concern for the device software, and if any additional biocompatibility and/or sterility testing is required.
- If clinical data is needed, provide information to show that the proposed study design and selected control groups are appropriate?
6. The FDA will attempt to review the De Novo request submission within 15 calendar days of receipt of the request to make a determination that the submission is declined or accepted for review. If they are unable to complete the review within the 15 days, your submission will automatically move to “accepted for review” status. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/de-novo-classification-process-evaluation-automatic-class-iii-designation
7. There are times when the FDA will refund your application fee. They have created a guidance document “User Fees and Refunds for De Novo Classification Requests” for the purpose of identifying:
- the types of De Novo requests subject to user fees
- exceptions to user fees
- the actions that may result in refunds of user fees that have been paid
When is a De Novo request subject to a user fee?
When will the FDA refund a De Novo user fee?
What fee must be paid for a new device submission following a De Novo “decline” determination?
To continue reading this eBook including a detailed walk-through of all the Traditional 510(k) components, submission requirements and timelines, and an overview of the other 510(k) forms including the Abbreviated 510(k) and the Special 510(k), please register to download the full version.

The ultimate guide to the medical device single audit program (MDSAP)
This article is an excerpt from The ultimate guide to the medical device single audit program (MDSAP) ebook.
Table of contents
- What is MDSAP?
- History of MDSAP
- Who is responsible for the MDSAP?
- How does an MDSAP audit work?
- Audit sequence
- You got a nonconformity – now what?
- What does an MDSAP audit cost?
- Why choose the MDSAP certification process?
- Potential disadvantages of the MDSAP
- Ready to participate? – Here’s how to get started
- Completing a successful MDSAP audit
The Medical Device Single Audit Program (MDSAP) was designed and developed to allow a single audit of a medical device manufacturer to be applied to all country markets whose regulatory authorities are members of the program. The MDSAP provides efficient and thorough coverage of the standard requirements for medical device manufacturer quality management systems, and requirements for regulatory purposes (ISO 13485:2016). In addition, there are specific requirements of each medical device regulatory authority participating in the MDSAP that must be met:
- Conformity Assessment Procedures of the Australian Therapeutic Goods (Medical Devices) Regulations (TG(MD)R Sch3)
- Brazilian Good Manufacturing Practices (RDC ANVISA 16)
- Medical Device Regulations of Health Canada (ISO 13485:2003)
- Japan Ordinance on Standards for Manufacturing Control and Quality Control of Medical Devices and In Vitro Diagnostic Reagents (MHLW Ministerial Ordinance No 169)
- Quality System Regulation (21 CFR Part 820), and specific requirements of medical device regulatory authorities participating in the MDSAP program.
This means that a report from a single MDSAP audit of a medical device manufacturer would be accepted as a substitute for routine inspections by all the member Regulatory Authorities (RAs) across the world. There are currently five participating Regulatory Authorities (RA) representing the following countries: Australia, Brazil, Canada, Japan and the USA.
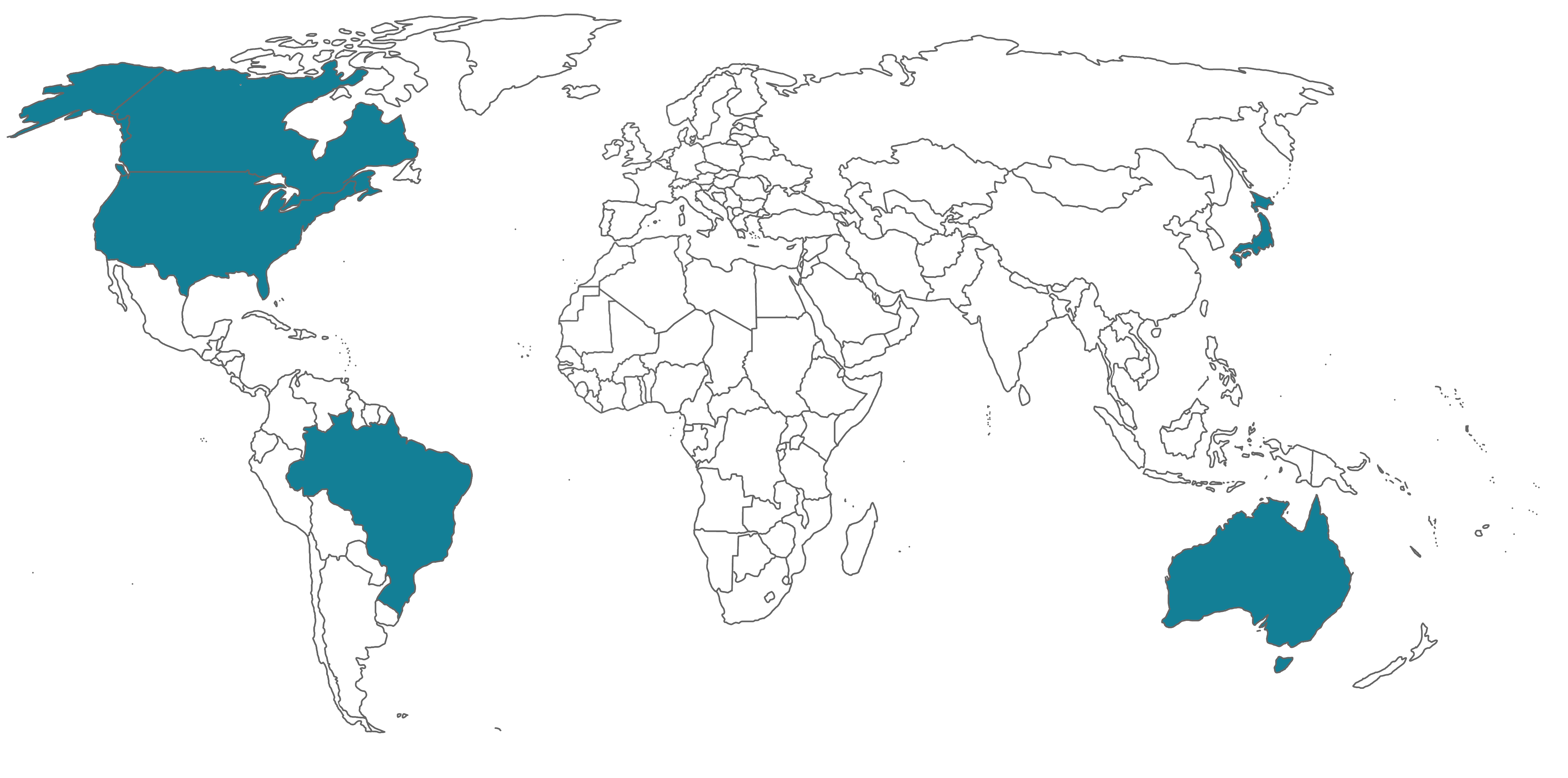
In April, 2021, the RAs released an “Audit Approach” document (MDSAP AU P0002.006) that combines the formerly separate MDSAP Audit Model and Process Companion documents into a single guidance document. It includes guidance for assessing the conformity of each process and includes an audit sequence, instructions for auditing each specific process, and identifies links that highlight the interactions between the processes.
In March 2012 the US FDA announced that they had approved a final pilot guidance document “Guidance for Industry, Third Parties and Food and Drug Administration Staff: Medical Device ISO 13485:2003 Voluntary Audit Report Submission Pilot Program.” This allowed the owner or operator of a medical device manufacturing facility to be removed from FDA’s routine inspection work plan for 1 year upon completing a ISO 13485:2003 audit. This guidance document went into effect in June 2012, and was intended as an interim measure while a single audit program was being developed.
This pilot program was not very successful and few companies signed up because they did not see any advantage in participating. The manufacturer had to pay for a third party to inspect their facilities, generate a report, and share the inspection results back to the FDA. Many companies were reluctant to contract “someone else” to perform their inspection when they could easily wait for the FDA to conduct an inspection for free.
During its inaugural meeting in Singapore in 2012, the International Medical Device Regulators Forum (IMDRF) appointed a working group to develop a set of documents for a harmonized third-party auditor system. Hence, the “Medical Device Single Audit Program” (MDSAP) was formed. The concept was similar to the FDA’s original idea of creating a third-party auditor to help reduce their workload of performing regulatory audits of medical device manufacturers’ quality management systems. This new approach would consist of a single audit that would review regulatory QMS compliance, conducted by a third-party, who would later be called an Auditing Organization (AO).
From January 2014 to December 2016, five countries participated in a Medical Device Single Audit Program Pilot. In June 2017, a report was generated summarizing the outcomes of prospective “proof- of-concept” criteria established to confirm the success of the program. The outcomes are documented in the final MDSAP Pilot Report and recommended that the program become fully active and open to any manufacturer who requested this type of audit.
The governing body of the MDSAP is the Regulatory Authority Council (RAC), which is composed of two senior managers (and a few other staff members) from each participating RA. They are responsible for executive planning, strategic priorities, setting policy, and making decisions on behalf of the MDSAP International Consortium. The RAC also reviews and approves documents, procedures, work instructions, and more. The mission of the MDSAP International Consortium is to jointly leverage regulatory resources to manage an efficient, effective, and sustainable single audit program focused on the oversight of medical device manufacturers on a global scale.
Other international partners that are involved in the MDSAP include:
MDSAP Observers:
- European Union (EU)
- United Kingdom’s Medicines and Healthcare products Regulatory Agency (MHRA)
- The World Health Organization (WHO) Prequalification of In Vitro Diagnostics (IVDs) Program
MDSAP Affiliate Members:
- Argentina’s National Administration of Drugs, Foods and Medical Devices (ANMAT)
- Republic of Korea’s Ministry of Food and Drug Safety
- Singapore’s Health Sciences Authority (HSA)
The observers and affiliate members are not the same as the participating member RA’s. The observers simply observe and/or contribute to RAC activities. Affiliate members, on the other hand, are interested in engaging in the MDSAP program and are subject to certain rules. They are only given access to a certain level of information about the manufacturers, audit dates, and information in audit reports.
They are also invited to attend sessions that are open to members, observers, and affiliates only.
Audits can also be conducted by MDSAP participating RAs at any time and for various reasons including:
- "For Cause" due to information obtained by the regulatory authority
- as a follow up to findings from a previous audit
- to confirm the effective implementation of the MDSAP requirements
The purpose of audits conducted by the RAs is to ensure appropriate oversight of the AOs MDSAP auditing activities. The AOs are appointed by the RAs and a list of the currently approved AO’s is published on the FDA website. Most AOs offer a broad range of management system certification services, beyond just medical devices. Manufacturers should verify that prospective AOs are clearly trained and perform MDSAP audits of medical devices.
AOs have the final word as to whether a manufacturer has met the requirements for the MDSAP during the execution of the audit and generation of the associated reports summarizing the results. MSDAP RAC participating RAs have the final decision regarding all development, implementation, maintenance, and expansion activities associated with the program.
Although an unannounced visit by an AO is rare, it can happen in circumstances where high-grade nonconformities have been detected.
To continue reading this eBook including a detailed look at the MDSAP audit process and grading, pros and cons of the approach, and how to get started please register to download the full version.

Taking SaMDs to market in the US: How is the FDA regulating adaptive machine learning algorithms?
Rimsys recently held a panel discussion, Taking SaMDs to market in the US. During it, Prabhu Raghavan, Principal at MDQR Solutions, and Rimsys Chief Solutions Officer, Brad Ryba, shared an overview of SaMDs and provided their insights about getting and maintaining market clearance for them in the United States. Topics ranged from FDA risk classifications and submissions, cybersecurity best practices, and machine learning algorithms, which brought about an important question: How is the FDA currently regulating adaptive machine learning algorithms in SaMDS?
Adaptive machine learning algorithms use post-market data in real time and evolve their models based on the data they're consuming. As such, every patient utilizing a device with adaptive machine learning algorithms may have a new model compared to the previous patient. While the FDA doesn't have any formal guidance on the subject just yet, manufacturers can work with the FDA to get a plan in place for maintaining a state of validation post market.
Watch the snippet from the webinar to learn about taking a staged approach with the FDA to get a proper validation plan in place.
To watch all discussion topics, download the webinar replay here.

The five guiding principles for machine learning-enabled medical devices using PCCPs
On October 24th, 2023, the FDA, Health Canada, and the MHRA published a joint document providing harmonization for machine learning-enabled medical devices (MLMD) that use predetermined change control plans (PCCPs). PCCPs are plans proposed by the manufacturer that state the specific modifications to a MLMD, the process for implementing these modifications, and the assessment of impacts from them.
The document details five guiding principles for MLMDs in an effort to set a foundation for PCCPs and encourage collaboration on them. According to the UK government’s website, these principles are:
- Focused and Bounded: Describing specific changes that a manufacturer intends to implement.
- Risk-based: The intent, design, and implementation of a PCCP are driven by a risk-based approach that adheres to the principles of risk management.
- Evidence-based: Demonstrating that benefits outweigh the risks throughout the product lifecycle.
- Transparent: Provide clear and appropriate information and detailed plans for ongoing transparency to all stakeholders, from patients to healthcare professionals.
- Total Product Lifecycle Perspective: Improve the quality and integrity of a PCCP by continually considering the perspectives of all stakeholders.
Here are some examples of how these principles could be applied:
- Focused and bounded: A manufacturer of an MLMD that diagnoses cancer might develop a PCCP to implement a change to the algorithm that improves its accuracy in detecting a specific type of cancer.
- Risk-based: A manufacturer of an MLMD that monitors a patient's vital signs might develop a PCCP to implement a change to the algorithm that reduces the likelihood of false alarms.
- Evidence-based: A manufacturer of an MLMD that delivers medication to patients might develop a PCCP to implement a change to the algorithm that improves the accuracy of the dosage.
- Transparent: A manufacturer of an MLMD might publish a white paper that describes the device's algorithm and how it was developed and tested. The manufacturer might also make available a user manual that provides clear instructions on how to use the device safely and effectively.
- Total product lifecycle perspective: A manufacturer of an MLMD might collect feedback from patients and healthcare professionals on how the device is performing after it is marketed. The manufacturer might also use this feedback to identify and address any potential problems with the device.
The five guiding principles for MLMDs using PCCPs are based on the 10 guiding principles for Good Machine Learning Practices (GMLP) published in 2021, which were designed to help medical device manufacturers develop and deploy machine learning models that are safe, effective, and high quality. Similarly, the goal of these five guiding principles is to help MLMD manufacturers develop and maintain safe and effective products that meet the needs of patients and healthcare professionals. They are also intended to streamline the regulatory process for MLMDs, making it easier for manufacturers to bring new products to market and make updates to existing products in a timely manner.
If you’re looking for additional information about MLMD requirements in the US, join Rimsys and MDQR Solutions for Taking SaMDs to market in the US on Thursday, November 30th, at 1 PM ET. We’ll discuss the various types of SaMDs, considerations to make when obtaining market clearance, and how the FDA is regulating AI/ML in devices. Those interested in attending can register here: Taking SaMDs to market in the US.

An overview of the Accreditation Scheme for Conformity Assessment (ASCA) Program
Introduced in 2021, the Accreditation Scheme for Conformity Assessment (ASCA) program is a voluntary program that allows device manufacturers to use an ASCA-accredited testing laboratory to conduct testing to be included in premarket submissions to the FDA. The ASCA program is designed to speed up FDA review times, reduce guesswork on documentation to provide in a premarket submission, and improve the quality of testing conducted.
In September of 2023, the program was converted from a pilot to a permanent volunteer program. This blog post provides an overview of the ASCA program to help medtech companies determine if they should consider participating.
How the ASCA program works
Under the ASCA program, the FDA grants ASCA Recognition to qualified accreditation bodies, which certifies testing laboratories to perform premarket testing for medical device companies. The FDA uses international conformity assessment standards and FDA-identified ASCA program specifications to assess ASCA Accreditation status to qualified testing laboratories.
The ASCA program includes FDA-recognized consensus standards and related test methods across two scopes:
- Biocompatibility: This scope includes standards for testing the safety of medical devices when they come into contact with human tissues.
- Basic safety and essential performance: This scope includes standards for testing the safety and performance of medical devices.
It’s important to note that medical device manufacturers should continue to reference additional FDA-recognized standards and provide declarations of conformity in their premarket submissions, but standards outside of biocompatibility and basic safety and essential performance are not eligible for ASCA program benefits.
Medical device manufacturers can choose an ASCA-accredited laboratory to conduct its testing for FDA premarket submissions. The ASCA-accredited testing laboratory will provide the manufacturer with all information listed in the ASCA program specifications.
For each eligible FDA-recognized consensus standard and test method, the manufacturer will need to document results on an ASCA Summary Test Report. Upon testing completion, the device manufacturer will receive an ASCA Summary Test Report from the laboratory. When the manufacturer prepares their premarket submission for FDA approval, they will need to include a declaration of conformity with the ASCA Summary Test Report as part of their submission.
Benefits of participating in the ASCA program
Medical device companies that participate in the ASCA program can see a variety of benefits, including:
- Faster FDA review times: The FDA has stated that it expects to review premarket submissions that include ASCA Summary Test Reports more quickly than submissions that do not.
- More clarity on documentation needed in a premarket submission: The ASCA Summary Test Report template provides a guide on what information to include in the report, which can help device manufacturers avoid having to guess what the FDA is looking for. This can save time and resources and help to ensure that the premarket submission is complete and accurate.
- Improved quality of testing conducted: ASCA-accredited testing laboratories must meet rigorous standards for competence and impartiality.
- Enhanced credibility: Participating in the ASCA program can help device manufacturers enhance their credibility with the FDA and other stakeholders. This is because the ASCA program is designed to improve the quality and efficiency of the premarket review process.
While the FDA has not announced any plans to make the ASCA program mandatory, it has stated that it intends to review premarket submissions that include an ASCA Test Summary report faster. This means that device manufacturers who participate in the ASCA program may be more likely to receive faster approvals.
In addition, some countries outside of the United States require device manufacturers to use accredited testing laboratories for premarket approval. By participating in the ASCA program, device manufacturers can demonstrate that their testing is conducted to high standards and more easily meet the requirements of these countries.
Funded through the MDUFA V User Fee program, the FDA does not charge an additional fee for manufacturers to participate in the ASCA program. For more guidance and training on the ASCA program, see the FDA’s website.

Your eSTAR submission questions answered by FDA experts
Your eSTAR submission questions answered
Starting on October 1, 2023, all 510(k) submissions, unless exempted, will need to be submitted electronically using eSTAR. eSTAR, which is short for Electronic Submission Template and Resource, is a dynamic PDF submission template that contains automation, guides, integrated databases, integrated policies and procedures, and automatic verification to help users prepare a comprehensive medical device submission.
Rimsys recently hosted a webinar, eSTAR submissions overview and live Q&A with FDA, to help the medtech community prepare for the quickly approaching eSTAR deadline. Patrick Axtell, Assistant Director for Tools and Templates Team, and Sajjad Syed, Software Engineer for Tools and Templates Team, from FDA joined our panel of Rimsys regulatory experts to provide an overview of eSTAR, demo, and live question and answer session. If you're interested in watching the webinar replay, you can find it here.
Due to high participation, we couldn't answer all questions live. This blog post provides Patrick's and Sajjad's answers to questions we couldn't get to during the webinar. Read below to learn what other industry professionals want to know about eSTAR!
Q: In the past, the sections were numbered Section 10 - Device Description. How should section(s) be numbered in the eSTAR?
You may number attachments how you prefer, this includes numbering attachments according to the previous section numbers of the Format for Traditional and Abbreviated guidance document. However, the numbering hierarchy in the IMDRF documents, which the attachment questions in eSTAR correlate to, may be better, since that numbering scheme is internationally harmonized. So for example, if you are using the nIVD eSTAR, the IMDRF document specifies the Comprehensive Device Description would be numbered 2.04.01: https://www.imdrf.org/sites/default/files/docs/imdrf/final/technical/imdrf-tech-190321-nivd-dma-toc-n9.pdf.
Q: After October 1, 2023, will the In Vitro Diagnostics eSTAR Version be acceptable to use for a Dual 510(k) and CLIA Waiver by Application (Dual Submission)?
Yes, the next IVD eSTAR update will fully support Dual submissions, and the workaround we were previously implementing will no longer be needed. CLIA waiver submissions will continue to be submitted via current methods, and are not required to use eSTAR after Oct 1, 2023.
Q. It is not possible to view eSTAR online from cloud storage platforms. eSTAR like IFU Form and CDRH Form cannot be viewed online without users downloading these files. I will highly appreciate if someone from the panel can give a solution to this. I understand these are special pre-programmed PDFs but it will immensely help if these can be shared for online viewing without having users download these files.
You need to choose to open eSTAR in the default application from within the cloud application you use, you can’t just click on it, since it will open in the browser, and browsers can’t open dynamic PDFs like eSTAR, Form 3881, Form 3514, and other forms FDA uses.
We can’t say that all cloud applications will work, but using Box does work. Once eSTAR is loaded in, click on the three dots, then choose the application you have installed, see screenshots below. I have Adobe Acrobat 2017 and Adobe Acrobat Pro both installed.
Be aware that every time you save the form, you upload it back into the cloud, so depending on how large it is and how fast your internet connection is, it may take many seconds to save each time, though you will see a progress bar.
Q: Does the eSTAR submission need to be submitted by only the US Agent or can this be done by the Foreign Manufacturer?
510(k) eSTARs, like 510(k) eCopies, can be submitted by foreign applicants.
Q: Will it be mandatory to use the eStar template for PMA supplement submissions?
Yes, by the end of 2023, eSTAR for PMA will be released. Similar to eSTAR for 510(k), there will be a pilot, guidance, and a transition period before it becomes mandatory.
Q: When do you expect to release the next version or sub-version of the non-IVD eSTAR template (4.x or 5.0) and will it be more elaborate on cybersecurity?
We are at the mercy of updating when policies are updated, which is irregular and unpredictable. Please email CDRH management and recommend a more consistent (e.g. quarterly) policy deployment schedule so that eSTAR can also have a more predictable deployment schedule. We are updating approximately one to every two months currently. Please be sure to read the Version History on page 1 of eSTAR regarding versioning and which version to use.
Q: How should a device categorized as a breathing gas pathway device in accordance with ISO 18562 be handled in eSTAR? One cannot find ISO 18562 categorization for a device in the eSTAR form.
This is a regulatory and policy question, and we are not sure what is meant by the statement “be handled.” Please reach out to OPEQ Submission Support OPEQSubmissionSupport@fda.hhs.gov with this question and they will direct your question appropriately.
Q: How often does FDA anticipate revising the eSTAR form?
eSTAR updates are almost exclusively timed according to policy updates, which the Tools and Templates Team has no control over. The T&T Team, as well as other groups in CDRH, are pushing for a quarterly update, where any policy updates in a quarter have an implementation date set for the beginning of the next quarter (e.g. Oct 1st, Jan 1st, Apr 1st). Please email Center management and promote this.
Q: The guidance Format for Traditional and Abbreviated 510(k)s: Guidance for Industry and FDA staff recommends a specific format for submissions. Biocompatibility, for example, is section 15. Should attachments in the eSTAR submission cross-reference the sections cited in the guidance? (So still using biocompatibility as an example, attachment 15.1 would be Cytotoxicity, attachment 15.2 would be Sensitization and so on)
The Format guidance, as well as the eCopy guidance, RTA guidance, and many other guidances, no longer apply for eSTARs. Eliminating the applicability of guidances - simplifying the preparation process - is something we are proud of doing. You do not need to follow the numbering of this guidance. See response above, where we recommend using the IMDRF numbering scheme instead, which is internationally harmonized.
Q: Where is information for risk assessments and management best included?
Depending on the type of submission you have selected (e.g. 510(k), De Novo, then Traditional, Special, Abbreviated), certain sections will become visible and active and some of them will be disabled. However, the bookmark pane will show all the sections in the entire eSTAR. Hence, please fill the sections that are visible to you since they are based on your selections. For example, if you have selected a Traditional 510k, the “Benefits, Risks and Mitigation Measures” section is not applicable. Hence it is not displayed and clicking on it leads a user back to the first page of the eSTAR. However, if you select “Special” as the 510k you are submitting, the “Benefits, Risks and Mitigation Measures” section does become applicable, and is displayed if you click on the bookmark. Also note that if you select De Novo, the section does become active as well. Another point to note is that for a Traditional 510(k) submission, if you do want to submit a Risks, Mitigations, Benefits report, you can attach it in the Performance Testing --> Bench Testing section of the eSTAR and identify it as “Other Non-Clinical Evidence”.
Q: Under Biocompatibility any idea when there will be endpoint questions for hemocompatibility or coagulation?
Hemocompatibility endpoint information, including Hemolysis, Complement Activation, and Thrombogenicity information, will appear depending on the tissue contact type and duration you choose. If you choose Implant Device >30 days, you will see this tab appear for example. The tabs that show are based on our Biocompatibility guidance.
Q: Guidance and Special Controls Adherence Section: According to the 510(k) electronic submission guidance document, identification of any applicable device-specific guidance documents or special controls should be included, which was done in the past; however, eSTAR requires each specific special control regulation or guidance recommendation to be listed. Can you please specify to what level of detail needs to be included within this section? The text box contains a character limitation, so it is unclear how specific information needs to be.
There are four textboxes like this in eSTAR. The specifications, characteristics, etc., of the device should not be provided in these textboxes. Instead, these textboxes should be used to refer to the attachments where these controls or recommendations are found, per each specific recommendation. These textboxes do expand as text is typed in, if needed. Keep in mind that the special controls, device-specific guidance citations would be divided among these four textboxes.
Q: Why are Advertisements included within the Labeling section, when they should be considered promotional materials? Could reference to advertisements be removed from this section?
Advertisements and promotional materials are included under the Other Labeling, and within the sub-attachment type "Other Labeling and promotional material" since this is consistent with Internationally harmonized document N9 and N13. As the help text states, whether you actually need to attach it at all (it is an optional attachment) is dependent on the jurisdiction. For FDA, you would need to reach out regarding your device and current policy at opeqsubmissionsupport@fda.hhs.gov.
Q: I need to know how eSTAR would promote efficiency.
There are many efficiency gains with eSTAR, this is not an exhaustive list. The eSTAR website also includes some of this information.
-eSTAR complements the reviewer's internal smart review template used to review the submission (i.e. the questions correlate). Therefore the reviewer is getting what they are expecting
-eSTAR provides a standardized format so that the reviewer (and applicant) always know where to find certain information
-eSTAR auto-updates many aspects of the submission, most notably it ensures the content is present negating the need for an RTA review by the reviewer, and therefore RTA holds
-eSTAR autocompletes information that you enter, ensuring you never need to enter the same information twice
-eSTAR includes built-in databases that ensure you see the information pertinent to your device (e.g. device-specific guidances), classification information automation, and the standards database, for auto-filling standards info accurately
-eSTAR has many forms built in, so that you don’t need to attach or upload them (e.g. T&A statement, Form 3514, 510(k) Summary, Declaration of Conformity, Form 3881)
-eSTAR guides the applicant through what is needed throughout, ensuring we collect everything that needs to be provided, and you know how to do it correctly
-eSTAR is collecting submission data in a structured format, which will help automate many aspects of our processing. This will provide applicants and reviewers many benefits (e.g. automating the submission log-in process, therefore permitting reviewers to receive a submission within minutes of when the submission is uploaded in the CDRH Portal)
-eSTAR is intended to be used as a resource also, since it consolidates all the information needed (e.g., regulation links, guidance links, submission process information) to prepare a submission

Rimsys ranks number 156 on the 2023 Inc. 5000
Rimsys is happy to announce today that it has been ranked number 156 on the 2023 Inc. 5000 list of fastest-growing private companies in America. Companies on the 2023 Inc. 5000 are ranked by their percentage of revenue growth over a three-year period. With exponential growth over the past three years, Rimsys joins a prestigious list of organizations across the country.
Rimsys, a cloud-based software for end-to-end regulatory process digitization and automation, is designed by and for medtech regulatory affairs professionals who experienced the painful challenges and compliance risks that result from not having a centralized, collaborative hub for regulatory information.
"Being included on this year's Inc. 5000 list of fastest-growing private companies is a tremendous milestone for Rimsys," said James Gianoutsos, Founder and CEO. "I created Rimsys to help medical device manufacturers overcome the challenges I experienced in the industry, and our regulatory information management (RIM) platform has increasingly gained the trust of leading medical device manufacturers globally over the past few years. I'm excited to continue our mission of improving global health by accelerating delivery and increasing availability of life-changing medical technologies with the support of our team members, customers, and partners."
The top 500 companies, including Rimsys, are featured in the September issue of Inc. Magazine, which will be available beginning August 23. To see the complete results of the Inc. 5000, view company profiles, and access an interactive database of honorees, visit www.inc.com/inc5000.
About Rimsys
Rimsys is improving global health by accelerating delivery and increasing availability of life-changing medical technologies. Rimsys Regulatory Information Management (RIM) software digitizes and automates regulatory activities, helping medtech regulatory affairs teams to plan more effectively, execute more quickly, and confidently ensure global regulatory compliance. Unlike complex, color-coded spreadsheets, or expensive external consultants, Rimsys centralizes all regulatory information, automates submission processes, and provides detailed visibility into product registrations, expirations, relevant standards, and global regulations. Traditional approaches to regulatory affairs can’t keep pace with the growing complexity of the global landscape, and overburdened teams face increasing compliance risks. Rimsys is designed around medtech regulations and workflows and supports a full breadth of regulatory activities including registrations, submissions, UDI, essential principles, standards management, and regulatory intelligence in a single, integrated platform. Leading global medtech companies including Johnson & Johnson, Siemens Healthineers, and Omron rely on Rimsys to better manage regulatory projects and resources, get new products to market more quickly, and reduce revenue risk of non-compliance, product recalls, and unexpected expirations. For more information, visit www.rimsys.io.
About Inc.
Inc. Business Media is the leading multimedia brand for entrepreneurs. Through its journalism, Inc. aims to inform, educate, and elevate the profile of our community: the risk-takers, the innovators, and the ultra-driven go-getters who are creating our future. Inc.’s award-winning work reaches more than 50 million people across a variety of channels, including events, print, digital, video, podcasts, newsletters, and social media. Its proprietary Inc. 5000 list, produced every year since 1982, analyzes company data to rank the fastest-growing privately held businesses in the United States. The recognition that comes with inclusion on this and other prestigious Inc. lists, such as Female Founders and Power Partners, gives the founders of top businesses the opportunity to engage with an exclusive community of their peers, and credibility that helps them drive sales and recruit talent. For more information, visit www.inc.com.

An overview of the Medical Device Discovery Appraisal Program (MDDAP)
Regulatory affairs within the medtech industry is uniquely challenging. Teams are faced with external changes in the form of new regulations (such as the MDR/IVDR in the European Union), growing information needs (including expanding unique device identification requirements around the world), newer audit requirements (MDSAP), and post-market surveillance requirements.
While there are emerging technologies that can help teams better address these issues, building more effective and efficient regulatory teams requires more than better tools. The traditional, largely manual approach to regulatory procedures and processes must also evolve, including the measurement of regulatory maturity. The Medical Device Discovery Appraisal Program (MDDAP) was born out of the recognition that regulatory compliance does not, by itself, guarantee the highest quality devices and was designed to enable device makers to measure their capability to produce high-quality devices.
This blog post provides a high-level overview of the Medical Device Discovery Appraisal Program (MDDAP) to help organizations assess the benefits of participating.
What is the Medical Device Discovery Appraisal Program (MDDAP)?
MDDAP provides the model for the Case for Quality Voluntary Improvement Program, a collaborative program between the FDA Center for Devices and Radiological Health (CDRH), the Medical Device Innovation Consortium (MDIC), Information Systems Audit and Control Association (ISACA), and the medical device industry. The Case for Quality program was initially started by the FDA in 2011 to identify manufacturers that consistently produce high-quality devices along with the practices that have the greatest impact on quality.
A timeline of MDDAP:
- 2011 – FDA launches Case for Quality
- 2018 – FDA and the Medical Device Innovation Consortium (MDIC) implement a voluntary pilot program for medical device manufacturing sites using the key business process and best practices detailed in the ISACA Capability Maturity Model Integration (CMMI) system
- 2022 – On May 5, 2022 the FDA issued the draft guidance, Fostering Medical Device Improvement: FDA Activities and Engagement with the Voluntary Improvement Program.
The draft guidance issued in May, 2022 outlines the transition from the pilot program to a permanent program, titled the Case for Quality Voluntary Improvement Program. The Voluntary Improvement Program (VIP) is facilitated through the Medical Device Innovation Consortium (MDIC) which will utilize the Capability Maturity Model Integration (CMMI) to evaluate the capability and performance of the participating medical device manufacturers. Originally developed to optimize an organization’s software development process, CMMI is now used in many industries to provide a methodology and assessment tools for continuous improvement.
The MDDAP program is voluntary and leverages the CMMI model to provide medical device manufacturers, who are already in compliance with FDA quality and regulatory requirements, with insight into how they perform against selected best practices. The program is designed to help already-compliant organizations identify gaps in existing processes and increase the ability to react to change and avoid previously unforeseen quality and process issues.
Requirements for MDDAP participation
Any facility which “designs, manufacturers, fabricates, assembles, or processes a finished device” in the United States is eligible for participation, with the following additional requirements:
- The organization must have a prior compliance history or compliance profile through an FDA inspection or MDSAP audit.
- The participating site must be registered with the FDA.
- The device must be listed with the FDA.
- The organization cannot currently be under an Official Action Indicated (OAI) or subject to judicial action.
Participants in the program can include those with Class I, II, or III devices and, in addition to being device manufacturers, may also perform contract sterilization, relabeling, remanufacturing, repacking, or specification development.
Visit the ISACA MDDAP site to see if your organization qualifies.
Advantages for manufacturers participating in MDDAP
The MDDAP program provides operational advantages by streamlining multiple regulatory processes with the FDA, as well as longer-term benefits to product quality and organizational processes. The program is built on the CMMI framework which identifies processes most effective in decreasing risk and improving product quality.
MDDAP participants who have completed appraisals through the program will have the following advantages when working with the FDA:
1. Reduced inspection activities - A risk-based approach to FDA inspections allows manufacturers to bypass routine FDA surveillance audits and PMA preapproval inspections for participating sites. The participating site will have demonstrated compliance with the quality system regulations verified by FDA through inspection (a prerequisite for joining the program). MDDAP appraisals and quarterly checkpoints evaluate the participating organization against a proven set of best practices and provide guidance on continuous improvement projects.
Participants should understand that the FDA will still perform “for cause” or directed inspections as required based on safety signals or as a follow-up to a safety event. Also, note that organizations participating in the MDSAP program will still need MDSAP audits.
2. Streamlined change notices - Participants in the MDDAP program have access to modified submission formats for:
- PMA and HDE 30-day change notices. Resources permitting, the FDA intends to review changes within the reduced timeframe of 14 calendar days.
- PMA and HDE manufacturing site change supplements.
- PMA or HDE manufacturing modules.
3. A more collaborative process with the FDA - The FDA’s Case for Quality program was designed to provide a more collaborative approach to medical device compliance and quality. This differs from the traditional oversight models by going beyond regulatory compliance and focusing on continuous improvement projects that optimize device quality.
Perhaps the biggest benefit of participating in the MDDAP program, however, is that the program provides a methodology for evaluating an organization's “maturity level” and implementing continuous improvements toward the goal of optimizing quality. Participating organizations are committing to a proven continuous improvement program. This results in the ability to consistently ensure a high-level of quality that can yield significant competitive and market advantages.
Are you looking for more tips to help your regulatory team achieve competitive advantages? Read our ebook, Regulatory strategy as a competitive advantage.





