

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

The beginner's guide to the FDA 510(k)
This article is an excerpt from The beginner's guide to the 510(k) ebook.
Table of Contents
- Introduction
- 510(k) basics
- Contents of a Traditional 510(k)
- 510(k) submission and timelines
- Other 510(k) forms
Congratulations! You have successfully developed a new medical device. Now you need to take it to market. In the United States, this often means submitting a 510(k). A 510(k) is a structured package of information about your device and its performance and safety that you submit to the Food and Drug Administration (FDA) for “clearance” before you can sell your device in the U.S. In order to receive clearance from the FDA, your 510(k) will need to demonstrate that your medical device is substantially equivalent to another legally marketed device (called a predicate device). The substantial equivalence approval process is a simple equation that looks something like this:

The 510(k) is generally the most efficient route to market clearance in the U.S. because you show your device is safe and effective based on this substantial equivalence standard, instead of needing to present more extensive clinical trial data.
There are three types of 510(k): Traditional, Abbreviated, and Special. This eBook will begin with a general overview of the 510(k) process, including its purpose and benefits. Next, we will explore the Traditional 510(k) and the sections and components required in depth. Finally, we will look at the Special and Abbreviated 510(k).
FDA: background and device oversight
Before we explain what a 510(k) is let’s first talk generally about the FDA and device oversight. The FDA is the U.S. governmental agency responsible for overseeing medical devices, drugs, food, and tobacco products. When it comes to medical devices, the FDA’s mission is to “protect the public health by ensuring the safety, efficacy, and security of…medical devices.” At the same time, the FDA also has an interest in “advancing public health by helping to speed innovations.” In other words, the FDA’s goal is to make sure devices are safe and effective for public use, while also ensuring that devices have a quick and efficient path to market.
In order to achieve this balance of safety and efficiency, the FDA has three different levels of oversight depending on the risk level of the device: (1) exempt from premarket submission, (2) Premarket Notification, also known as 510(k), and (3) Premarket Approval (PMA).
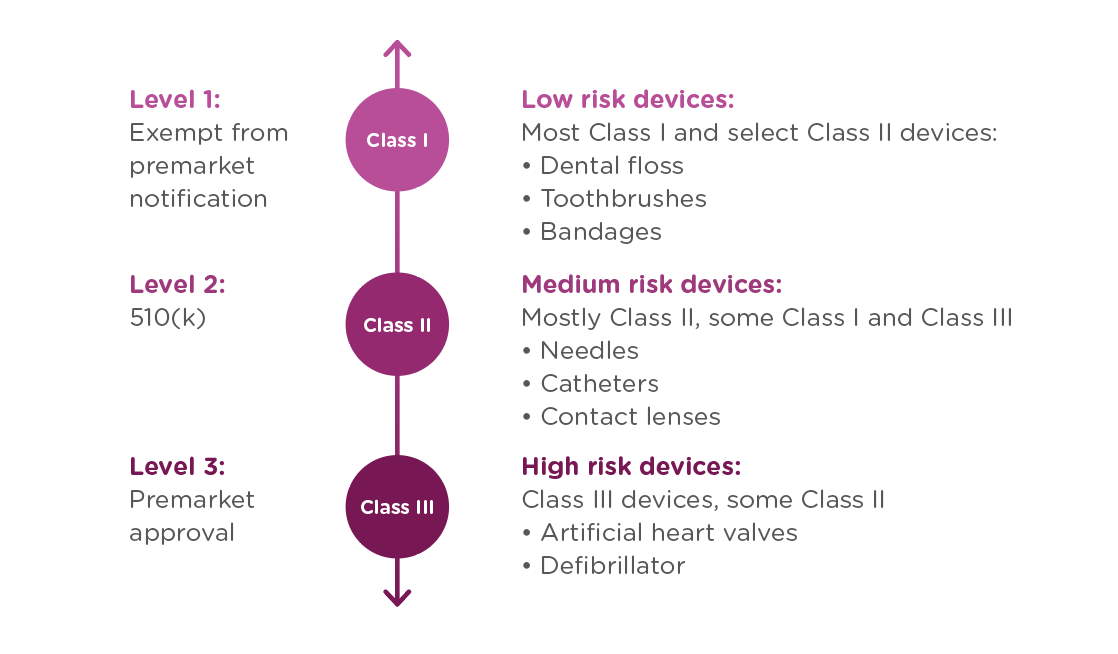
When is a 510(k) required?
A 510(k) is required for medium risk devices that have a predicate on the market which can be used to demonstrate the safety and effectiveness of the new device. Meanwhile, a PMA is required for high-risk or novel devices which require a higher level of scrutiny to be confirmed safe and effective.
A 510(k) is not only required for new devices, but also for devices that have been modified in a way that could impact safety or effectiveness. This could include changes to the:
- Design
- Components
- Materials
- Chemical composition
- Energy source
- Manufacturing process
- Intended use
You must submit your 510(k) at least 90 days before marketing the device.
What Exactly is Substantial Equivalence?
Now that we know what a 510(k) is, let’s talk about the substantial equivalence standard. You’ll recall from the introduction that your 510(k) must show that the new (or modified) device is substantially equivalent to at least one other legally marketed device, called a predicate device. Substantial equivalence looks at the intended use and the technological characteristics of the two devices.
More specifically, you must show:
- that the new device has the same intended use as the predicate, and
- the differences between the two devices do not raise questions about the safety and effectiveness of the new device.
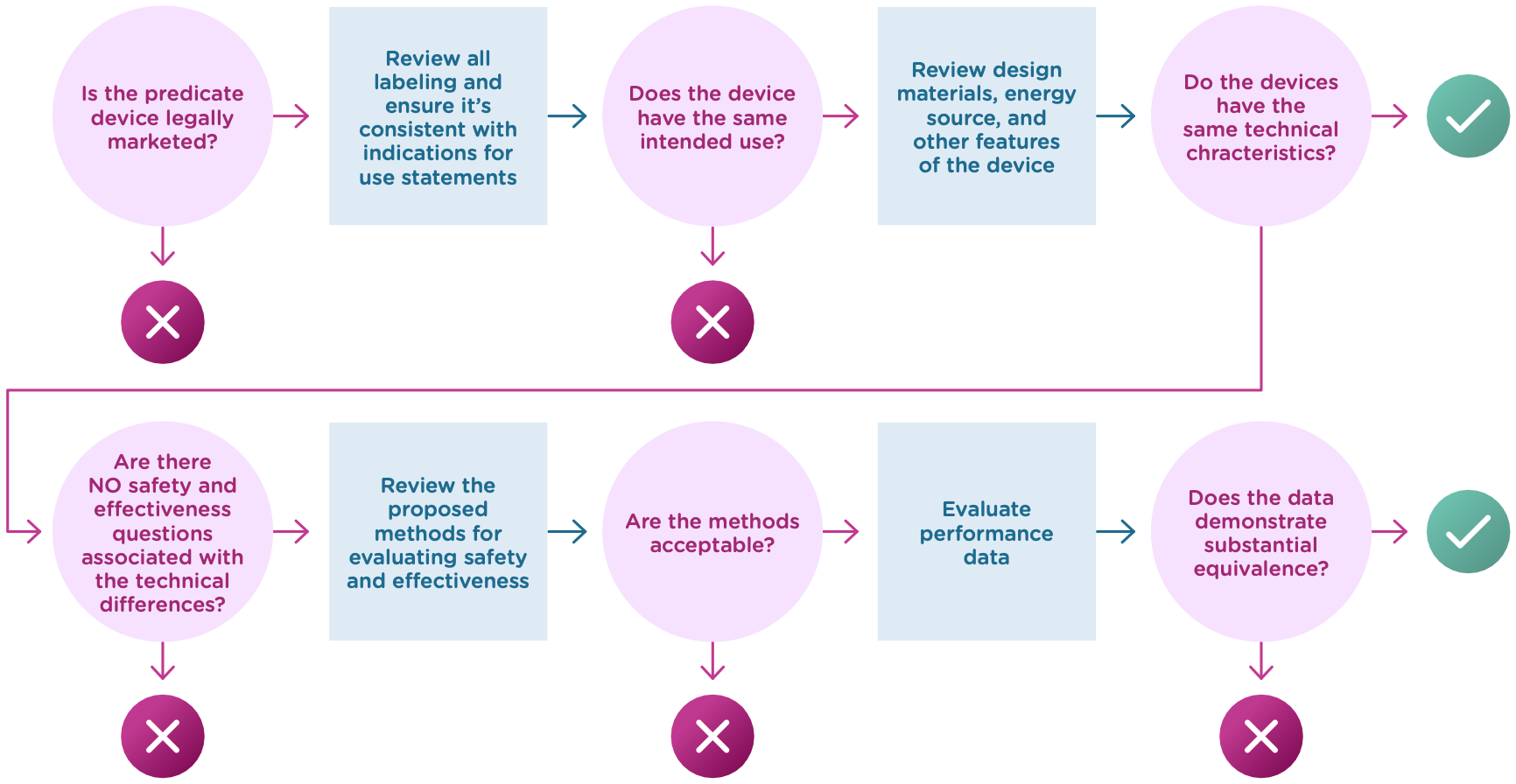
Now let’s take a closer look at intended use and technological characteristics.
Intended use
Intended use means the general purpose or function of the device. The FDA will look at your proposed labelling and your Indications of Use section of the 510(k) to determine the intended use of your device (this is covered in Chapter 2). Intended use includes:

Technological characteristics
Once the FDA has determined that a predicate device exists and that the new device and the predicate device have the same intended use, it will move on to compare the technological characteristics. Technological characteristics include:
- Materials
- Design
- Energy source
- Other device features
The two devices do not have to be identical, and in fact they almost never are. The key here is to demonstrate that any differences do not have a significant impact on safety or effectiveness. Here’s what to cover when you compare your device’s technological characteristics with that of the predicate device:
Overall description of the device design
- Engineering drawings or diagrams to explain the device and component parts.
- List of component parts and explanation of how each component contributes to the overall use and function of the device.
- Physical specifications: dimensions, weight, temperature, tolerances, etc.
Materials
- Detailed chemical formulation used in all materials of constructions (especially those that come into contact with a patient).
- Any additives, coatings, paint, or surface modifications.
- How materials have been processed and what state they’re in.
Energy Sources
- Use of batteries, electricity, etc.
Other technological features
- Software/hardware
- Features
- Density
- Porosity
- Degradation characteristics
- Nature of reagents
- Principle of the assay method
In deciding whether the differences in technological characteristics impact safety or effectiveness, the FDA will typically rely on descriptive information about the technological characteristics as well as non-clinical and clinical performance data.
Let’s look at an example: A manufacturer submits a 510(k) for a new type of contact lens. Both the new device and the predicate device are indicated for daily wear for the treatment of astigmatism. The predicate device is only available in a clear lens, but the new device comes in a line of colors, including purple tinted lenses.

Who is responsible for submitting a 510(k)?
The following four types of organizations may be responsible for submitting a 510(k):
Manufacturers
- End-of-line device manufacturers who will be placing a device on the U.S. market.
- Note: Does not apply to component part manufacturers unless components will be marketed independently.
Specification developers
- Companies that develop the specifications for a finished device which has been manufactured elsewhere
Repackers or relabelers
- Required to submit a 510(k) if they significantly alter the labeling or condition of the device, including modification of manuals, changing the intended use, deleting or adding warnings, contraindications, sterilization status.
- Note: This is rare. The manufacturer, not the repackager or labeler, is typically responsible for the 510(k) submission.
Importers
- Importers that introduce a new device to the U.S. market may need to submit a 510(k), if it hasn’t already been submitted by the manufacturer.
Now that we’ve covered the basics, let’s explore what actually goes into your 510(k).
A Traditional 510(k) should contain all the following components in the list below. In some cases, a particular section may not apply to your device. When that happens, it’s a good idea to include the section anyway and just state “This section does not apply” or “N/A” under that heading.
To continue reading this eBook including a detailed walk-through of all the Traditional 510(k) components, submission requirements and timelines, and an overview of the other 510(k) forms including the Abbreviated 510(k) and the Special 510(k), please register to download the full version

The ultimate guide to the China UDI system and database
This article is an excerpt from The ultimate guide to the China NMPA UDI system and database ebook.
Table of Contents
- Overview
- UDI basics and benefits
- UDI format requirements and issuing entities
- UDI database and submission requirements
- Implementation of UDI and the UDI database in China
The current Chinese medical device regulatory regime kicked-off in 2014 with the Regulation on Supervision and Administration of Medical Devices. This core set of registration requirements, modeled after the United States and European Union systems, established a set of device classifications (class I, II, and III) based on risk and procedures for obtaining market clearance for each type of device.
Medical devices in China are regulated by the National Medical Products Administration (NMPA). Class I devices, such as clinical laboratory equipment or non-invasive skin dressings, require only notification to the NMPA for marketing authorization, and that authorization does not expire. Class II and III devices such as implantable devices or devices with a measuring function require full registration and a formal review before market clearance can be obtained.
These initial regulations have been expanded since their introduction, adding accelerated pathways to market for certain products in certain regions, easing acceptance of clinical data from overseas, and more specific roles and responsibilities for local agents of international manufacturers. In addition, in 2019, the regulations added a provision that medical devices carry a unique device identification (UDI). China’s UDI requirements are similar to those in the US and European Union. They establish specific device ID and labeling requirements, as well as a central, state-administered database of devices.
This eBook walks through the basics of medical device UDIs, the specifics of China’s implementation, and how MedTech companies who market their devices in China can prepare for the full rollout of these regulations in the coming years.
A UDI is a unique alphanumeric code that is designed to identify medical devices sold in a particular country/region from manufacturing, through distribution, to use by a patient. Like other aspects of the medical device regulatory regime, the UDI system in China follows the approach taken by the United States FDA and European Commission, and is based on the guidance from the International Medical Device Regulators Forum (IMDRF). Generally, UDI systems are designed to improve patient safety and optimize care by:
- Increasing the traceability of medical devices, including field safety corrective actions
- Providing an unambiguous identification method for medical devices throughout distribution and use
- Making adverse event reports more accessible
- Reducing medical errors by providing detailed information related to the device
- Simplifying medical device documentation and making it more consistent
There are three components to the UDI system in China:
- UDI code: The actual UDI code can be assigned by one of three (3) issuing agencies and contains information about the product, it’s expiration date, and the manufacturing batch/lot it’s associated with.
- UDI labeling: Put simply, medical devices must carry the UDI code on them. The regulations stipulate how devices and their packaging must be labeled for compliance.
- UDI database: In addition to labeling, all device UDIs must be submitted to a central database that is administered by the NMPA.
The following sections explore each of these components in more detail.
The UDI code
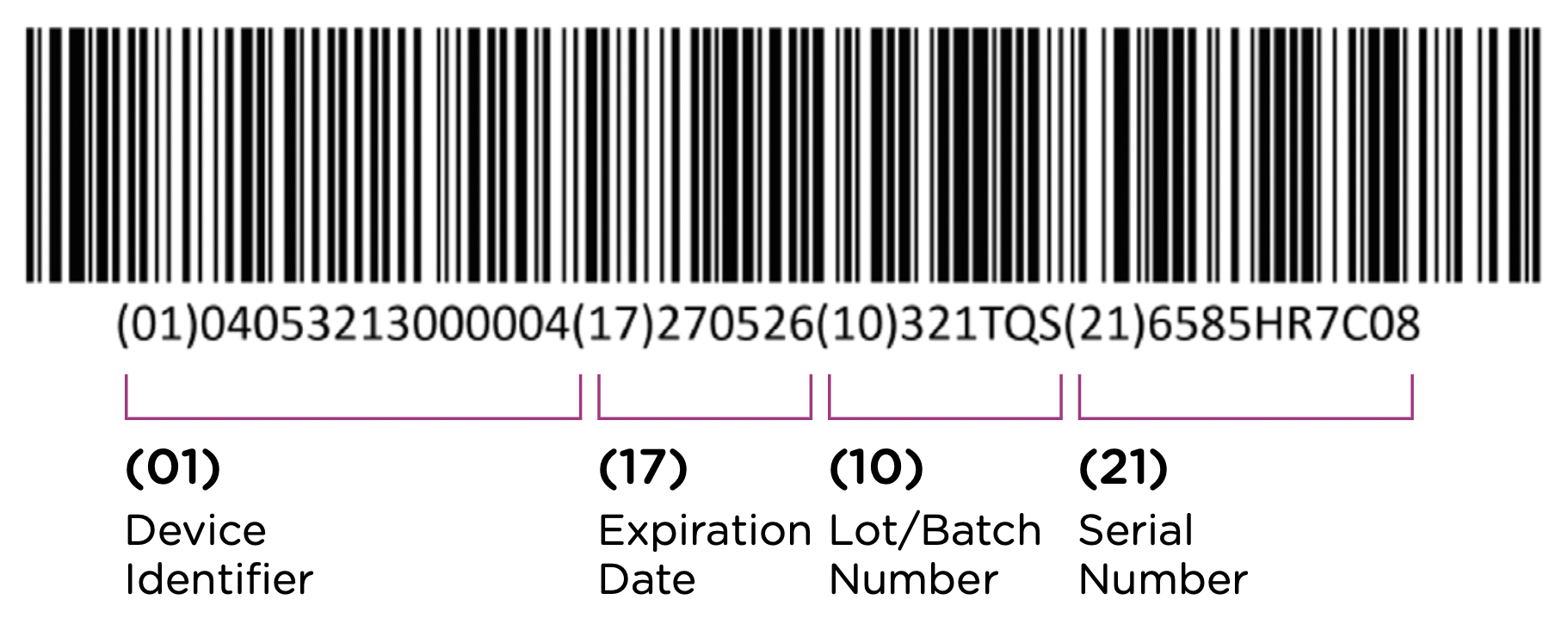
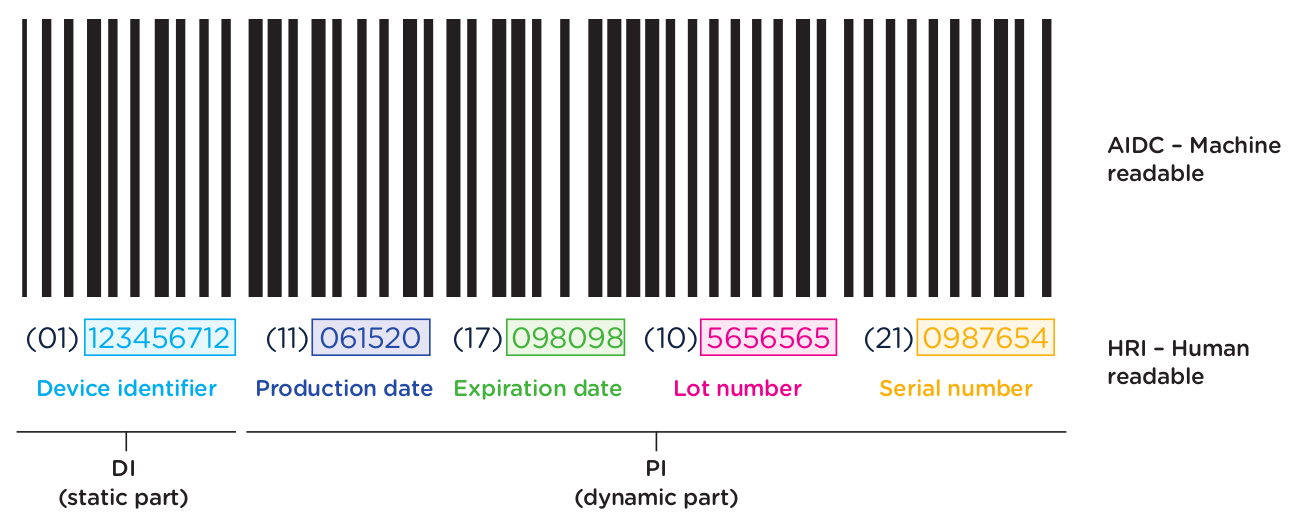
The first element of the UDI system is the code itself. The UDI code is the alphanumeric identifier that is associated with a specific medical device. UDI codes have two (2) elements to them, the UDI device identifier (UDI-DI) or static portion, and the UDI production identifier (UDI-PI) or dynamic portion. You can see the two components in the UDI diagram below:

The UDI-DI contains information about the issuing entity—the organization that is authorized to assign UDI codes. In China, this can be one of three entities: GS1, an international barcode and electronic data interchange standards organization, and two domestic organizations: the Zhongguancun Industry & Information Research Institute (ZIIOT), and AliHealth. Additional details about the issuing agencies are covered in Chapter 2. In addition, the UDI-DI contains information about the manufacturer and the specific model or version of the device.
The UDI-PI contains information about the manufacturing and production of the device. This typically includes information about the lot or batch number in which the device was manufactured, the manufacturing date and expiration date for the device (if applicable), and the specific serial number for the device. Here you can see all of the components marked up using the same UDI example:
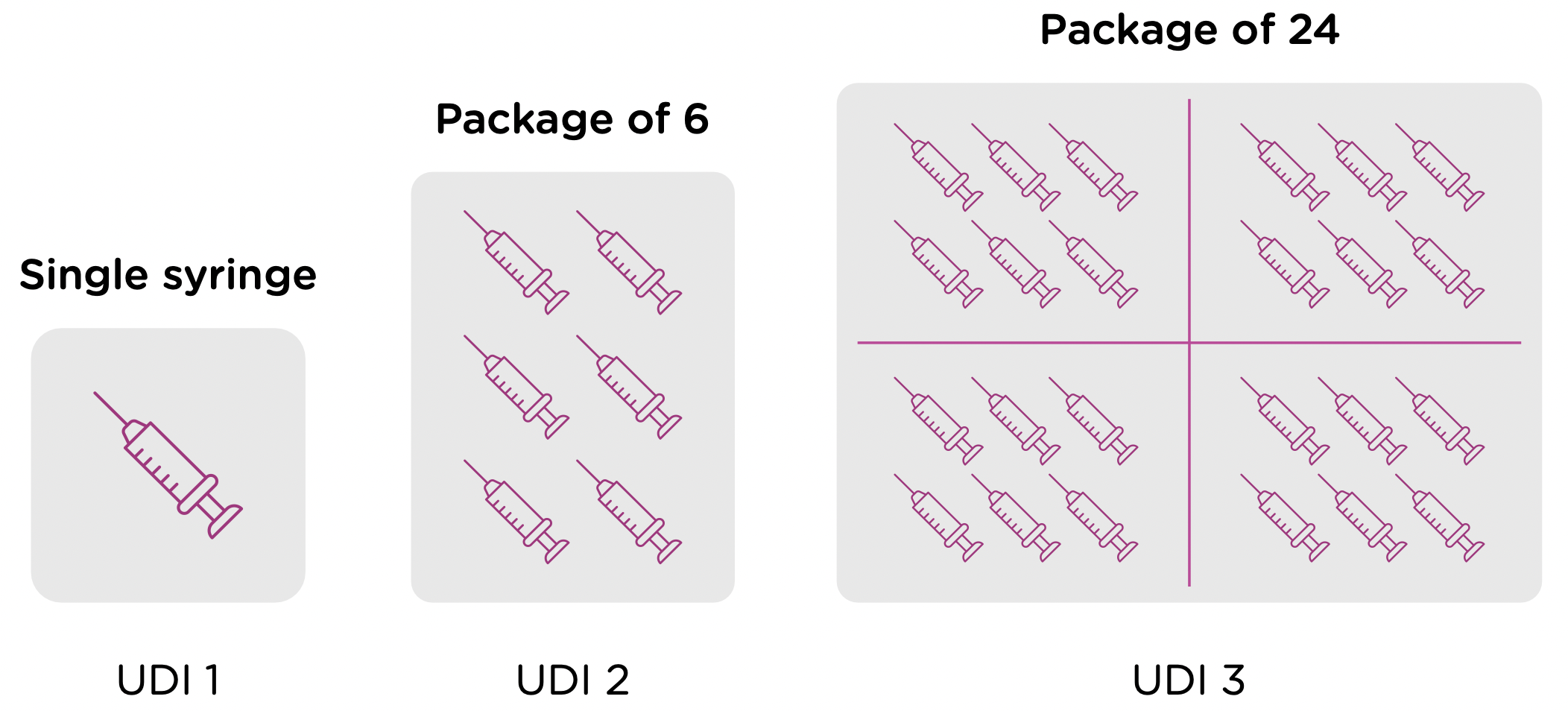
Note that each packaging permutation and level for a given device will need to be assigned its own UDI. So for example, let’s say that a company manufactures 5ml enteral (oral) syringes in two packaging options: 1 – packaged individually and 2 – packaged in a box of 5. Each packaging option would need its own UDI, despite the fact that the underlying product is the same.
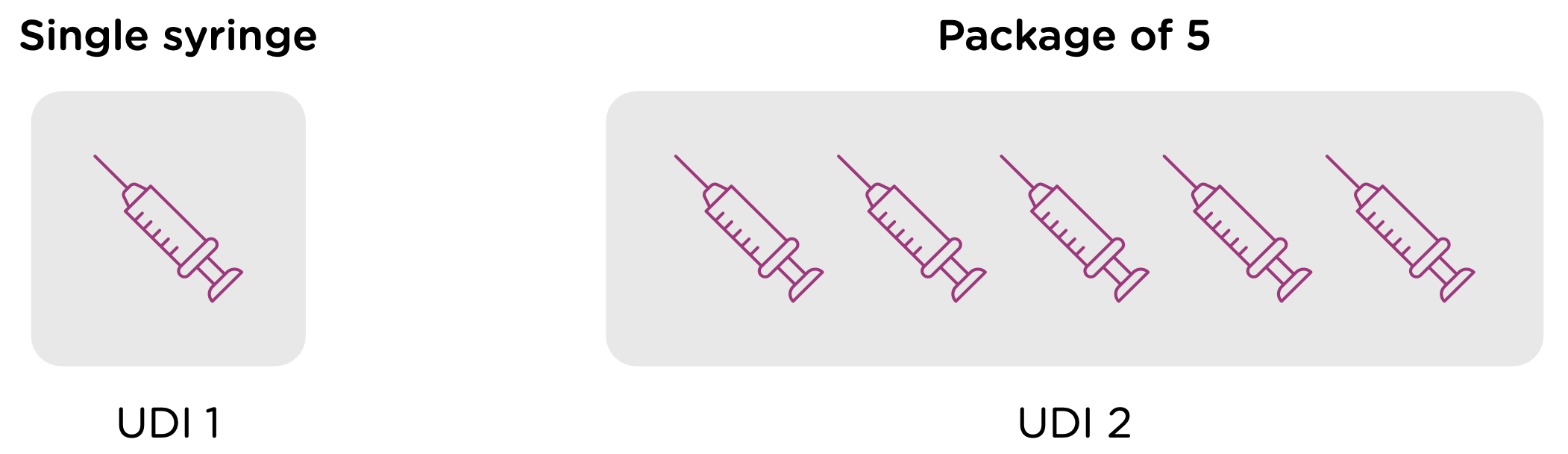
Now looking at packaging levels, let’s assume that the manufacturer packages the single syringe offering into boxes of 6, and again into larger containers of 24. Each of those packaging options needs its own UDI as well.
Labeling
In addition to obtaining UDI code for each device as outlined in the previous section, medical device manufacturers are required to ensure that devices are appropriately labeled with the assigned UDI. This label is called the UDI Carrier. The UDI is represented in two forms on the UDI Carrier: a machine-readable form and a human-readable form.
The machine-readable form or automatic identification data capture (AIDC) is a barcode or some other technology that can be used to automatically capture UDI information. The NMPA regulations support 3 types of machine-readable formats: 1-dimensional barcode, 2-dimensional barcode, and radio-frequency identification (RFID).

The regulations note that “use of advanced automatic identification and data collection technologies is encouraged”—prompting manufacturers to use more modern 2D and RFID machine-readable carriers where possible. Note, however, that if a device uses RFID, the UDI Carrier must also include the UDI in barcode format.
The human-readable form or human-readable interpretation (HRI) is the numeric or alphanumeric code for the UDI that can be read and manually entered into systems.

The UDI Carrier should be included on the device and on all levels of packaging. The UDI Carrier must be clear and readable during the operation and use of devices. If there isn’t room on the device for both the human and machine-readable forms of the UDI, then manufacturers should prioritize the machine-readable form.
UDI database
The third component of the NMPA UDI system is the UDI database. This is a centralized database of UDI and product information, administered by the NMPA. Manufacturers are required to submit UDI information into the database within 60 days after a product is approved (for sale in China) and before it is commercialized. The database contains a more detailed product record than what is included in the UDI itself, and it is the responsibility of the manufacturer (and/or their in-country representative) to submit the information correctly, and ensure that it’s kept up to date.
Chapter 3 of this eBook goes into detail about the specific fields and data requirements for UDI database submissions.
To continue reading this eBook including information about UDI format requirements and issuing entities, implementation timelines, and affected device types, please register to download the full version.

The ultimate guide to the EU MDR/IVDR unique device identifier (UDI) System
This article is an excerpt from The ultimate guide to the EU MDR/IVDR UDI ebook.
Table of contents
- Overview
- UDI basics and benefits
- UDI format requirements and issuing entities
- UDI rules for specific device types
- Implementation of UDI and UDAMED in the European Union
- US vs EU UDI comparison
The EU Medical Device Regulation (2017/745) (“MDR”) and EU In Vitro Diagnosis Regulation (2017/746) (“IVDR”) introduce two new systems for information exchange: UDI (Unique Device Identifier) for device identification and EUDAMED (European Databank on Medical Devices) to centralize and disseminate information. UDI is a specific code assigned to all devices and higher levels of packaging. This will allow for devices being sold in the European market to be identified and traced through a globally harmonized approach. EUDAMED is the IT system developed by the European Commission to replace the EUDAMED2 database previously in place under the Medical Device Directives (MDD). EUDAMED is a multi-functional system that will be used to coordinate device registration, provide information about devices to industry professionals and the public, and highlight necessary safety details.
The EU MDR and IVDR UDI system is based upon the guidance of the International Medical Device Regulators Forum (IMDRF). It’s a globally harmonized system that’s designed to increase patient safety and optimize care.
UDI system goals
Increase patient safety
- Improve tracing of devices
- Reduce the presence of counterfeit devices
Ensure access to accurate information
- Unambiguous identification of devices throughout distribution and use
Improve post-market surveillance
- Improve accessibility of adverse event reports
Enhance supply chain Management
- Streamline supply chain process and inventory management
- Simplify medical device documentation processes
The UDI system has four key elements
Element 1: Assignment of UDI (UDI Components)
The first element of the UDI system is the assignment of a UDI. The UDI is a code of alphanumeric characters that acts as the access key to information about a specific medical device on the market. The EU MDR and EU IVDR requires that a UDI be assigned to all medical devices except for custom-made or investigational devices. There are three components of a UDI:
- Basic UDI-DI
- UDI (consisting of UDI-DI and UDI-PI)
- Packaging UDI (Note: This is not an official term used in the EU MDR and IVDR, but we’re using it to help explain the concept. The Packing UDI is part of the UDI itself.)
1. Basic UDI-DI
The Basic UDI-DI identifies the device group that a particular device fits into. A device group is a group of products that all share the same intended purpose, risk class, essential design, and manufacturing characteristics. A device group is generally classified by medical device manufacturers as a “Product Family” or “Product Category,” depending on the internal nomenclature used within the company. The Basic UDI-DI functions as a parent or higher-level descriptor of a device.
NOTE: There can only be one Basic UDI-DI per UDI-DI.
The Basic UDI-DI is not printed on the product itself or on the packaging of a product, but rather it must be included in the following documents and applications:
- Certificates (Including Certificate of Free Sale)
- EU Declarations of Conformity
- Techical Documentation
- Summary of Safety and Clinical Performance
2. UDI (UDI-DI and UDI-PI)
The second component is the UDI itself, which consists of two parts:
Device Identifier (DI)
Production Identifier (PI)
The UDI-DI (Device Identifier DI, also referred to as “static”) identifies specific, detailed information about a particular device. If any of the below details should change, the device will need a new UDI-DI.
- Name or trade name of the device
- Device version or model
- If labelled as a single use device
- Packaged as sterile
- Maximum number of uses
- Need for sterilization before use
- Quantity of devices provided in a package
- Critical warnings or contra-indication
- CMR/endocrine disruptors
NOTE: There can be several UDI-DIs for one Basic UDI-DI.
Meanwhile, the UDI-PI (Production Identifier PI, also referred to as "dynamic") contains manufacturing information (including serial number, lot/batch number, software identification, and manufacturing or expiry date or both types of dates.)
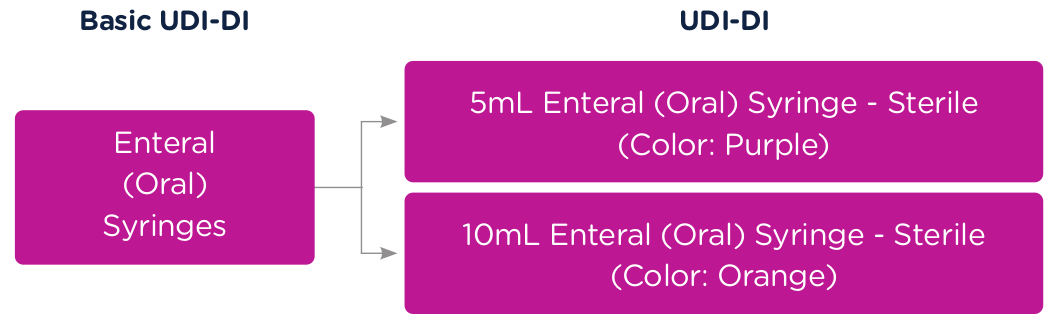
To better illustrate this concept of Basic UDI-DI and UDI (UDI-DI and UDI-PI), let’s use a syringe as an example. The Basic UDI-DI would identify the category of a syringe, for example, "Enteral (Oral) Syringe."
A 5ml Enteral (Oral) Syringe – Sterile (Color: Purple) would get a unique UDI-DI and a 10m Enteral (Oral) Syringe – Sterile (Color: Orange) would get a unique UDI-DI. Both products would be associated to the same Basic UDI-DI. In this case, the "Enteral (Oral) Syringe," which defines the category.
Each time that 5ml Enteral (Oral) Syringe – Sterile (Color: Purple) is manufactured at the same revision, it will get a new UDI-PI per lot. See the graphic below.

Each product is identical and therefore has the same UDI-DI. However, the UDI-PI changes to reflect the manufacturing date, lot number, expiry date, and serial number, as applicable.
The UDI will contain all device-specific information and have the same functions as the comparable database (GUDID) of the United States FDA. The main difference (in EUDAMED) is that the UDI data is divided into components of Basic UDI-DI, UDI, and Packaging UDI.
3. Packaging UDI
The third component of UDI is the Packaging UDI. (Note: This is not an official term used in the EU MDR and IVDR, but we’re using it to help explain the concept.)
Each level of packaging, except shipping containers, must receive its own unique UDI. Packaging UDI refers to the unique UDI assigned to higher levels of packaging instead of the device itself.
In the event of significant space constraints on the unit of use packaging, the UDI Carrier may be placed on the next higher packaging level.
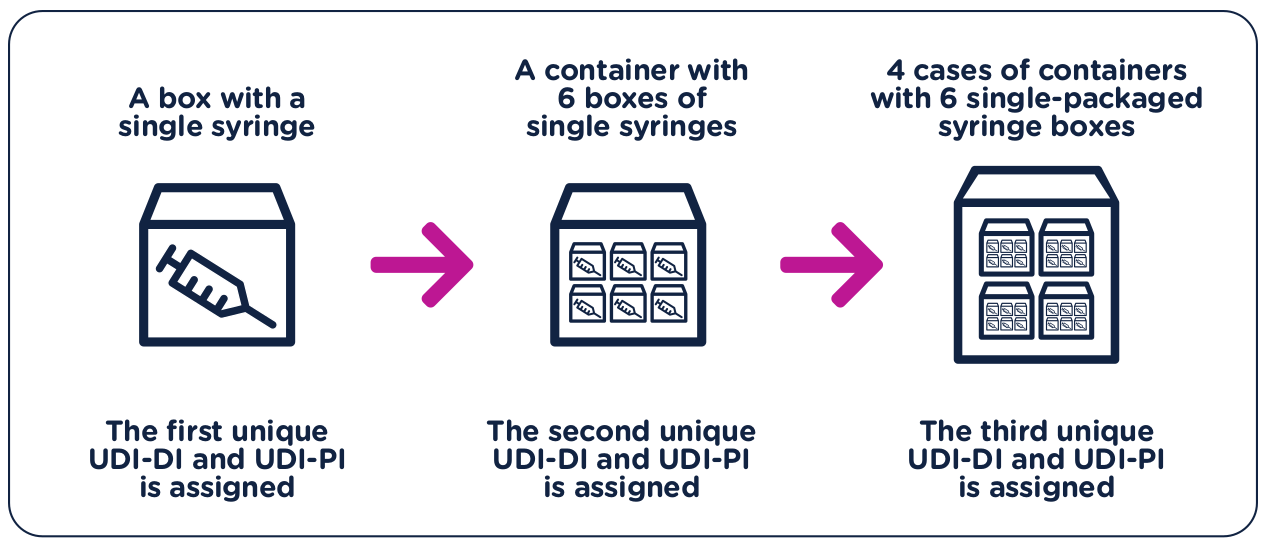
Returning to our earlier example of syringes, if a manufacturer first packages a single sellable syringe into an individual box, this package would receive its own UDI-DI and UDI-PI.
If then the manufacturer packages those individual boxes into containers of six (6), those containers would receive their own UDI-DI and UDI-PI.
And finally, if the manufacturer packages those six (6) containers into cases of four (4), those cases would receive their own UDI-DI and UDI-PI.
Each of those levels of packaging must be assigned its own UDI-DI and UDI-PI. The initial syringe did not change, but the way it is packaged did, therefore, requiring its own UDI-DI and UDI-PI.
Element 2: Placing UDI on the device and/or packaging
The second element to the UDI system is the placing of the UDI on the device or on its packaging through what is referred to as a “UDI Carrier.” The UDI Carrier is the part of the label that contains the UDI information that is applied directly to the device or included on the device packaging. The UDI Carrier should have both a machine-readable portion (AIDC) and a human-readable portion (HRI). (Specific details about each element of the UDI will be covered in Chapter 2.)
- Machine-readable form – AIDC – (Automatic Identification and Data Capture) is a barcode or other machine-readable technology that can be accessed automatically by scanning the UDI information.
- Human-readable form – HRI – (Human Readable Interpretation) is the numeric or alphanumeric code, which can be manually entered into the system for access to the UDI information.
If there are space constraints limiting the use of both the AIDC and HRI on the label, then only the AIDC is required to appear. However, on devices that are intended to be used in home-health care or other non-medical facility settings, the HRI would be required to appear.
Single-use devices may contain the UDI Carrier on its lowest level of packaging rather than on the device itself.
Reusable devices must include the UDI Carrier on the device itself, unless any type of direct marking would interfere with the safety or performance of the device, or if it is not technologically feasible to directly mark the device. If so, this should be properly documented in your design history file.
Most importantly, the UDI Carrier must be readable for the intended lifecycle of the device.
Below is an example of a GS1 AIDC and HRI barcode label.
Element 3: Storage of UDI information by Economic Operators
Storage of UDI information by "Economic Operators" is the third element of the UDI system. 2017/745 Articles 2(35), 22(1), and 22(3) define an economic operator as:
- A manufacturer
- An authorized representative
- A distributor
- An importer
- An investigator for clinical investigations
- A person who sterilizes systems or procedure packs
Class III, implantable device:
According to EU MDR 2017/745 Annex II, the manufacturer shall keep an updated list of all UDIs that it has assigned. Economic operators and all health institutions are required to store, preferably by electronic means, the UDI of all the devices for which they have supplied or with which they have been supplied.
For Devices Other than Class III:
Member States are encouraged, and in some cases require, health institutions to store, preferably by electronic means, the UDI of the devices with which they have been supplied. The UDI must also be included in any field safety notice for reporting serious incidents and field safety corrective actions.
The EU MDR and EU IVDR also give the European Commission authority to make additional requirements regarding the submission or maintenance of UDI information. In making those decisions, the European Commission must consider six (6) areas:
- Confidentiality and data protection
- Risk-based approach
- Cost-effectiveness of the additional measures
- The need to avoid duplications in the UDI system
- The needs of the healthcare systems of the member states
- Harmonization with other medical device identification systems
To continue reading this eBook including information about the EUDAMED database, UDI format requirements and issuing entities, implementation timelines, and key differences between the EU and US UDI systems, please register to download the full version

Key steps to help you streamline regulatory process management
Regulatory affairs is constantly changing, and these changes span the entire product lifecycle - from pre-market awareness of global regulations to managing varying and changing market placement and post-market activities. Managing all types of regulatory changes without streamlined processes and methods in place is a daunting task and one that can set regulatory affairs teams up for risks, including project delays, non-compliances, financial impacts, and employee turnover.
With regulatory affairs interactions and activities often spanning various departments, office locations, and external stakeholders, many regulatory affairs teams are left wondering how they can better streamline their process management.
We recapped core methodologies from our recent webinar, Navigating regulatory change: Why streamlined process management is critical for medtech regulatory teams, to help RA teams review their existing processes, identify gaps, and put together a remediation and implementation plan that will enable them to reduce risks and maintain regulatory continuity.
Phase 1 – Assess your team’s current processes
Assigning resources to provide an honest assessment of your regulatory processes can be tough. We've seen the most success by having awareness and involvement from all of your team’s stakeholders, as many of them have a vested interest in your regulatory processes and information. Additionally, we've seen even greater success when those efforts are supported by an executive sponsor who's committed to streamlining these processes and making the changes.
When we talk about assessing current processes, we don’t just mean reading common SOPs and flow charts. We're talking about an honest review of the adequacy of your regulatory inputs. Some questions you can ask to help with your assessment are:
- Do we have everything that we need when we need it?
- Does this process generate valuable deliverables? How hard are we working to generate these outputs?
- Do we have the right tools in place? If not, what tools on the market can help us achieve our goals?
It’s safe to say you can spend less time on processes that you've identified are working well. If you don’t know exactly where to start in a process assessment, it's helpful to think about where you spend most of your time. When focusing on the processes that are taking up most of your time, it’s easier to see where the inefficiencies lie.
Once the opportunities for improvement are identified, it's time to consider all potential risks. Compliance risk is an important thing to consider, but there’s also business risk with slow processes, inefficient processes, or even worse, ineffective processes where RA professionals spend a lot of time arriving at the wrong output or no output at all.
As risk is assessed for these opportunities, you should then consider the effort that it's going to take to resolve each. This measurement doesn't have to be highly specific. The intent for it is to ultimately help you prioritize which regulatory process changes you want to execute with your team first.
Phase 2 – Planning for Improvement
While there is some planning that needs to take place at the time that you kick off the assessment phase to help you understand and establish who's going to assess which processes., this phase is intended to focus on planning for improvement. It starts with prioritizing your inefficient processes from highest to lowest. Gaps should be prioritized and resourced first without trying to “boil the ocean.” This will help your team set itself up for a successful implementation phase next. From our experience, it’s hard to appropriately focus on process improvement when trying to change too many processes at once.
Within the planning phase, you can review the results of the assessments with all of the regulatory stakeholders and any other stakeholders who are involved with the outputs of the processes. The process owners can then take this opportunity to break down silos by understanding the relationship that their processes have with other departments and the effect their outputs have on these departments. Taking the time to understand the impact your processes have on other departments will allow you to be able to make any necessary adjustments to the inputs and outputs of your processes and hone your communication strategies.
From here, you can assign resources and tasks to manage the overall effort with regular check-ins.
Phase 3 – Implementation
Implementation, the easy part as we jokingly say. As you work through the implementation tasks identified in the planning phase and check in with your stakeholders, it's a good idea to test and iterate on your changes to ensure that they continue to make sense. This is also crucial to verify that your changes are advancing the project or the task toward your overall process management goals.
Training is a key element in the implementation phase as well. Any upfront communication that can be provided to the users of the process ahead of the training is going to be beneficial for change adoption and change management. It’s important to not only communicate that process changes are coming but also why they are coming and what benefits the end users of the process and the consumers of the outputs expect to see as a result.
Once those process changes have been implemented, and training is completed, it’s important to measure and monitor the process for the effectiveness of the changes. Some key questions to consider here are:
- Did we gain efficiencies?
- Did we make our lives easier with these process changes?
- What impact have these process changes had on our team, other departments, and our organization?
If you can’t answer these questions positively, it’s important to go back through the assessment planning phase activities to make sure all improvement opportunities and tasks were properly identified and assigned. When you’ve found those changes have made a positive impact, you should communicate those successes with all relevant teams to build a success story within your organization and to encourage additional adoption of these changes.
Our webinar replay, Navigating regulatory change: Why streamlined process management is critical for medtech regulatory teams, has more tips to help you optimize your process management and explains how regulatory tools such as RIM systems can help RA teams automate, track, and manage their processes across global internal and external teams. Download the full replay here.

Rimsys UDI Overview
UDI: More than Just a Barcode and Label
Unique Device Identification (UDI) is a global requirement mandated by regulatory bodies in various countries to facilitate the easy tracking of key medical device information throughout the supply chain. This system ensures traceability from the moment a device is manufactured until it is used in a medical facility or at home.
The UDI system mandates specific labeling requirements, including the placement of a UDI number, a barcode, and essential device information on the medical device's label. This facilitates the straightforward identification and tracking of the device.
The importance of UDI to regulatory affairs teams
The obligations of the device labeler extend beyond just labeling. Manufacturers are also required to submit and update device information in regulatory databases specific to each country where the device is marketed. This part of the regulation underscores the importance of managing UDI data effectively, as it is critical for legally marketing and maintaining medical devices in different markets. As a result, the responsibility for UDI compliance is increasingly recognized as falling within the purview of regulatory affairs departments within manufacturing companies.

The Unique Device Identification (UDI) system, initially introduced by the US FDA, has since been adopted by regulatory authorities worldwide. These authorities are developing their own UDI programs tailored to their countries to deliver similar patient benefits. Each country's UDI program typically mandates specific labeling and device data reporting and maintenance. While there are overlapping elements for the UDI data required, individual countries have set up additional, localized requirements. This creates a layer of digital complexity through unique regulatory database requirements, interfaces for data entry, and the need for machine-to-machine submissions to handle large-scale reporting.
The increasing blend of shared and unique UDI data requirements, along with country-specific regulatory database needs, highlights the importance of developing comprehensive compliance solutions. The drive for digital transformation in this area is fueled by the intensive data demands from both regulators and manufacturers. This transformation aims to simplify the management of expanding requirements and address the growing complexity as more countries adopt UDI programs.
As UDI programs and the necessity for database reporting become mandatory in more countries, manufacturers and labelers must be ready to establish and maintain UDI datasets for both new and existing products in those markets.
How Rimsys can help
Rimsys regulatory management software offers a platform that simplifies the creation, maintenance, and reporting of UDI data. It also provides tools to oversee and manage the entirety of a company's UDI program through a unified solution.
Business outcomes supported by the Rimsys UDI module:
- Remove the risk of data entry error that comes with keeping identical data sets manually in sync - The Rimsys solution allows users to create and manage UDI attribute data from a centralized location and then apply that information to global UDI requirements, where the data requirements overlap multiple markets.
- Reduce the burden of keeping up to date with each country's UDI program - Rimsys monitors global UDI regulatory changes and adds new country requirements directly into the platform as UDI programs are implemented and become required. Rimsys also keeps up with the latest changes to supported UDI programs for the US (FDA) EU (MDR), Saudi Arabia (SFDA), China (NMPA), South Korea (MFDS), and Singapore (HSA) and updates the required fields directly into the platform.
- An open API ecosystem allows "source of truth" data to be integrated into Rimsys as a "post go-live" phase - Ensure data is up to date and locked at the source of truth, yet centralized for application to global UDI requirements in Rimsys. Data that is required but not controlled in a customer's source system can be managed within Rimsys.
- Built-in support of machine to machine (M2M) transmission to GUDID (FDA) with EUDAMED (EU) coming soon - Rimsys alleviates the need to manually upload UDI data into databases when relevant information changes to ensure compliance. Acknowledgments from machine-to-machine interactions are saved directly to Rimsys and associated to each UDI record.
- Leverage the existing product hierarchy in Rimsys to efficiently manage Basic UDI to reduce non-compliance risk for EU MDR - Data requirements for Basic UDI established in Rimsys are included with the M2M process - Coming Soon
- Facilitate impact assessments in Rimsys since UDI information sits alongside product and registration data - Eliminate the need to manually combine disparate data sets.
Ready to see how Rimsys software can help you create and manage the complexities of UDI data? Schedule a custom demo here.

A look at the FDA Total Product Life Cycle Advisory Program (TAP)
The Total Product Life Cycle Advisory Program (TAP) is a voluntary pilot program launched by the FDA’s Center for Devices and Radiological Health (CDRH) in October 2023. The TAP Pilot is one of the commitments between the FDA and industry as part of the MDUFA V reauthorization, which aims to provide faster patient access to safe and effective medical devices, increase innovation, improve patient safety through enhanced surveillance and data collection, and provide a more efficient regulatory process for FDA and industry.
Taking a medical device from concept to commercialization in the United States is often a long and challenging process that involves participation and adoption from FDA, clinicians, payers, and patients. The TAP program is addressing the obstacles that device manufacturers often encounter throughout this process with:
- Early and frequent interactions: FDA will provide more opportunities for sponsors to interact with the agency early in the development process, which can help to identify and address potential issues early on.
- Strategic input from stakeholders: The program will involve input from a variety of stakeholders, including clinicians, patient advocates, and payers, which can help to ensure that the development of new devices is meeting the needs of patients and the healthcare system.
- Proactive, strategic advice from CDRH: FDA will provide proactive and strategic advice to sponsors throughout the development process, which can help to reduce the risk of regulatory delays.
Currently, TAP program membership includes the companies or individuals developing the medical devices, the medical device sponsors, dedicated staff within the CDRH, stakeholders consisting of clinicians, patient advocates, payers, and academic experts, and independent advisors. However, It is unclear if independent advisors will continue to be part of the program as TAP expands. Additionally, specific individuals involved in the TAP program at a given time will vary based on the device being developed and the stage of the development process.
While currently still in its pilot phase, the TAP program is open to a limited number of medical devices. To be considered for the program, device manufacturers must have breakthrough designation with no previous pre-submission meetings. The TAP program started with 15 cardiovascular devices last fall and is now at 31 enrolled devices as of February 2024. Enrollment could reach as high as 60 devices by the end of 2024.
As part of the MDUFA V reauthorization, the TAP Program shares the goal of facilitating the development of high-quality, safe, effective, and innovative medical devices. Additionally, the TAP Program strives to reduce device development time and costs through early and frequent feedback from FDA, increase innovation with more predictable and efficient regulatory pathways for new devices, and improve patient access to new devices.
Overall, the TAP program’s focus is on improving the medical device landscape by addressing various challenges and opportunities throughout the product lifecycle. The program's success will be measured by its ability to expedite development, foster innovation, ensure device quality, and ultimately, improve patient access to these potentially life-changing technologies. For more information about the TAP program and enrollment in it, visit FDA’s website.
_thumbnail.avif)
Are FDA risk classifications and submissions any different for SaMDs? [VIDEO]
The number of software as a medical device (SaMD) is growing and with it are questions about how to effectively obtain market clearance for them. One question we hear often is, “Are FDA risk classifications and submissions any different for SaMDs?” Currently, the FDA is regulating SaMDs the same way it’s regulating traditional medical devices. As a result, you’ll still have the same three risk classifications, Class I, Class II, and Class III.
The submission process is also the same. Most Class I devices are 510(k) exempt, and most class II devices would fall under a 510(k) or De Novo submission depending on whether or not substantial equivalence can be made to another US-marketed device. Most Class III devices require PMA submissions.
There have been discussions about FDA pre-certification programs and following IMDRF guidelines for SaMDs, which would alter the submission process and also the risk stratification of SaMDs. However, none of these discussions have matured. The FDA continues to mirror their risk classification and submission guidelines for SaMDs and traditional medical devices.
Watch the full answer to this question from our recent panel discussion with subject matter expert, Prabhu Raghavan of MDQR Solutions, below.
You can also download the full replay here to get answers to other common SaMD questions such as:
- How is the FDA regulating AI/ML in SaMDs?
- What is a Predetermined Change Control Plan (PCCP) for machine learning-enabled medical devices?
- What cybersecurity considerations sh ould be made when taking SaMDs to market?

An introduction to standards for medtech companies
A standard is a published document that is established by consensus and is approved by a recognized body (ISO, IEEE, UL, etc.). It outlines requirements, specifications, guidelines, or characteristics that are used in a repeated way to ensure that materials, products, processes, and services are developed for a specific purpose. Think of it as a formula for an agreed upon way of doing something that establishes the best way of performing a function. It could be developing a product, managing a process, or even supplying materials to a manufacturer.
Why are standards important?
Standards enable technology to work seamlessly across industries and markets and help to build consumer trust that products and services are designed to work together in an efficient way. They form the fundamental building blocks for product development by establishing consistent requirements that can be universally applied, practiced, and understood. For example:
- Quality standards reduce product failures on the assembly line.
- Environmental standards reduce environmental impacts, reduce waste, and provide sustainability.
- Health and safety standards reduce accidents in the workplace.
- Food safety standards prevent food from being contaminated.
To ensure standards stay relevant and are aligned with technology changes, many standards organizations require that their standards be reviewed periodically and updated as necessary.
Is an industry standard the same as a regulation?
No, but there is a very close relationship between the two. Simply put, a standard is a guideline whereas a regulation includes laws. Industry standards are used voluntarily (although strongly encouraged) while regulations are not voluntary because they are a requirement from a government agency or similar authority, i.e. ISO 13485 is a standard and building codes are regulations.
- Standards are technical documents, driven by consensus that are crafted by experts.
- Regulations are sometimes based on standards, created by a variety of individuals and entities, while overseen by federal, state, and/or municipal authorities.
It's important to note that while standards aren't mandated by law, many regulatory authorities recognize standards and recommend adherence to them in order to promote safety and quality.
What kind of standards are medtech manufacturers responsible for tracking?
The medical device industry has the responsibility for the design and manufacturing of a wide range of products used to diagnose and treat illnesses to improve health in patients. Medical device standards help ensure that a manufacturing or design process can consistently produce the quality required to serve patients and healthcare professionals around the world.
Some of the more common standards used by the medtech industry include, but are certainly not limited to:
ISO 9001 – A general standard (not industry specific) for quality management and implementing a rigorous quality system. For medtech specifically, it helps with the management of the quality control process by helping to keep costs low, improve accountability and simplify regulatory compliance.
ISO 13485 – This standard is designed for medtech specifically and expands on the framework set up by ISO 9001. Compliance with this standard helps with quality control, process validation, and risk management, often referred to as the risk management standard.
ISO 14971 – This standard specifies terminology, principles, and a process for the risk management of medical devices, including software and in vitro. This helps to identify hazards that may be associated with devices and to minimize those risks.
IEC 60601-1 – Medical Electrical Equipment, general requirements for basic safety and essential performance of equipment.
ISO 10993 – Biological evaluation of medical devices that includes biocompatibility testing of materials used to design product parts that would come into contact with a patient, testing for skin sensitization, and irritabilities.
ISO 15223 – Symbols to be used with information to be supplied by the manufacturer. This standard identifies symbols that are globally accepted to be used in a broad spectrum of medical devices. These symbols can be placed on the device, on the packaging, or on any accompanying information such as instructions for use.
ISO 45001 – This standard outlines the requirements for occupational health and safety management systems that can be employed in the medical device industry to help reduce occupational risk.
Where do I get these standards?
Standards used to design and build medical devices need to be purchased, and you must always maintain the most current revision of that standard to ensure proper adherence to it. They can be purchased as electronic copies, or you can request a paper copy for your files. You can purchase them directly from the standard organization (ISO, IEC, ASTM, UL, etc.). There are also organizations services that will provide standards from many organizations, serving as your to be your one-stop shop.
How do I know when standards change?
The best way to manage how you receive information about changes to industry standards would be to implement an electronic standard tracking system. These systems help to:
- Give you early notifications of changes
- Mitigate your company's risk by ensuring you're up-to-date
- Save you time by eliminating the tracking on your own
- Ensure your standards are up-to-date
Using manual processes such as spreadsheets to manage standards updates can be difficult, time-consuming, and lead to compliance risks - especially when a high number of standards and markets are involved. There are a variety of standards management tools to help medtech companies monitor and manage global standards, including Rimsys.
How can Rimsys help?
Rimsys’ regulatory management software offers standards management to help you stay ahead of the mayhem by providing:
- Access to a library of over 1.6 million global standards through a partnership with IHS Markit
- The ability to link standards to individual products to more easily assess the impact of changes across your product portfolio
- Automatic alerts when standards are changed, superseded, or withdrawn to reduce compliance risks and enable faster reaction times
- Bulk updates to your essential principles/GSPR tables when standards change for easier maintenance and compliance
For more information, visit www.rimsys.io/solutions/standards-management.

Why should you invest in your regulatory team? Easy Medical Device podcast interview
Recently, our Founder and CEO, James Gianoutsos, was a guest on an episode of the Easy Medical Device podcast. Hosted by Monir El Azzouzi, a quality and regulatory professional with over 16 years of industry experience, the Easy Medical Device podcast explores a wide range of topics, news, and challenges to help medtech quality and regulatory professionals gain valuable insights that will help them excel in their roles.
In the episode, Why should you invest in your regulatory team?, James and Monir explore the limitations of traditional cost-center approaches to resourcing and preparing budgets for regulatory affairs teams and discuss the benefits of treating regulatory affairs as a revenue function. Hear their thoughts about:
- How regulatory affiars teams are typically structured
- The importance of the RA job function on revenue
- The impact AI will have on regulatory affairs
- How digital tools can enable RA teams
James also provided tips RA professionals can use to convince their stakeholders to invest in regulatory affairs teams. He emphasized that getting buy-in often involves a mindset shift that will change the dynamic of the conversation. For example, when planning for a renewal, think about the financial impacts of missed renewals rather than the sheer volume of renewals you're doing.
When you're trying to convince your leadership team, don't talk about how many renewals I did for this product in a particular month. talk about the dollar figures you saved the company or retained on the market.
For more tips, listen to the full interview on the Easy Medical Device website.




