

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

To build or to buy: evaluating options for Regulatory Information Management
Your regulatory team needs dedicated software to manage market entry activities, maintain regulatory integrity, and ensure post-market compliance. While small medtech companies often start out managing regulatory data in spreadsheets, this quickly becomes unwieldy.
Can you develop a system that tracks product information and registration expiration dates? Yes, absolutely – especially if your medical device company has internal software development capabilities as part of your IT team. However, a strong RIM system will also give you the ability to completely manage market entrance documents and regulatory workflows. And building a RIM system will also require significant input from your regulatory and quality teams, in addition to IT resources.
Admittedly, we are a bit biased here, but this is the reason we started Rimsys – to create regulatory order in the medtech community and help regulatory professionals automate processes and digitize information so that they can spend more time on activities that truly make a difference for their organizations.
Before you begin a project to build your own RIM system, or to modify an existing system to meet regulatory needs, consider the entire size and scope of the project. This article discusses the common areas where custom-built RIM projects can run into unanticipated costs or issues.
Meeting software regulatory requirements
RIM systems are the source of information used by your regulatory team to provide accurate and timely information to regulators and auditors to ensure that your organization is compliant with existing regulations. This means that the software system itself needs to meet certain requirements. To ensure a compliant and secure RIM system, you need the following:
- ISO 9001 certification
Your organization may already be ISO 9001 certified, but in developing your own software to manage internal data and processes, you are greatly expanding the scope of your ISO 9001 project.
- ISO/IEC 27001 certification
ISO/IEC 27001 is the global standard for information security management, including data protection and cyber security and resilience. You will need to obtain ISO/IEC 27001 certification for your RIM system.
- 21CFR Part 11 compliance (US) and EU annex 11 (EU)
21 CFR Part 11 is the portion of US federal regulation that addresses electronic records and electronic signatures as related to FDA processes and documents. The EU Annex 11 is the equivalent regulation in the EU. A good RIM system is designed with Part 11 and Annex 11 compliance in mind and can easily be validated to the regulations. You will need to demonstrate procedures that ensure all electronic records kept in the RIM system are controlled, authentic, and can be verified. Features such as data audit trails and specific electronic signature requirements need to be implemented.
- SOC II Type 2
SOC II Type 2 may be used in place of ISO/IEC 27001 to demonstrate suitable data security, particularly in cloud-based systems. SOC II Type 2 reports prove a company’s controls, but are not a certification provided by an independent registrar. SOC II Type 2 also requires an Informational Security Management System (ISMS), which is the framework focused on risk management and risk mitigation.
- GDPR compliance (EU)
While often associated with email marketing activities, the EU General Data Protection Regulation requires companies that store any information about an EU citizen to have specific safeguards in place. In particular, if your RA team includes EU citizens then their personal data is subject to GDPR and, among other things, they have the right to request their data is deleted from the system if they leave the company. All personal data needs to be protected from outside access as well.
Reducing overall cost of ownership
Building a RIM system from scratch or building RIM features into a QMS or PLM system is not a one-time endeavor. Consider the following on-going activities that will be required:
- Addressing regulatory changes
Global medtech regulations are constantly changing. For example, Rimsys created an entirely new module to handle Unique Device Identifier (UDI) requirements as countries announced compliance dates related to UDI labeling and databases. In this example, and in others, each country has different requirements regarding the data that needs to be stored, the format of that data, and the ways in which it is to be reported.
A RIM system is not just a software development project. It requires the attention of regulatory professionals who can ensure that the system is properly handling the requirements of each country in which your device is marketed.
- Managing validation documentation
As with a medical device, a validated RIM system cannot be modified without following specific and documented procedures designed to ensure the system’s integrity. Any time a new feature is added, or a change is made to the system – whether it be a small bug fix or the addition of a major new function to address an updated regulation – the affected part of the system will need to be revalidated.
- System support
The cost of maintaining and supporting a system as complex as a RIM system is significant. Such costs include not only the development costs, but the cost to train and support users of the system on an ongoing basis. If you are using internal resources, as many companies do, it is important that you include the lost opportunity cost for your development team in cost calculations. What are your developers not working on while they build your RIM system?
Consider carefully whether your IT team is positioned to become a software development team in the long-term. An IT team that is advocating for an in-house solution should be able to provide a plan for how often new features will be provided, how the system will be supported, and how an ongoing product roadmap will be managed.
Reasons not to build a RIM system in-house
Considering the above information, the primary arguments you can make against building a RIM system in-house are:
- Building a RIM system is not just a software development project. We will need to stay on top of changing regulations and requirements and be prepared to update the system frequently. Note that this is the primary argument to be made when an IT team is pushing for an in-house solution (a situation we see frequently).
- A RIM system built with internal resources builds your existing regulatory process into the system. Are you sure that those processes can’t be improved upon? A RIM system that is used by many medtech companies not only includes built-in industry best practices but will evolve to support new workflows and processes as the industry changes. A custom-built RIM system will have none of those advantages.
- The system will need to be validated and certified according to several standards and regulations, like our medical devices. This has the potential to significantly increase the scope of our ISO-related processes and other internal procedures.
- Purchasing a dedicated RIM system from a company that is solely focused on providing up-to-date functionality for regulatory professionals is a safer and simpler choice.
We have worked with a number of companies that ultimately chose to implement Rimsys after attempting to build a RIM system in-house. Faced with the unexpected complexity of the development project, they ultimately chose to go with a packaged solution. Be sure to carefully evaluate all potential costs, including on-going costs, when making the build vs buy decision.

Post-market surveillance for medical devices in the European Union
This article is an excerpt from Post-market surveillance for medical device in the European Union.
Table of Contents
- What is post-market surveillance?
- What classes of medical devices require post-market surveillance?
- Components of a successful post-market surveillance plan
- PMS data requirements
- Post-market surveillance system goals
- Required post-market surveillance reporting
- Embracing post-market surveillance as an integral part of your quality program
- Getting started with post-market surveillance
Post-market surveillance (PMS) is designed to monitor the performance of a marketed medical device by collecting and analyzing field use data. Article 10 of the EU MDR and IVDR requires all device manufacturers to have a post-market surveillance system in place. The main elements of the PMS are laid out in Article 83, and additional details for lower-risk and higher-risk devices are covered in articles 84 and85, respectively.
In general, a PMS system consists of both proactive activities and reactive, or vigilance, activities. While post-market surveillance and vigilance are sometimes used interchangeably, vigilance consists of separate activities that feed post-market surveillance programs.
Post-market surveillance systems are used to collect and analyze data not only about the manufacturer’s device but also about related competitors’ devices that are on the market. Data collected through PMS procedures is then used to identify trends that may lead to, among other things, quality improvements, updates to user training and instructions for use, and identification of manufacturing issues.
Note that “market surveillance” encompasses activities performed by a Competent Authority to verify MDR compliance, and should not be confused with the topic of this ebook,“post-market surveillance," which is performed by the manufacturer.
All medical devices marketed in the EU require some level of post-market surveillance, and all medical device manufacturers must implement a post-market surveillance system (PMS). The requirements of the PMS, however, vary and should be “proportionate to the risk class and appropriate for the type of device” (MDR Chapter VII). In particular, the type and frequency of reporting vary based on a device’s risk class.
A post-market surveillance plan (PMS) is an integral part of a manufacturer’s quality management system and provides a system for compiling and analyzing data that is relevant to product quality, performance, and safety throughout the entire lifetime of a device. The PMS should also provide methods for determining the need for and implementing any preventative and corrective actions. A PMS system should include and define:
Surveillance data sources
With the increased focus on proactive risk identification in the MDR, it is important to design post-market surveillance systems that actively acquire knowledge and detect potential risks. It is not sufficient to rely solely on spontaneous reporting by healthcare providers, patients, and other stakeholders.
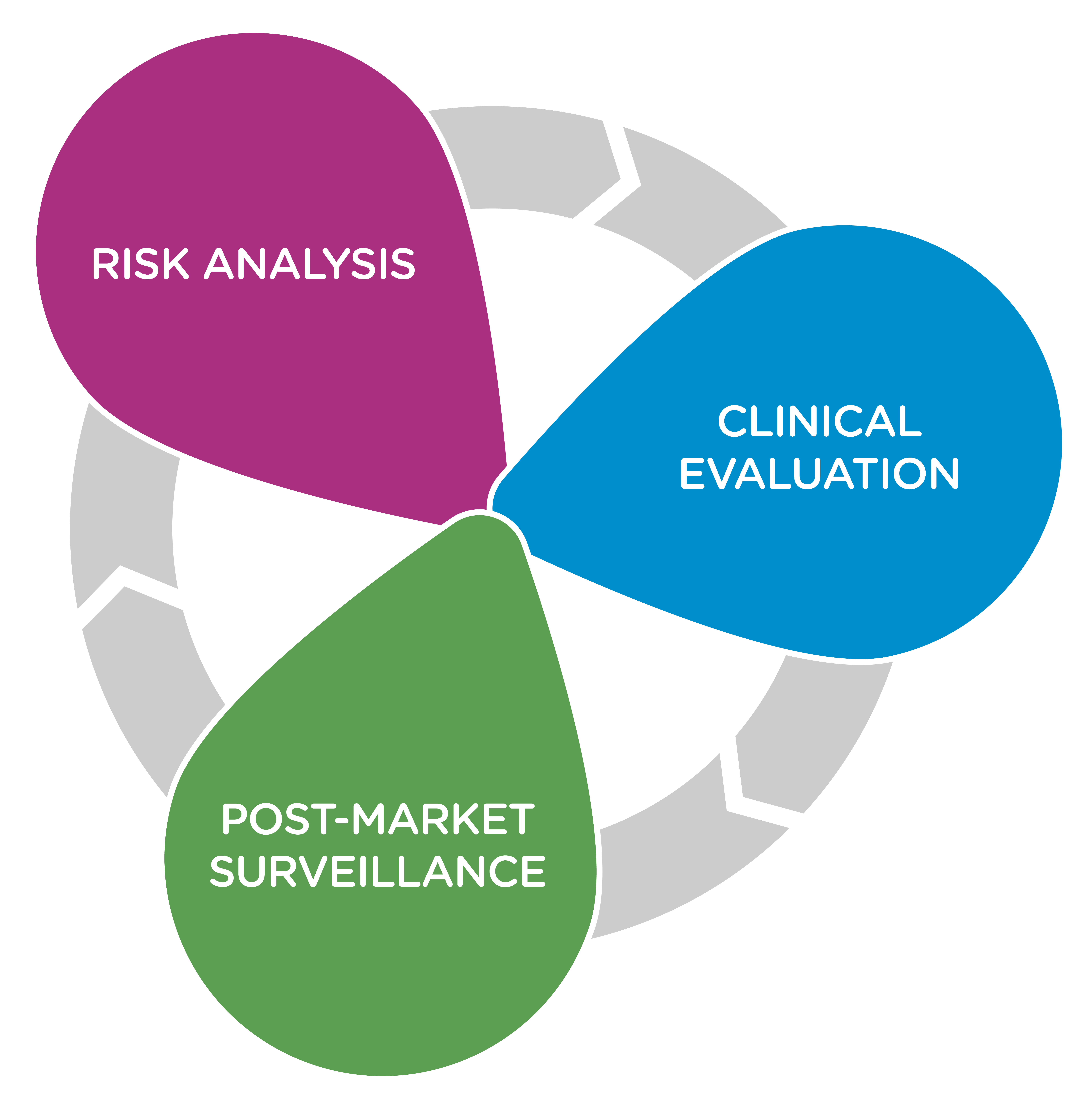
In addition to information coming from Clinical Evaluation Reports and complaint and adverse event reporting, typical sources of surveillance data include:
• Social media networks: Because many of your stakeholders may be communicating on social media networks, it is important to employ social listening techniques and/or tools to identify issues and concerning trends as they develop.
• Industry and academic literature: Any studies, academic papers, and other literature that addresses similar devices or the specific use cases for which your device is designed should be evaluated. In particular, risk factors and adverse events identified with similar devices should be closely examined. It is also important to identify newer technologies that may affect the benefit-risk ratio and establish a new definition of “state of the art” for the device type.
• EUDAMED: While the European Database on Medical Devices (EUDAMED) is not yet fully functional, it is intended to provide a living picture of the lifecycle of all medical devices marketed in the EU. Manufacturers should take special care to consider information for similar devices made available through the EUDAMED system in the future.
• Registries: Patient, disease, and device registries can provide information that informs the clinical evaluation process which provides input into the post-market surveillance system.
Data analysis methodology
A well-defined data analysis methodology will accurately identify trends and lead to defendable decisions in the application of post-market experience. Once the necessary information has been identified and collected, and potentially cleaned of incomplete or otherwise unusable data, the data needs to be analyzed.
The goal is to identify meaningful trends, correlations, variations, and patterns that can lead to improvements in the safety and efficacy of the device. There are many data analysis tools available that can assist with:
• Regression analysis that will identify correlations between data (e.g. the device location/geography correlates to battery life).
• Data visualization that can be useful in spotting trends in the data.
• Predictive analytics, which can be particularly useful with large data sets, to identify future trends based on historical data.
• Data mining, which is also normally used with large datasets, to organize data and identify data groups for further analysis.
Benefit-risk indicators and thresholds
The MDR requires that medical device manufacturers not only demonstrate the clinical benefit of their device but also quantify the benefit-risk ratio. The benefit of a device must be shown to clearly outweigh the risk for it to gain market approval. Article 2 (24) of the MDR defines the benefit-risk determination as “the analysis of all assessments of benefit and risk of possible relevance for the use of the device for the intended purpose when used in accordance with the intended purpose given by the manufacturer.”
A PMS system should clearly define benefit-risk calculations and the data used to support them. Post-market surveillance activities are critical in order to re-evaluate and maintain the benefit-risk calculations and determinations of a device throughout its life. Information that is gained through a PMS system can lead to:
• Identification of new risk factors.
• Adjustments to risk frequency and/or severity values based on actual use data.
• Adjustments to established risk calculations based on new “state of the art” technologies becoming available.
• Adjustments to established benefit calculations based on actual use data.
While complaint handling and other feedback tracking are more often described as part of post-market vigilance systems, they play a role in the more proactive post-market surveillance processes as well. A PMS system should define ...
To continue reading this ebook, download the full version.

Regulatory should be a revenue function
“Regulatory has a seat at the table.” This quote, while seeming innocuous, is actually quite meaningful. The context is a discussion about regulatory digital transformation. At RegUP Boston, one of our customers was discussing the organizational conditions that led to a large-scale digital transformation initiative across regulatory affairs for an enterprise medical device manufacturer. The change was precipitated by a clear awareness of the impact that regulatory affairs has on both the top and bottom lines of the business. As a result, the regulatory team was given a voice in business strategy, and an opportunity to invest in growth.
The challenges of ‘cost-center’ thinking
At some level, this would seem obvious. Of course regulatory affairs is strategic in a highly-regulated industry. Yet despite this, most RA teams are treated primarily as a cost center—a function that doesn’t directly contribute to revenue or profit. This means that the business is continually looking to minimize the cost incurred by regulatory activities, but more importantly, it leads to significant friction between teams. Rather than a go-to-market partner, regulatory is viewed as an operational impediment to revenue generation by sales and marketing teams.
Cost-center thinking also manifests itself in poor measurement and objectives for regulatory teams. Often regulatory teams are assessed by volume-based metrics: number of submissions completed, speed of submission completion, and percentage completed ‘on-time’. While these are decent measures of output, they don’t have a direct correlation with revenue. Submissions or renewals that can be completed quickly aren’t necessarily associated with the highest-value products or markets.
Staffing levels also aren’t well optimized in a cost-center mindset. Apart from attempting to minimize full-time regulatory staff, many companies allocate regulatory headcount based on device risk classes or number of markets served. Again, these are good benchmarks for ongoing work volume, but they don’t necessarily align with future go-to-market plans or revenue targets for different product lines. This approach can leave RA teams under-resourced, and force businesses to rely on consultants when regulatory staffing levels “unexpectedly” don’t match got-to-market needs.
Regulatory is a key contributor to revenue and profit
Treating regulatory affairs as a cost center misses an important reality: regulatory clearance is an essential aspect of revenue generation in the medtech industry. It’s a prerequisite for growth, as any new product or geographic expansion of an existing product requires a new market submission and approval from health authorities before it can be sold. Regulatory affairs also has direct responsibility for sustaining revenue. The regulatory lifecycle of a product doesn’t end after market clearance. RA teams ensure the continued revenue stream from a product by keeping track of license expirations, relevant regulatory and standards changes, and managing post-market surveillance activities.
Unlike (necessary and valuable) support functions like accounting or IT, there’s a direct line between regulatory activities and revenue for the business. This means that RA functions are obviously important, but also that alignment between sales, marketing, and regulatory affairs is necessary for go-to-market success.
An analogy: sales & marketing alignment
There is a similar dynamic at almost any B2B business between sales and marketing teams. Nobody would question that both sales and marketing have responsibility for revenue generation, but that doesn’t always mean they are closely aligned. If teams don’t have an agreed-upon revenue target (X% of sales should be driven by marketing activities), marketing teams can end up measuring themselves on things that have less direct business impact such as website traffic or re-shares of social media posts. This leads to conflict between teams as different activities are prioritized, and sales teams perceive that marketing isn’t an active, helpful partner.
It’s not that marketing teams aren’t executing in the outlined scenario, it’s that the measures and priorities aren’t aligned. When sales and marketing share a defined revenue goal, upstream measurements like new lead and pipeline generation guide marketing activity prioritization. Marketing reports on results that are relevant to sales goals, and sales teams have clarity into how marketing is contributing. The result is an aligned and productive, rather than adversarial, go-to-market motion.
What does revenue-aligned regulatory affairs look like?
Note that this is not an accounting discussion. Alignment here is not about how medtech companies should account for expenses associated with regulatory compliance. Rather it’s about how regulatory objectives and investments should be structured. Changing those structures to be revenue aligned produces two beneficial outcomes.
The first is in regulatory planning. When marketing activities are derived from revenue goals (like pipeline generation) the result is that activities that have the highest revenue impact are prioritized. If regulatory affairs teams carry a revenue target, the projects that are prioritized are those with the highest revenue impact. This simple criterion drives closer alignment between regulatory, marketing, and sales teams, and prevents priorities from being determined by the “loudest” voices in the room, or project length/complexity—which can happen when RA teams are measured on activity alone.
The second outcome is a shift in investment strategy. In a cost-center mindset, all investments are designed to minimize costs. In a revenue mindset, investments are driven based on expected returns. When regulatory projects are prioritized according to revenue impact, it’s easier to allocate headcount based on the anticipated workload as a return on each additional hire can be easily estimated. The same goes for investments in technology and tools. With a direct line between work product and revenue, it’s easier to make the business case for investment. In the same way that marketing campaigns require investment to generate sales opportunities, regulatory projects require investment to create revenue for the business.
Revenue alignment improves organization and focus for regulatory affairs teams. It allows them to effectively prioritize activities, and plan to adequately staff them. It reduces a focus on activity for activity’s sake and instead strengthens alignment across all go-to-market teams. It also makes it easier to justify investments in improving regulatory processes. From new tools to end-to-end digital transformation, regulatory affairs can be optimized to deliver on revenue projections.
Why give regulatory a “seat at the table”?
Medical device and in vitro diagnostic companies simply won’t have a choice. The current approach to managing regulatory affairs isn’t keeping up with the pace of change in the industry. The MDR and IVR rollout in the EU is expected to leave 50% to 76% of the products currently on the market behind. RA teams that are measured on work volume (as much as possible), at the lowest possible cost aren’t effective in this environment, and the organizational friction between them and other go-to-market teams will only further hinder execution.
Companies that succeed in today’s environment are those that take a different approach to regulatory affairs. By treating regulatory as a revenue function and aligning regulatory activities to financial goals, companies can more strategically plan for regulatory workload. They can prioritize projects that have the largest impact on the business while reducing churn and repetitive administrative work within the team. They can justify investments in productivity and process improvement by tying them to expected return for the business. And they create tight alignment across marketing, sales, and regulatory affairs in an integrated go-to-market motion for the business.

Ask us Anything ... about China submissions!
Your submission questions answered for imported devices in China
Our latest “Ask us Anything” webinar this week focused on the topic of medical device submission strategies for China, specifically for devices being imported into China. Karen Cohn, Regulatory Specialist here at Rimsys was on-hand to answer everyone’s questions. Karen specializes in international submission strategies and was a subject-matter expert on NMPA submissions while at Philips.
In this article, we have included the most common questions we received and put them together with their answers, along with related links to additional information that was mentioned during the webinar.
Remember that you can always ask us a question by using #AskRimsys on Twitter or LinkedIn - or using our short survey. Your questions help us select topics for upcoming webinars, and every week we select one question to answer directly on our social media channels.
The following list provides direct links to relevant CMDE information (Center for Medical Device Evaluation of the NMPA). These sites are in Chinese, but we have found that Google translate does a good job of providing English versions for purposes of researching regulations, but be sure to obtain verified translations for documents and regulations that are important to your organization.
- CMDE Main Page
- CMDE QMS Guidance
- Class I CMDE Guidance (requirements and instructions are in the documents at the bottom of the page)
- Clinical Evaluation Guidance (following the guidance depends on which Sub-Category your device falls under)
- Guiding Principles
- Innovative Device Pathway
Karen’s answers are below.
Q: Do I need to have submission documentation translated into Cantonese or Mandarin?
Cantonese and Mandarin are spoken dialects of Chinese. What you are looking for is a Simplified Chinese Translation, which is the written language. There is also a written language called Traditional Chinese, but the NMPA only accepts translations into Simplified Chinese. You will also need to provide the original document as well.
Q. How do I find the NMPA standards that may be applicable to my device?
The CMDE (Center for Medical Device Evaluation) has information on its standards and guiding principles (see additional links at the top of this article).
Standards for China have differences from their ISO and other standard association counterparts. There may be different labeling, testing, and other requirements. I would highly suggest that you get verified translations of these standards and review them during the developmental period of your device.
Q. Could you elaborate on essential principles and using the EU GSPRs instead?
You can try to use EU GSPRs, but it is important that you do a gap analysis first and note that the EU GSPR will be using ISO and Harmonized Standards, whereas the NMPA essential principles will need to use NMPA standards. Note that the NMPA is very particular about the organization of their submission information, which includes essential principles.
Q. Is there a pathway for combination products, or is China similar to the EU in that the drug and the device need to be approved separately?
The short answer is that there is a combination pathway, which is closest to the pathway available with the FDA. You are required to submit to the CMDE and the CD (Center for Drugs) and they coordinate with each other and with you. I would highly recommend having a pre-submission meeting to determine which agency should be leading the process.
Q. How do I let leadership know of the personnel cost of registering in China?
It is important to align with your manager and director if you are a specialist, especially if they are not currently aware of the process of registering a device in China.
Q. How do I effectively work with my China counterparts to coordinate my submission activities?
I’ve been in situations where you are working with a China counterpart - sending them detailed information and documents mainly by email and SharePoint - but you don’t get the final documentation back to review and don’t see the final submission product. The actual submission can become a bit of a black hole, which is a compliance risk. Using a tool like Rimsys can help, because it allows you to create separate submission templates such as the NMPA PTR and the NMPA eRPS ToC. You can then use these templates to easily create the submission documents. You also can clearly assign owners and track progress for each section of the submission to clearly indicate who should be doing the work.
Q: Could you elaborate on QMS requirements?
QMS requirements have recently been updated, and the NMPA is now requiring QMS information in submission documentation. For a while, there was no guidance regarding QMS requirements, but guidance has recently been released for the inspection criteria.
Q: Could you elaborate on clinical evaluation requirements?
Clinical evaluation requirements depend on the device type. There are a number of pathways listed on the CMDE website and your clinical team will need to understand which one is applicable to your device(s).
Q: For large devices, such as MRIs, are there any unique steps or additional information we need to provide?
Building and installing a device can take 18+ months and special site requirements need to be met before clinical trials can start.
You can certainly include this information in your submission. Strongly consider sending someone from your organization with the device when it is time for type testing. It is up to the manufacturer to make sure that the type testing is done correctly, to your standard, and under conditions that are compliant with your requirements.
Q: Does NMPA have an abbreviated pathway if the device has been 510(k) cleared or EU CE marked?
China requires that the device be approved/cleared in its country of origin. This doesn’t provide an abbreviated pathway.
Q: What is a PTR?
PTR stands for Product Technical Requirements. It is a document that outlines the device specifications and testing methods based on the applicable Chinese standards. This is an important document used for the in-country testing required for registration in China and for the NMPA submission. Once approved, the NMPA testing center will stamp the PTR and send a testing summary. Both items are needed for the market access submission for Class II and III regulated products. Class I devices also need a PTR even though they do not go through type testing.
Q: Do medical device registrations expire?
Class II and Class III Registrations expire after 5 years from their certificate approval date. The renewal must be submitted six months prior to the expiration date.
If the mandatory standards for medical devices have been revised, you may need to submit a change notification. Note that a change notification and a renewal cannot be submitted in parallel, so be conscious of the requirements and leave yourself enough time to get through the submission process.
Q: Are pre-submission consultations available?
Yes, pre-submission consultations are available through the NMPA. Ensure that you document all meetings and discussions, as it is required to include meeting notes in market-entry submissions for class II and class III devices.
You may also be interested in our Ultimate guide to the China UDI system and database.

FDA databases
The FDA maintains many publicly accessible databases that are valuable to medical device manufacturers preparing product submissions, compiling post-market data, researching guidance documents, and more. We have listed some of the most commonly used databases below, along with a summary of information they provide and how they can be used.
FDA databases useful for new medical devices
Product Classification
This database contains medical device names along with the three-letter device product code and device classification. Manufacturers may use this database to properly classify a new device.
- Search the CDRH product classification database
- Read A primer of medical device classification (includes device classification information for multiple countries)
Pre-market Notifications - 510(k)
The 510(k) database includes all released 510(k) submissions and can be searched by 510(k) number, type, product code, device name, and more. In order to use the 510(k) pre-market notification process, a new device requires that an existing predicate device that also used the 510(k) process be identified. Use this database to identify devices that are substantially equivalent to the new device and meet the requirements of a predicate device.
Pre-market Approvals (PMA)
The PMA database lists pre-market approvals, including supplements that have been approved. PMA data can be searched by PMA number, device name, decision date, supplement type, and more. A new device that is substantially equivalent to a PMA-approved device means that the new device will require a PMA and be classified as a Class III device.
- Search the Pre-market Approval (PMA) database
- Read The beginner’s guide to the FDA PMA submission process
De Novo
The De Novo database includes all De Novo classification orders and can be searched by De Novo number, product code, 510(k) number, device name, and more. The De Novo process allows medical device manufacturers to request reclassification for novel devices with low to moderate risk profiles, that would otherwise be automatically classified as Class III devices.
Devices@FDA
The Devices@FDA database provides a simple search of both the 510(k) database and the PMA database. While convenient for initial searches, it only allows searches by device name and approval date.
Standards and guidance documents
CFR Title 21
The FDA provides a searchable online reference database for Title 21 that provides an organized table of contents and timelines of changes to the regulation.
FDA guidance documents
FDA guidance documents provide the FDA’s interpretation of regulatory policies, discuss the application of regulations to specific products, and also provide guidance for industry. Guidance documents can be searched for based on product, topic, issue date, FDA organization, and more. In addition, users may browse a list of guidance documents by topic.
FDA recognized consensus standards
The FDA provides a searchable database of voluntary consensus standards to which the agency will accept a declaration of conformity. Because these are standards developed by different organizations, this database can be searched by standards and organization, along with keywords, product codes, and more.
Unique Device Identifier - UDI
GUDID
AccessGUDID provides searchable access to the GUDID database of device information, including the device identifier, device name, company name, and more.
- Learn more about GUDID and searching the GUDID database
- If you are looking for information on submitting UDI information to the GUDID database, see the FDA’s GUDID submission page.
FDA post-market databases
522 Post-market surveillance studies program
This database allows users to search post-market surveillance studies by manufacturer or device. The 522 post-market surveillance studies program defines requirements for the design, tracking, oversight, and review of studies mandated under section 522 of the FD&C act.
MedSun reports
The Medical Product Safety Network (MedSun) is an adverse event reporting program launched in 2002 designed to allow the CDRH to work collaboratively with the clinical community to identify, understand, and solve problems related to the use of medical devices.
Post-approval studies (PAS)
The post-approval studies (PAS) database contains information about studies that manufacturers are required to complete as a condition of device approval. The PAS database can be searched by applicant or device information.
Medical device recalls
The medical device recall database contains recall information since November 1, 2002, and can be searched by product, recall class, product code, recall date, root cause, and more.
MedWatch
MedWatch is the FDA safety information and adverse event reporting system that is available to health professionals, patients, and consumers. Note that in addition to medical devices, MedWatch is available for reporting on medicines, biologics, cosmetics, and food.
Additional FDA databases
Establishment registration and device listing
The registration and listing database contains information on all establishments engaged in the manufacture, preparation, propagation, compounding, assembly, or processing of medical devices. It also includes listings of medical devices in commercial distribution by both domestic and foreign manufacturers. Establishment owners are generally required to register their facilities and devices with the FDA annually.
Total product lifecycle (TPLC)
The TPLC database includes both pre-market and post-market data about medical devices, including PMA and 510(k) approvals, adverse events, and recalls. The TPLC database can be searched by device name or product code and includes full reports by product line.
Do your research!
In many cases, the same information is contained in multiple databases, so take the time to understand which databases provide the right combination of data for your needs. This article references only a portion of the FDA database. See the FDA’s Medical Device Databases listing for all of the available FDA databases.

IMDRF: International Medical Device Regulatory Forum
The medical device industry is vast and diverse, with each country or regional authority having its own regulatory body and requirements. For instance, medical devices are classified in ascending order in the United States, with Class I devices being the lowest risk class. Devices that pose the highest risk to users or patients and require the most regulatory oversight (pacemakers, defibrillators, etc.) are Class III devices, and devices that pose a moderate risk, such as syringes and catheters, are Class II devices. While this is a logical way to categorize medical devices, not every country uses this model.
Medical device regulations will likely never be universally uniform. Still, there is a concerted effort on behalf of regulatory experts from some of the largest medical device markets in the world to harmonize regulations and regulatory best practices. The International Medical Device Regulatory Forum (IMDRF) is a body of medical device regulatory specialists from around the world working together to standardize regulations and “accelerate international medical device regulatory harmonization and convergence.” While the IMDRF isn’t a regulatory body, its guidelines are often adopted by its member countries and adapted to fit their regulations and initiatives. In this article, we’ll give you an overview of the IMDRF, including its history and how its work impacts the global medical device industry.
The IMDRF’s beginning
The IMDRF was conceived in 2011 when Australia, Brazil, Canada, China, the EU, Japan, the US, and the World Health Organization (WHO) met to discuss the formation of this forum. One of their main goals was to build on and expedite the work of the Global Harmonization Task Force (GHTF), the organization that laid the foundation for the IMDRF.
The idea for the GHTF came from a meeting between representatives from the US, EU, Canada, and Japan in 1992. They met to discuss the possibility of forming a “consultative body” of regulatory specialists with the goal of “harmonizing medical device regulatory practices” worldwide. The GHTF held its first meeting in January 1993, forming study groups to evaluate different aspects of regulatory processes, including each member nation's quality management and Good Manufacturing Practices (GMPs). Today, the IMDRF continues to build on the strong foundation laid by the GHTF.
IMDRF members
As mentioned, the IMDRF works to harmonize medical device regulatory best practices worldwide. This mission requires the cooperation of regulators from many countries, performing in-depth studies and publishing guidelines that shape the global regulatory landscape. There are 11 members and their regulatory authorities that comprise the IMDRF:
- Australia - Therapeutic Goods Administration (TGA)
- Brazil - Brazilian Health Regulatory Agency (ANVISA)
- Canada - Health Canada
- China - National Medical Products Administration (NMPA)
- The European Union - European Commission (EC)
- Japan - Pharmaceutical and Medical Device Agency (PMDA)
- Russia - Russian Ministry of Health
- Singapore - Health Sciences Authority (HSA)
- South Korea - Ministry of Food and Drug Safety (MFDS)
- The United Kingdom - Medicines and Healthcare products Regulatory Agency (MHRA)
- The United States of America - US Food and Drug Administration (FDA)
IMDRF functions
The IMDRF is an international medical device regulatory body that offers guidance rather than implementing binding regulations, i.e., they don’t have the authority to mandate directives. According to the FDA, they “develop internationally agreed upon documents related to a wide variety of topics affecting medical devices.” Its members develop these documents by conducting studies via working groups, which we discuss in the next section.
IMDRF working groups
The IMDRF working groups are like subcommittees that focus on specific regulatory issues pertaining to medical devices. They address new technology, trends, and areas where harmonization of standards and regulations would be most beneficial. Their working groups conduct studies and research with the aim of providing regulatory best practices and universally applicable guidelines. There are currently seven active working groups:
- Adverse Event Terminology
- Artificial Intelligence Medical Devices
- Good Regulatory Review Practices
- Medical Device Cybersecurity Guide
- Personalized Medical Devices (PMD)
- Regulated Product Submission
- Software as a Medical Device (SaMD)
There are various participants in every active working group, and these groups can also include regulatory bodies that aren’t IMDRF members. Through their sharing and collection of data, working groups gain valuable insights that enable them to create guidelines that IMDRF members can relatively and easily implement around their existing medical device regulations. This helps to ensure the safety and efficacy of medical devices while promoting the harmonization of regulatory best practices and standards. These documents serve to accelerate the convergence of medical device regulations around the globe and have produced 69 guidances to date.
The IMDRF might not be a regulatory body or have any binding authority, but they are well respected within the medical device community. Their guidance documents help to shape regulatory standards in the world’s largest medical device markets. Staying abreast of IMDRF developments is a regulatory intelligence best practice (much like having a holistic regulatory information management system that provides structure for the medical device industry). Shameless plug aside, understanding the current undertakings of IMDRF working groups is a great way to better understand what’s going on in the medical device regulatory world and what changes might be coming down the road.
To learn more about the IMDRF, visit their website at imdrf.org.







