

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

Post-market surveillance for medical devices in the European Union
This article is an excerpt from Post-market surveillance for medical device in the European Union.
Table of Contents
- What is post-market surveillance?
- What classes of medical devices require post-market surveillance?
- Components of a successful post-market surveillance plan
- PMS data requirements
- Post-market surveillance system goals
- Required post-market surveillance reporting
- Embracing post-market surveillance as an integral part of your quality program
- Getting started with post-market surveillance
Post-market surveillance (PMS) is designed to monitor the performance of a marketed medical device by collecting and analyzing field use data. Article 10 of the EU MDR and IVDR requires all device manufacturers to have a post-market surveillance system in place. The main elements of the PMS are laid out in Article 83, and additional details for lower-risk and higher-risk devices are covered in articles 84 and85, respectively.
In general, a PMS system consists of both proactive activities and reactive, or vigilance, activities. While post-market surveillance and vigilance are sometimes used interchangeably, vigilance consists of separate activities that feed post-market surveillance programs.
Post-market surveillance systems are used to collect and analyze data not only about the manufacturer’s device but also about related competitors’ devices that are on the market. Data collected through PMS procedures is then used to identify trends that may lead to, among other things, quality improvements, updates to user training and instructions for use, and identification of manufacturing issues.
Note that “market surveillance” encompasses activities performed by a Competent Authority to verify MDR compliance, and should not be confused with the topic of this ebook,“post-market surveillance," which is performed by the manufacturer.
All medical devices marketed in the EU require some level of post-market surveillance, and all medical device manufacturers must implement a post-market surveillance system (PMS). The requirements of the PMS, however, vary and should be “proportionate to the risk class and appropriate for the type of device” (MDR Chapter VII). In particular, the type and frequency of reporting vary based on a device’s risk class.
A post-market surveillance plan (PMS) is an integral part of a manufacturer’s quality management system and provides a system for compiling and analyzing data that is relevant to product quality, performance, and safety throughout the entire lifetime of a device. The PMS should also provide methods for determining the need for and implementing any preventative and corrective actions. A PMS system should include and define:
Surveillance data sources
With the increased focus on proactive risk identification in the MDR, it is important to design post-market surveillance systems that actively acquire knowledge and detect potential risks. It is not sufficient to rely solely on spontaneous reporting by healthcare providers, patients, and other stakeholders.
In addition to information coming from Clinical Evaluation Reports and complaint and adverse event reporting, typical sources of surveillance data include:
• Social media networks: Because many of your stakeholders may be communicating on social media networks, it is important to employ social listening techniques and/or tools to identify issues and concerning trends as they develop.
• Industry and academic literature: Any studies, academic papers, and other literature that addresses similar devices or the specific use cases for which your device is designed should be evaluated. In particular, risk factors and adverse events identified with similar devices should be closely examined. It is also important to identify newer technologies that may affect the benefit-risk ratio and establish a new definition of “state of the art” for the device type.
• EUDAMED: While the European Database on Medical Devices (EUDAMED) is not yet fully functional, it is intended to provide a living picture of the lifecycle of all medical devices marketed in the EU. Manufacturers should take special care to consider information for similar devices made available through the EUDAMED system in the future.
• Registries: Patient, disease, and device registries can provide information that informs the clinical evaluation process which provides input into the post-market surveillance system.
Data analysis methodology
A well-defined data analysis methodology will accurately identify trends and lead to defendable decisions in the application of post-market experience. Once the necessary information has been identified and collected, and potentially cleaned of incomplete or otherwise unusable data, the data needs to be analyzed.
The goal is to identify meaningful trends, correlations, variations, and patterns that can lead to improvements in the safety and efficacy of the device. There are many data analysis tools available that can assist with:
• Regression analysis that will identify correlations between data (e.g. the device location/geography correlates to battery life).
• Data visualization that can be useful in spotting trends in the data.
• Predictive analytics, which can be particularly useful with large data sets, to identify future trends based on historical data.
• Data mining, which is also normally used with large datasets, to organize data and identify data groups for further analysis.
Benefit-risk indicators and thresholds
The MDR requires that medical device manufacturers not only demonstrate the clinical benefit of their device but also quantify the benefit-risk ratio. The benefit of a device must be shown to clearly outweigh the risk for it to gain market approval. Article 2 (24) of the MDR defines the benefit-risk determination as “the analysis of all assessments of benefit and risk of possible relevance for the use of the device for the intended purpose when used in accordance with the intended purpose given by the manufacturer.”
A PMS system should clearly define benefit-risk calculations and the data used to support them. Post-market surveillance activities are critical in order to re-evaluate and maintain the benefit-risk calculations and determinations of a device throughout its life. Information that is gained through a PMS system can lead to:
• Identification of new risk factors.
• Adjustments to risk frequency and/or severity values based on actual use data.
• Adjustments to established risk calculations based on new “state of the art” technologies becoming available.
• Adjustments to established benefit calculations based on actual use data.
While complaint handling and other feedback tracking are more often described as part of post-market vigilance systems, they play a role in the more proactive post-market surveillance processes as well. A PMS system should define ...
To continue reading this ebook, download the full version.

An overview of 21 CFR Part 820 - quality systems for medical device manufacturers
What is 21 CFR Part 820?
21 CFR 820 is the FDA federal regulation that pertains to quality systems for medical device manufacturers, and it is part of the agency’s set of Current Good Manufacturing Practices (CGMP) for industry. Also referred to as the FDA’s quality system regulation (QSR), the regulation defines design controls and quality processes at all stages of device development in order to ensure that all medical devices marketed in the United States are safe and effective.
21 CFR 820 consists of 15 subparts, which define quality system requirements for each stage and function within the medical device manufacturing process. We define each subpart below.
Federal regulations are organized as Title → Chapter → Subchapter → Part, which means that 21 CFR 820 is short-hand for:

21 CFR 820 vs ISO 13485
ISO 13485 is the de facto international quality system standard for medical device manufacturers, but this is not currently the standard in the United States. While Part 820 and ISO 13485 are structured differently, they have no conflicting requirements. Therefore, companies that are marketing medical devices in the U.S. and in other markets will need to comply with both ISO 13485 and the FDA’s QSR, as defined in 21 CFR 820.
However, the FDA is moving towards harmonizing these standards, and on February 23, 2022 issued a proposed rule to amend the QSR to align more closely with the international consensus standard for Quality Management Systems, primarily by incorporating reference to the ISO 13485 standard. The FDA has published FAQ’s about the proposed rule.
21 CFR Part 820 Requirements
Part 820: General Controls (subpart A)
The General Controls subpart contains three sections providing general information about the regulation, including the scope and applicability along with key definitions.
Scope
The regulation defines current good manufacturing practice (CGMP) requirements governing the methods, facilities, and controls used for the “design, manufacture, packaging, labeling, storage, installation, and servicing of all finished devices intended for human use." Specifically, this subpart defines:
- Applicability:
The requirements of this regulation are intended to ensure the safety and efficacy of all finished medical devices intended for human use that are manufactured in or imported into the United States. Manufacturers that are involved in some, but not all, manufacturing operations should comply with those requirements that are applicable to the functions they are performing.
Exceptions:
- This regulation does not apply to manufacturers of medical device components, but such manufacturers are encouraged to use this regulation as guidance.
- Class I medical devices are exempt from the Design Controls defined in this regulation, except for those listed in § 820.30(a)(2).
- Manufacturers of blood and blood components are not subject to this regulation but are subject to Biologics good manufacturing practices as defined in Subchapter F, Part 606 of the regulation.
Definitions
This section of the regulation contains definitions for a number of terms used throughout the document. The following are the major definitions related to quality records:
- Design history file (DHF): A compilation of records that describes the design history of a finished device.
- Design input: The physical and performance requirements of a device that are used as a basis for device design.
- Design output: The results of a design effort at each design phase and at the end of the total design effort. The finished design output is the basis for the device master record. The total finished design output consists of the device, its packaging and labeling, and the device master record.
- Device history record (DHR): A compilation of records containing the production history of a finished device.
- Device master record (DMR): A compilation of records containing the procedures and specifications for a finished device.
Quality System
The section of the regulation sets the basic requirement for a quality system by stating that “Each manufacturer shall establish and maintain a quality system that is appropriate for the specific medical device(s) designed or manufactured, and that meets the requirements of this part.”
The term “appropriate” is used throughout this regulation and can be open to interpretation. A manufacturer, however, should assume that all requirements are appropriate and applicable except in cases where non-implementation of the requirement can be shown to have no effect on the product's specified requirements or ability to carry out necessary corrective actions.
Quality system requirements (subpart B)
This section of the regulation defines the overall responsibilities and the resources required for the management of the quality system.
Management responsibilities
Executive management is responsible for establishing a quality policy and ensuring adequate resources to effectively maintain and manage the quality system. In addition, management is responsible for establishing a specific quality plan, consisting of relevant practices, resources, activities, and procedures.
Quality audit
Periodic audits of the quality system are required to be conducted by personnel not directly responsible for the activities being audited. The dates and results of each audit need to be documented, along with the results of the audit. It is expected that corrective actions and, when necessary, reaudits, be performed for any identified noncompliances.
Personnel
Manufacturers are responsible for assigning sufficient personnel with appropriate experience and training to perform all tasks required by the quality system plan.
Design controls (subpart C)
Manufacturers of all class II and class III medical devices, along with the specific class I devices listed in paragraph (a)(2) of this regulation, are required to establish design control procedures that ensure design requirements are met as specified.
Design controls shall define:
- Design and development planning - Plans that describe the design and development activities, and responsibilities for these activities and their implementation.
- Design input - Procedures that ensure design requirements are appropriate and address the intended use of the device.
- Design output - Procedures that document design output, including acceptance criteria, so that conformance to design input requirements can be adequately evaluated.
- Design review - Formal and documented reviews of the ensign results that include participation from representatives of all.
- Design verification - Procedures for verifying the device design that confirm that the design output meets the design input requirements.
- Design validation - Procedures for validating the device design, ensuring that devices conform to defined user needs and intended uses, and including testing of production units under actual or simulated conditions.
- Design transfer - Procedures to ensure that the device design is correctly translated into production specification.
- Design changes - Procedures for identifying, documenting, validating, and managing the verification and approval process of all design changes before they are implemented.
- Design history file - A design history file (DHF) is required for each type of device and should include or reference the records necessary to demonstrate that the design was developed in accordance with the approved design plan and device requirements.
Document controls (subpart D)
Medical device manufacturers are required to put in place document controls for all documents required in this regulation.
Document approval and distribution
One or more people must be assigned to review and approve documents prior to issuance. The approval must be documented, include a date and the signature of the approver, and be made available at all locations where applicable. Procedures must also be in place to ensure that obsolete documents are removed and/or prevented from being used.
Document changes
Similar to document approval procedures, changes to documents must be approved, reviewed, and documented. Records of all changes must be maintained.
Purchasing controls (subpart E)
To continue reading this Regulatory Brief, including a definition of the remaining subparts and a comparison of 21 CFR 820 to ISO 13485, please download the full brief.

The ultimate guide to the China NMPA UDI system and database
This article is an excerpt from The ultimate guide to the China NMPA UDI system and database ebook.
Table of Contents
- Overview
- UDI basics and benefits
- UDI format requirements and issuing entities
- UDI database and submission requirements
- Implementation of UDI and the UDI database in China
The current Chinese medical device regulatory regime kicked-off in 2014 with the Regulation on Supervision and Administration of Medical Devices. This core set of registration requirements, modeled after the United States and European Union systems, established a set of device classifications (class I, II, and III) based on risk and procedures for obtaining market clearance for each type of device.
Medical devices in China are regulated by the National Medical Products Administration (NMPA). Class I devices, such as clinical laboratory equipment or non-invasive skin dressings, require only notification to the NMPA for marketing authorization, and that authorization does not expire. Class II and III devices such as implantable devices or devices with a measuring function require full registration and a formal review before market clearance can be obtained.
These initial regulations have been expanded since their introduction, adding accelerated pathways to market for certain products in certain regions, easing acceptance of clinical data from overseas, and more specific roles and responsibilities for local agents of international manufacturers. In addition, in 2019, the regulations added a provision that medical devices carry a unique device identification (UDI). China’s UDI requirements are similar to those in the US and European Union. They establish specific device ID and labeling requirements, as well as a central, state-administered database of devices.
This eBook walks through the basics of medical device UDIs, the specifics of China’s implementation, and how MedTech companies who market their devices in China can prepare for the full rollout of these regulations in the coming years.
A UDI is a unique alphanumeric code that is designed to identify medical devices sold in a particular country/region from manufacturing, through distribution, to use by a patient. Like other aspects of the medical device regulatory regime, the UDI system in China follows the approach taken by the United States FDA and European Commission, and is based on the guidance from the International Medical Device Regulators Forum (IMDRF). Generally, UDI systems are designed to improve patient safety and optimize care by:
- Increasing the traceability of medical devices, including field safety corrective actions
- Providing an unambiguous identification method for medical devices throughout distribution and use
- Making adverse event reports more accessible
- Reducing medical errors by providing detailed information related to the device
- Simplifying medical device documentation and making it more consistent
There are three components to the UDI system in China:
- UDI code: The actual UDI code can be assigned by one of three (3) issuing agencies and contains information about the product, it’s expiration date, and the manufacturing batch/lot it’s associated with.
- UDI labeling: Put simply, medical devices must carry the UDI code on them. The regulations stipulate how devices and their packaging must be labeled for compliance.
- UDI database: In addition to labeling, all device UDIs must be submitted to a central database that is administered by the NMPA.
The following sections explore each of these components in more detail.
The UDI code
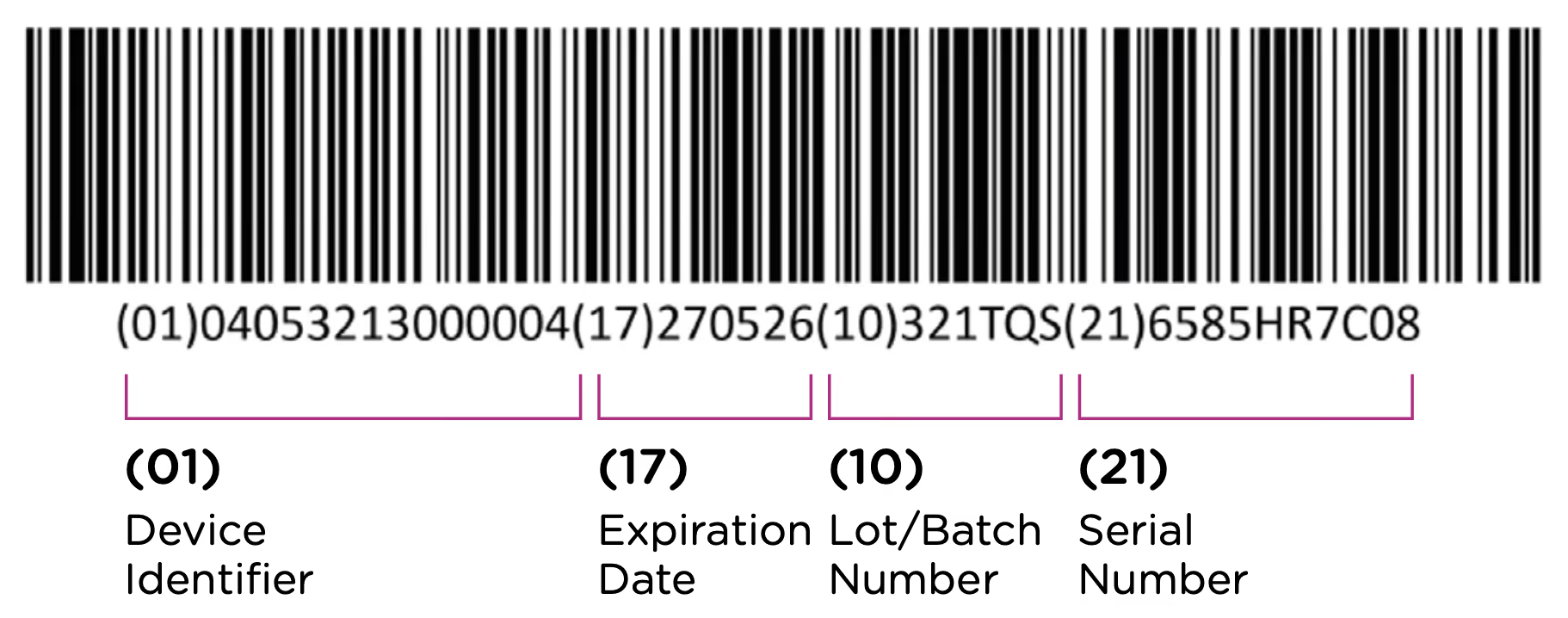
The first element of the UDI system is the code itself. The UDI code is the alphanumeric identifier that is associated with a specific medical device. UDI codes have two (2) elements to them, the UDI device identifier (UDI-DI) or static portion, and the UDI production identifier (UDI-PI) or dynamic portion. You can see the two components in the UDI diagram below:

The UDI-DI contains information about the issuing entity—the organization that is authorized to assign UDI codes. In China, this can be one of three entities: GS1, an international barcode and electronic data interchange standards organization, and two domestic organizations: the Zhongguancun Industry & Information Research Institute (ZIIOT), and AliHealth. Additional details about the issuing agencies are covered in Chapter 2. In addition, the UDI-DI contains information about the manufacturer and the specific model or version of the device.
The UDI-PI contains information about the manufacturing and production of the device. This typically includes information about the lot or batch number in which the device was manufactured, the manufacturing date and expiration date for the device (if applicable), and the specific serial number for the device. Here you can see all of the components marked up using the same UDI example:
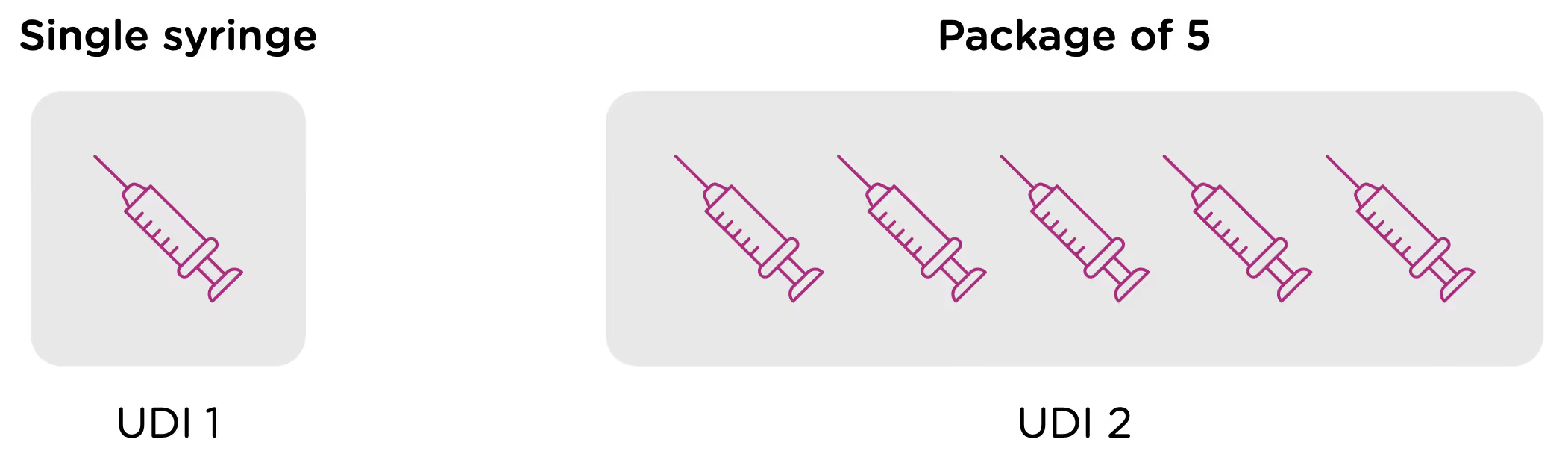
Note that each packaging permutation and level for a given device will need to be assigned its own UDI. So for example, let’s say that a company manufactures 5ml enteral (oral) syringes in two packaging options: 1 – packaged individually and 2 – packaged in a box of 5. Each packaging option would need its own UDI, despite the fact that the underlying product is the same.
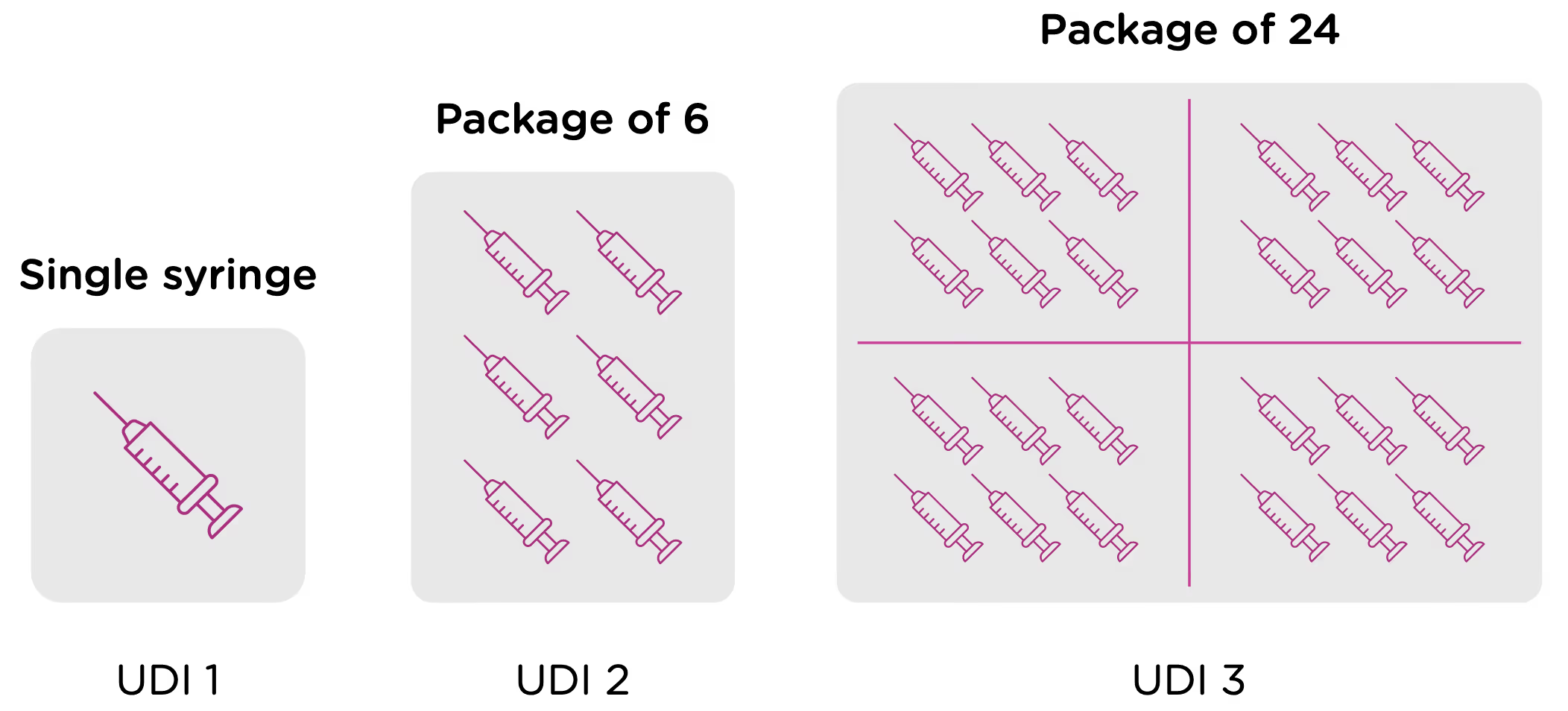
Now looking at packaging levels, let’s assume that the manufacturer packages the single syringe offering into boxes of 6, and again into larger containers of 24. Each of those packaging options needs its own UDI as well.
Labeling
In addition to obtaining UDI code for each device as outlined in the previous section, medical device manufacturers are required to ensure that devices are appropriately labeled with the assigned UDI. This label is called the UDI Carrier. The UDI is represented in two forms on the UDI Carrier: a machine-readable form and a human-readable form.
The machine-readable form or automatic identification data capture (AIDC) is a barcode or some other technology that can be used to automatically capture UDI information. The NMPA regulations support 3 types of machine-readable formats: 1-dimensional barcode, 2-dimensional barcode, and radio-frequency identification (RFID).

The regulations note that “use of advanced automatic identification and data collection technologies is encouraged”—prompting manufacturers to use more modern 2D and RFID machine-readable carriers where possible. Note, however, that if a device uses RFID, the UDI Carrier must also include the UDI in barcode format.
The human-readable form or human-readable interpretation (HRI) is the numeric or alphanumeric code for the UDI that can be read and manually entered into systems.

The UDI Carrier should be included on the device and on all levels of packaging. The UDI Carrier must be clear and readable during the operation and use of devices. If there isn’t room on the device for both the human and machine-readable forms of the UDI, then manufacturers should prioritize the machine-readable form.
UDI database
The third component of the NMPA UDI system is the UDI database. This is a centralized database of UDI and product information, administered by the NMPA. Manufacturers are required to submit UDI information into the database within 60 days after a product is approved (for sale in China) and before it is commercialized. The database contains a more detailed product record than what is included in the UDI itself, and it is the responsibility of the manufacturer (and/or their in-country representative) to submit the information correctly, and ensure that it’s kept up to date.
Chapter 3 of this eBook goes into detail about the specific fields and data requirements for UDI database submissions.
To continue reading this eBook including information about UDI format requirements and issuing entities, implementation timelines, and affected device types, please register to download the full version.

The ultimate guide to the EU MDR/IVDR UDI
This article is an excerpt from The ultimate guide to the EU MDR/IVDR UDI ebook.
Table of contents
- Overview
- UDI basics and benefits
- UDI format requirements and issuing entities
- UDI rules for specific device types
- Implementation of UDI and UDAMED in the European Union
- US vs EU UDI comparison
The EU Medical Device Regulation (2017/745) (“MDR”) and EU In Vitro Diagnosis Regulation (2017/746) (“IVDR”) introduce two new systems for information exchange: UDI (Unique Device Identifier) for device identification and EUDAMED (European Databank on Medical Devices) to centralize and disseminate information. UDI is a specific code assigned to all devices and higher levels of packaging. This will allow for devices being sold in the European market to be identified and traced through a globally harmonized approach. EUDAMED is the IT system developed by the European Commission to replace the EUDAMED2 database previously in place under the Medical Device Directives (MDD). EUDAMED is a multi-functional system that will be used to coordinate device registration, provide information about devices to industry professionals and the public, and highlight necessary safety details.
The EU MDR and IVDR UDI system is based upon the guidance of the International Medical Device Regulators Forum (IMDRF). It’s a globally harmonized system that’s designed to increase patient safety and optimize care.
UDI system goals
Increase patient safety
- Improve tracing of devices
- Reduce the presence of counterfeit devices
Ensure access to accurate information
- Unambiguous identification of devices throughout distribution and use
Improve post-market surveillance
- Improve accessibility of adverse event reports
Enhance supply chain Management
- Streamline supply chain process and inventory management
- Simplify medical device documentation processes
The UDI system has four key elements
Element 1: Assignment of UDI (UDI Components)
The first element of the UDI system is the assignment of a UDI. The UDI is a code of alphanumeric characters that acts as the access key to information about a specific medical device on the market. The EU MDR and EU IVDR requires that a UDI be assigned to all medical devices except for custom-made or investigational devices. There are three components of a UDI:
- Basic UDI-DI
- UDI (consisting of UDI-DI and UDI-PI)
- Packaging UDI (Note: This is not an official term used in the EU MDR and IVDR, but we’re using it to help explain the concept. The Packing UDI is part of the UDI itself.)
1. Basic UDI-DI
The Basic UDI-DI identifies the device group that a particular device fits into. A device group is a group of products that all share the same intended purpose, risk class, essential design, and manufacturing characteristics. A device group is generally classified by medical device manufacturers as a “Product Family” or “Product Category,” depending on the internal nomenclature used within the company. The Basic UDI-DI functions as a parent or higher-level descriptor of a device.
NOTE: There can only be one Basic UDI-DI per UDI-DI.
The Basic UDI-DI is not printed on the product itself or on the packaging of a product, but rather it must be included in the following documents and applications:
- Certificates (Including Certificate of Free Sale)
- EU Declarations of Conformity
- Techical Documentation
- Summary of Safety and Clinical Performance
2. UDI (UDI-DI and UDI-PI)
The second component is the UDI itself, which consists of two parts:
Device Identifier (DI)
Production Identifier (PI)
The UDI-DI (Device Identifier DI, also referred to as “static”) identifies specific, detailed information about a particular device. If any of the below details should change, the device will need a new UDI-DI.
- Name or trade name of the device
- Device version or model
- If labelled as a single use device
- Packaged as sterile
- Maximum number of uses
- Need for sterilization before use
- Quantity of devices provided in a package
- Critical warnings or contra-indication
- CMR/endocrine disruptors
NOTE: There can be several UDI-DIs for one Basic UDI-DI.
Meanwhile, the UDI-PI (Production Identifier PI, also referred to as "dynamic") contains manufacturing information (including serial number, lot/batch number, software identification, and manufacturing or expiry date or both types of dates.)
To better illustrate this concept of Basic UDI-DI and UDI (UDI-DI and UDI-PI), let’s use a syringe as an example. The Basic UDI-DI would identify the category of a syringe, for example, "Enteral (Oral) Syringe."
A 5ml Enteral (Oral) Syringe – Sterile (Color: Purple) would get a unique UDI-DI and a 10m Enteral (Oral) Syringe – Sterile (Color: Orange) would get a unique UDI-DI. Both products would be associated to the same Basic UDI-DI. In this case, the "Enteral (Oral) Syringe," which defines the category.
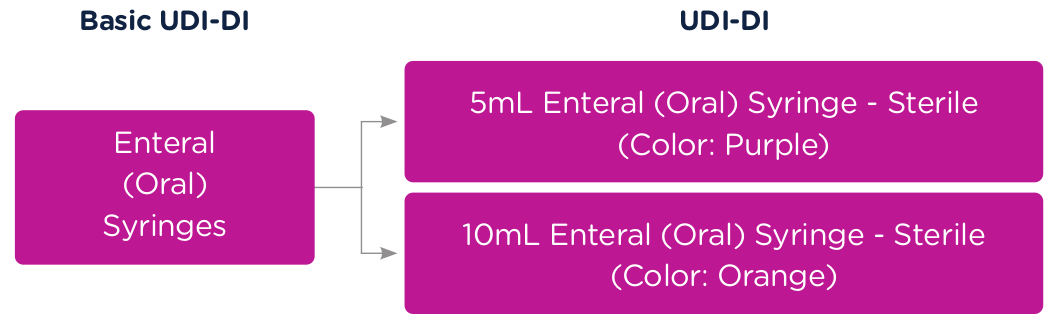
Each time that 5ml Enteral (Oral) Syringe – Sterile (Color: Purple) is manufactured at the same revision, it will get a new UDI-PI per lot. See the graphic below.

Each product is identical and therefore has the same UDI-DI. However, the UDI-PI changes to reflect the manufacturing date, lot number, expiry date, and serial number, as applicable.
The UDI will contain all device-specific information and have the same functions as the comparable database (GUDID) of the United States FDA. The main difference (in EUDAMED) is that the UDI data is divided into components of Basic UDI-DI, UDI, and Packaging UDI.
3. Packaging UDI
The third component of UDI is the Packaging UDI. (Note: This is not an official term used in the EU MDR and IVDR, but we’re using it to help explain the concept.)
Each level of packaging, except shipping containers, must receive its own unique UDI. Packaging UDI refers to the unique UDI assigned to higher levels of packaging instead of the device itself.
In the event of significant space constraints on the unit of use packaging, the UDI Carrier may be placed on the next higher packaging level.
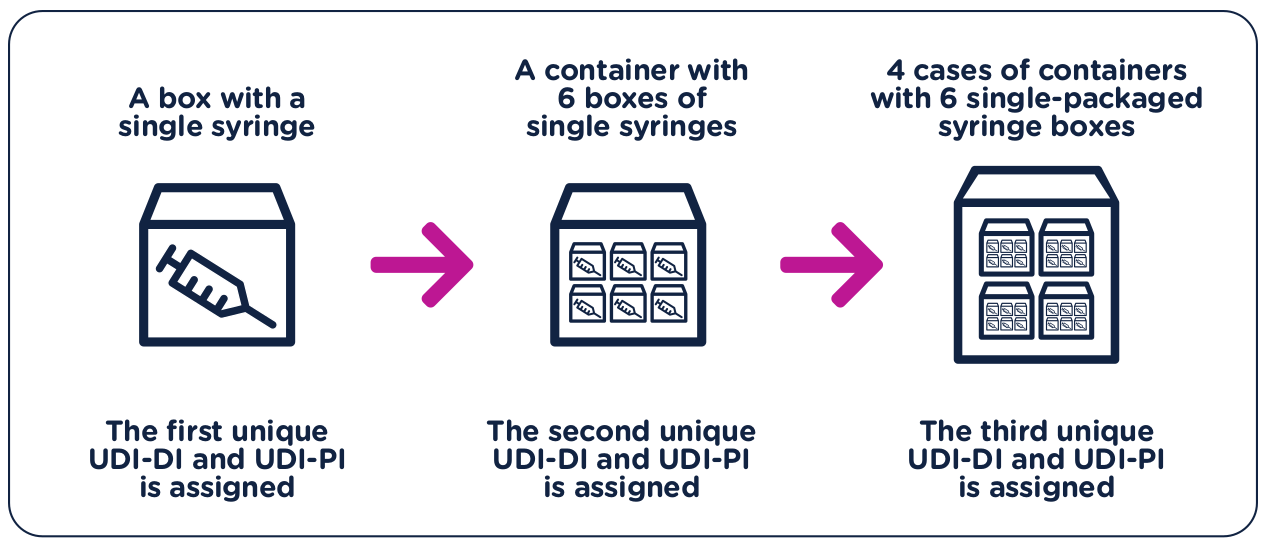
Returning to our earlier example of syringes, if a manufacturer first packages a single sellable syringe into an individual box, this package would receive its own UDI-DI and UDI-PI.
If then the manufacturer packages those individual boxes into containers of six (6), those containers would receive their own UDI-DI and UDI-PI.
And finally, if the manufacturer packages those six (6) containers into cases of four (4), those cases would receive their own UDI-DI and UDI-PI.
Each of those levels of packaging must be assigned its own UDI-DI and UDI-PI. The initial syringe did not change, but the way it is packaged did, therefore, requiring its own UDI-DI and UDI-PI.
Element 2: Placing UDI on the device and/or packaging
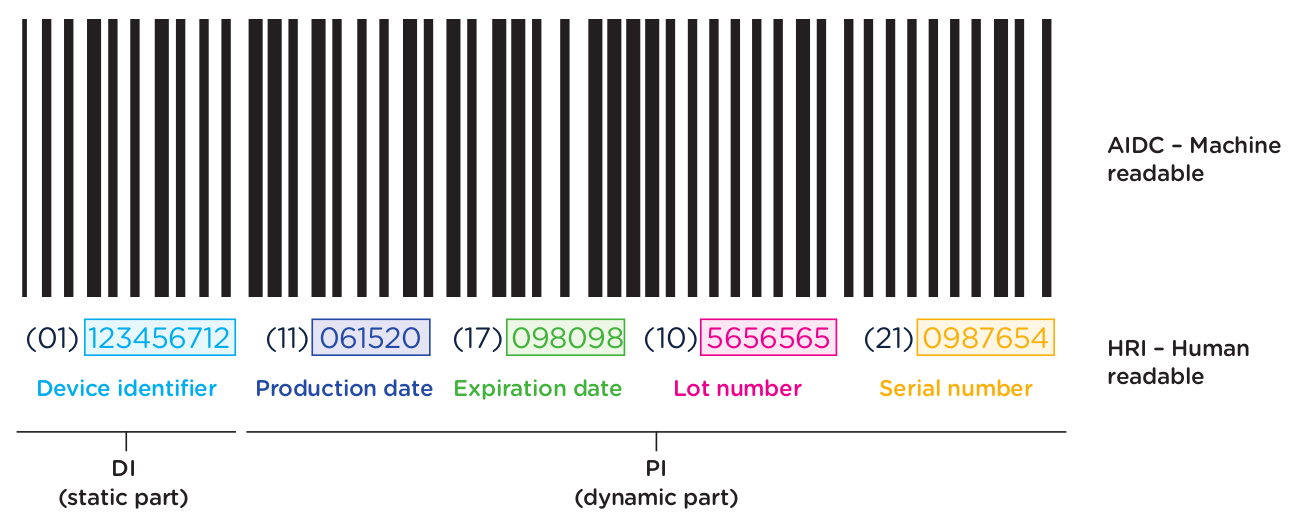
The second element to the UDI system is the placing of the UDI on the device or on its packaging through what is referred to as a “UDI Carrier.” The UDI Carrier is the part of the label that contains the UDI information that is applied directly to the device or included on the device packaging. The UDI Carrier should have both a machine-readable portion (AIDC) and a human-readable portion (HRI). (Specific details about each element of the UDI will be covered in Chapter 2.)
- Machine-readable form – AIDC – (Automatic Identification and Data Capture) is a barcode or other machine-readable technology that can be accessed automatically by scanning the UDI information.
- Human-readable form – HRI – (Human Readable Interpretation) is the numeric or alphanumeric code, which can be manually entered into the system for access to the UDI information.
If there are space constraints limiting the use of both the AIDC and HRI on the label, then only the AIDC is required to appear. However, on devices that are intended to be used in home-health care or other non-medical facility settings, the HRI would be required to appear.
Single-use devices may contain the UDI Carrier on its lowest level of packaging rather than on the device itself.
Reusable devices must include the UDI Carrier on the device itself, unless any type of direct marking would interfere with the safety or performance of the device, or if it is not technologically feasible to directly mark the device. If so, this should be properly documented in your design history file.
Most importantly, the UDI Carrier must be readable for the intended lifecycle of the device.
Below is an example of a GS1 AIDC and HRI barcode label.
Element 3: Storage of UDI information by Economic Operators
Storage of UDI information by "Economic Operators" is the third element of the UDI system. 2017/745 Articles 2(35), 22(1), and 22(3) define an economic operator as:
- A manufacturer
- An authorized representative
- A distributor
- An importer
- An investigator for clinical investigations
- A person who sterilizes systems or procedure packs
Class III, implantable device:
According to EU MDR 2017/745 Annex II, the manufacturer shall keep an updated list of all UDIs that it has assigned. Economic operators and all health institutions are required to store, preferably by electronic means, the UDI of all the devices for which they have supplied or with which they have been supplied.
For Devices Other than Class III:
Member States are encouraged, and in some cases require, health institutions to store, preferably by electronic means, the UDI of the devices with which they have been supplied. The UDI must also be included in any field safety notice for reporting serious incidents and field safety corrective actions.
The EU MDR and EU IVDR also give the European Commission authority to make additional requirements regarding the submission or maintenance of UDI information. In making those decisions, the European Commission must consider six (6) areas:
- Confidentiality and data protection
- Risk-based approach
- Cost-effectiveness of the additional measures
- The need to avoid duplications in the UDI system
- The needs of the healthcare systems of the member states
- Harmonization with other medical device identification systems
To continue reading this eBook including information about the EUDAMED database, UDI format requirements and issuing entities, implementation timelines, and key differences between the EU and US UDI systems, please register to download the full version

Dispatches from RAPS Convergence: The state of regulatory tools
A few weeks ago we attended (virtually) the RAPS Euro Convergence conference. The event, despite the virtual format, still brought together regulatory professionals from across the European region for several days of immersive learning. At Rimsys, we took advantage of the opportunity to explore the state of regulatory tools and processes in the region, and see how they compare to those of North American teams.
Visitors to the Rimsys booth, both at RAPS Euro Convergence and at last year’s North American RAPS Convergence event were invited to fill out a short survey in exchange for the opportunity to win an Amazon gift card. While the respondents were self-selected, and the results aren’t statistically significant, they still showcase some interesting differences in the tools and mindset of regulatory affairs professionals in each region.
Regulatory tools used
Across both regions, Excel is the most commonly used tool by regulatory teams. Over half of respondents across both regions reported using Excel to manage regulatory information and processes. European regulatory affairs professionals were much more likely to use an electronic quality management system (eQMS) to manage their work, and about one-third of respondents to both surveys indicated that they used physical paper-based documents.
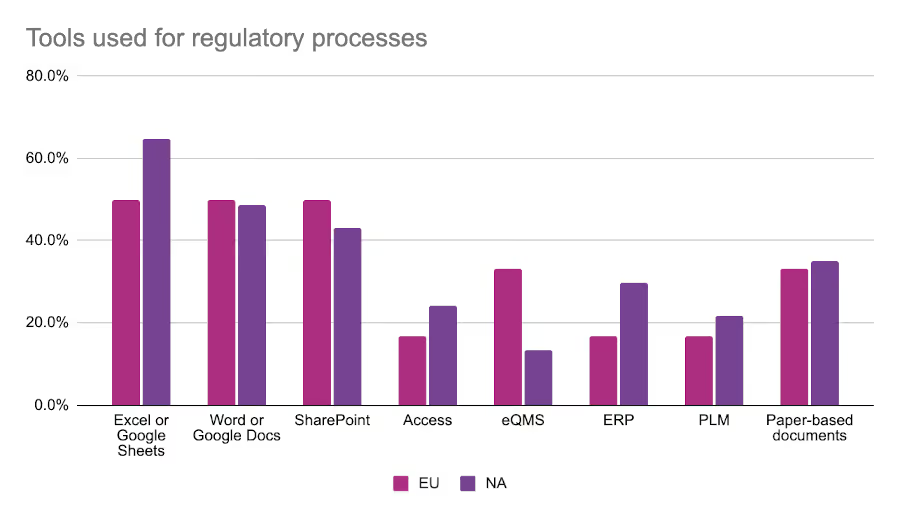
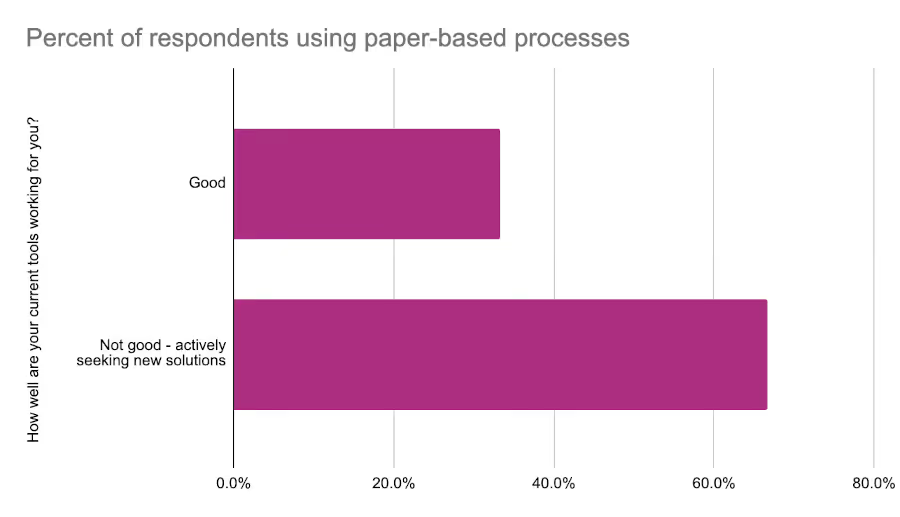
We also asked respondents how well their tools were working. European respondents were generally more content with their toolset with 66% saying their tools were “good”. By comparison only 22% of North American respondents felt the same. One thing that was clear was the impact of paper-based processes on satisfaction. Respondents who reported struggling with their current tools were nearly twice as likely to use paper-based processes as part of their regulatory activities.
Work efficiency and satisfaction
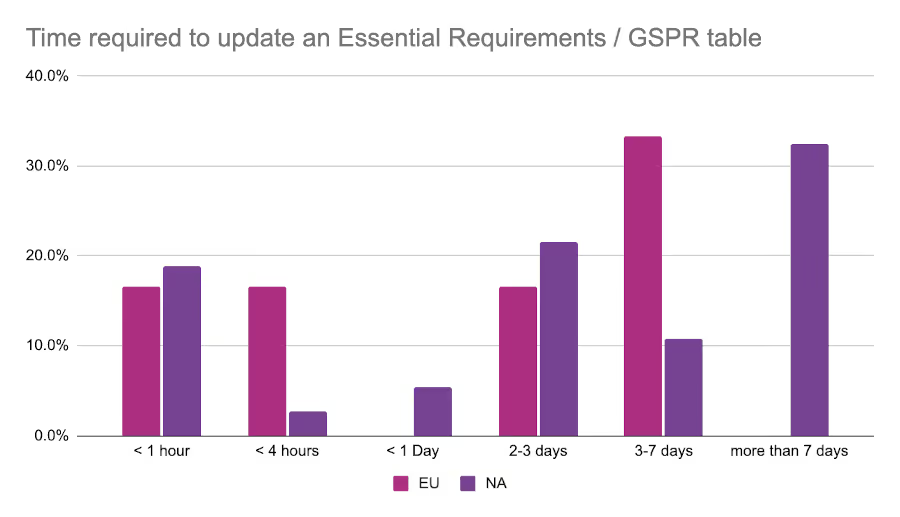
Just over 50% of all respondents indicated that the tools they use “could be better”. This may have something to do with the amount of manual work that RA teams find themselves doing. As part of the survey we asked respondents how many hours it takes on average for them to update one of their Essential Principles / General Safety and Performance Requirements (GSPR) tables. The most common response from the EU event was 3-7 days, while those at the North American event were most likely to report greater than 7 days for the same task.
Given their slightly better estimated performance when it comes to regulatory processes, respondents from the EU were less likely to express frustration with their roles and dissatisfaction with their productivity. We asked attendees at both conferences to rate their “regulatory frustration” and satisfaction with their productivity on a scale from 1 to 5 with 1 being very frustrated/unsatisfied, and 5 being very satisfied/not frustrated at all.
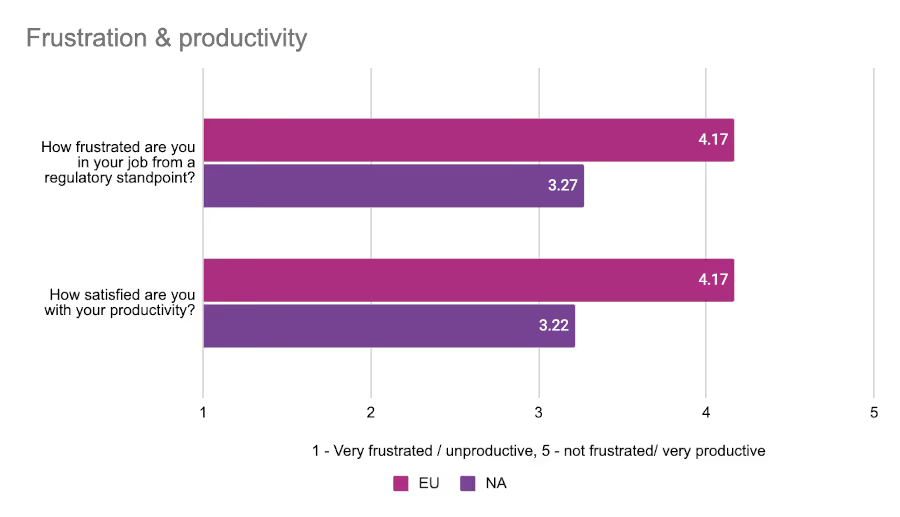
EU respondents were noticeably more positive in their assessments than our North American respondents, although everyone generally had a favorable view of their productivity.
Implications
Looking at the survey results, there are some interesting discrepancies. Teams in both regions heavily use poorly-suited tools and manual approaches to regulatory processes, yet have generally favorable views of their productivity. This points to two likely conclusions. One, that regulatory affairs professionals are particularly comfortable with a lot of manual administrative work, and two, that they’re largely unaware of the new class of regulatory information management (RIM) solutions that are specifically designed to automate and streamline regulatory workflows.
RIM platforms (like Rimsys) provide a digitized central repository for regulatory information and content, allowing RA professionals to streamline product submission, authorization, and maintenance processes (such as updating GSPR tables that we discussed earlier). RIM platforms can reduce the time and resources required to complete regulatory activities by more than 50%.

Building a business case for a RIM system
While the space is growing quickly, regulatory information management (RIM) systems are still relatively new to a lot of medtech companies. RIM systems help companies digitize and automate regulatory activities associated with their products. They provide a central information repository for all regulatory content and streamline activities like market registrations, data capture and transmission of unique device identification (UDI), and building essential principles tables while tracking associated standards.
Given that many regulatory affairs teams still manually manage these processes through complex spreadsheets and disjointed documents, the value of an automated solution is pretty obvious, but quantifying that value—especially for teams that aren’t seasoned software acquirers—can be a bit harder. This guide provides a framework for regulatory affairs teams to quantify the potential benefits of a RIM system, and build an internal business case for investment.
Challenges with the traditional approach to regulatory affairs
While functional, there are a number of painful inefficiencies that come along with traditional approaches to regulatory processes. Without an automated way to keep track of all the information and supporting documents associated with these processes, companies struggle with:
- Lack of visibility into regulatory data, clearance/approval status for different markets, and time-to-market metrics
- Compliance gaps driven by disconnects between go-to-market, distribution, and regulatory affairs teams
- Difficulty assessing the impact of, and responding to changes in standards or regulations
- Accumulation of “tribal” knowledge among individuals that limits continuity and visibility across the organization
These pains represent specific costs to MedTech companies in the form of:
- Staffing: Companies must over-staff regulatory affairs teams to support highly-manual processes. Expensive consultants are often brought in to help address in-house resource shortages.
- Productivity: Regulatory affairs teams lose huge amounts of time repeatedly hunting for information (up to 50% of their time spent).
- Lost revenue: Long application times and lack of process visibility delay market entry for new products. Non-compliance can lead to fines, or the need to pull products out of specific markets.
The automation and data consolidation/integration provided by a RIM system can significantly reduce these costs, and provide a clear, measurable return on investment.
Additional benefits of a RIM system
In addition to the addressing the pains outlined above, RIM systems can provide valuable benefits across MedTech companies:
- IT teams: Without a bespoke platform to manage regulatory processes, regulatory affairs teams rely on a broad collection of tools to support their day-to-day work. This can include specific software to create and manage UDIs and access regulatory intelligence, as well as use of software designed for other functions: enterprise resource planning (ERP), product lifecycle management (PLM), or quality management systems (QMS)—highly-configured to try and support regulatory activities. A comprehensive RIM system (like Rimsys) provides support for multiple regulatory functions, saving IT teams the cost of acquiring and maintaining separate systems. With functionality specifically designed for regulatory processes, a RIM system is easier to support than customizations to tools designed for other functions.
- Go-to-market teams: Sales and marketing organizations can also benefit from the adoption of a RIM system. In addition to bringing new solutions to market more quickly, RIM systems can also help with planning and forecasting. Visibility into the time and cost required to enter different markets, and the specific regulations associated with each market, can help go-to-market teams better prioritize target markets, and set revenue projections for their product lines. RIM systems can also provide workflows for project requests, allowing go-to-market teams to better coordinate registrations to support planned product launches.
- Distributors, in-country sponsors, and notified bodies: One-off email communications with external parties is not only time consuming for regulatory affairs teams, but inefficient for partners. RIM systems can provide controlled access for external parties, allowing them to login and directly access needed information without sending an email, and waiting (sometimes days) for a response. Streamlining these communications allows partner organizations to move more quickly, and ultimately accelerate the delivery of products into new markets. This helps to grow revenue while also improving the productivity of regulatory affairs teams who no longer have to interrupt their work for every internal or external information request.
Building your business case
Putting together the challenges and benefits outlined in the previous section (as applicable to your team), will feed the primary content of your business case. Next we’ll take a look at how to structure the content.
Part 1 – Your current situation and challenges
One of the best places to start when putting together a business case for a RIM system is to look at your current situation. What are the challenges that your organization faces? How much time is spent looking for information, submitting marketing applications, completing regulatory impact assessments, creating and maintaining the new MDR / IVDR GSPRs? How long does it take your team to complete new registrations? How well do internal teams communicate and coordinate go-to-market activities for new products and markets? How many products and countries does your team support today? What would happen if that number increased significantly?
Next, look at the business implications of the current situation. Does your team have a backlog of requests from go-to-market or other management teams? How often do you bring in external consultants to help with workload? Note the associated costs of your project backlog (delays in receiving market clearance), and the amount spent each year on consultants. Are there any other business risks such as continuity or non-compliance that are associated with your current approach? Does your team have the ability to identify regulatory roadblocks associated with markets you’re planning to enter?
Part 2 – Recommended solution
Here’s where you make the primary case for a RIM system. Highlight the specific capabilities and benefits of your solution of choice, and how it will address the challenges of your current situation. Highlight areas where other teams will benefit from the solution, and opportunities to drive greater organizational alignment.
Part 3 – Costs and expected ROI
Estimating costs and returns can be difficult, but it’s one of the key ways to improve the credibility of your business case. If you’re unsure about costs you can generally assume that a full-featured RIM system will cost the equivalent of 1 to 3 full-time regulatory affairs professionals, and use that as a starting point.
When calculating returns start with time-savings. If regulatory activities could be completed 50% more efficiently, how would that impact the number of consulting hours you use or hiring plans? If you could reduce pre-market clearance time by 6 weeks, how much additional revenue could be generated? Have you had instances where you incurred fines or had to remove products from a market due to compliance issues (i.e. shipping to markets where the product hasn’t been approved)? What were the associated costs?
Next, look at what your RIM system is replacing. Do you have individual tools for UDI or other functions that could be replaced? What about other tools? Does your team have seats in ERP or QMS tools that won’t be needed any more? All of these are relevant cost savings. In most cases, a RIM system will show strong ROI once you’ve estimated all of these savings.
Part 4 – Alternatives
Here’s where you want to demonstrate that you’ve done your due diligence in support of your recommendations. Have you evaluated multiple software solutions? Enumerate the specific capabilities or approach that led you to recommend your preferred vendor. Are there other ways to address your current challenges: expanding the regulatory affairs team, or implementing other types of software (rather than a RIM system)? Explain why you think these solutions will be less effective.
Putting it together
RIM systems can provide significant benefits specifically to regulatory affairs teams and broadly across MedTech companies. With a bit of legwork, it’s easy to put together a compelling case for investment in a solution. What can sometimes lead to difficulty in quantifying potential benefits—the fact that many MedTech companies don’t have a robust measurement framework for Regulatory affairs processes—means that you’ll be in a much better position to assess the performance of your team and impact of your investment once the analysis is complete.
Ready to get started? Download our RIM business case template. Questions? Our team is happy to work through the process with you and validate your estimates based on the experiences of our clients.

5 ways a RIM system can accelerate time-to-market for MedTech companies
Like all products, time-to-market is a critical success factor for medical technology (medtech). Product research and development ties up capital investment that can only be recouped when products start selling. For start-up and early stage companies, time-to-market can be the difference between success and failure. With a limited capital runway, they must demonstrate market success to access additional funding and growth opportunities. Unlike other products, however, medical devices have an added hurdle of regulatory clearance that must be obtained before products can be marketed and sold.
Underestimating the regulatory burden
It’s easy to underestimate the amount of time and effort required for regulatory activities related to New Product Introductions (NPI), especially during the development process. Policies, procedures, and submission processes can vary significantly between countries and regions, and almost all of them require government clearance before devices can be marketed. For larger companies it’s not unusual to operate in 100+ countries, creating an enormous array of standards and applications that must be manually managed.
These challenges are exacerbated by unclear lines of communication and siloed information across systems. Product development and testing information is stored within Product Lifecycle Management (PLM) and Quality Management System (QMS) solutions. Sales forecasting, marketing, and production information is stored within Enterprise Resource Planning (ERP) and Customer Relationship Management (CRM) systems. And regulatory, standards information, and compliance documents are stored across various file systems.
Regulatory submissions must synthesize information from R&D, QA, and go-to-market teams, and data from across all of these systems. Any mis-steps, or missing information can result in significant go-to-market delays, and even rejections from regulatory bodies. Coordinating regulatory status between go-to-market regulatory affairs teams can be a particularly vexing challenge. Without clarity about what markets have been cleared/approved, companies run the risk of moving too quickly ahead of the regulatory process, or unnecessarily delaying market entry.
What is a RIM system?
Despite all of the complexity associated with regulatory submissions, many teams rely on rudimentary approaches to coordinate and manage them. Submissions and associated data are managed via individual documents or complex color-coded spreadsheets, with no central repository of submission records or status.
Regulatory Information Management (RIM) systems are software solutions designed specifically to help companies streamline the submission, authorization, and maintenance process. They provide a digitized central repository for all regulatory information and content, allowing companies to automate and maintain compliance in the global market.
Using a RIM system to manage regulatory submissions and compliance can improve efficiency and productivity, reduce the risk of rejected submissions and noncompliance, and provide greater visibility into ongoing registration processes and status across the organization. While RIM systems provide specific benefits to regulatory affairs teams, they also provide company-wide benefits—specifically when it comes to accelerating time-to-market for new products.
5 ways a RIM system can accelerate time-to-market for new products
Full-featured RIM systems (like Rimsys) provide a number of capabilities that MedTech companies can take advantage of to get new products to market more quickly—and keep them there.
- Regulatory intelligence. Medical device registration requirements and standards vary across regions and countries. Understanding market entry requirements and timelines for regulatory submissions are necessary for any go-to-market planning, but finding and keeping track of this information can be challenging—especially for early-stage companies. RIM systems can provide up-to-date information about regulatory requirements without a lot of manual research and document management. Go-to-market teams can leverage the breadth of regulatory information in the system to develop rollout strategies for different markets, and identify the most attractive markets based on size and regulatory complexity.
- Digital forms and templates. Each regulatory submission requires multiple templates and forms, many of which aren’t available digitally. Finding the correct, current form, and manually filling it for each country/region can add significant time to the regulatory submission process. RIM systems can take the guesswork out of finding forms, by providing a library of digital templates for different markets that companies can easily access and fill. Even offline forms can be loaded into the system for digital filing and storage. With a modern RIM system, regulatory teams can easily access, fill, and track progress for submission forms in all the markets they’re looking to enter.
- Centralized visibility and information storage. RIM systems can function as a “digital hub” and single source of truth for all of the information associated with the regulatory process. Submissions require detailed product information, testing results, labeling, and other information that is often stored in other systems. Rather than sourcing this information over and over again, RIM systems provide regulatory teams with an organized repository that they can reuse across global applications. RIM access can be extended to other teams, and even external partners (like in-country distributors) to provide visibility into regulatory information and the status of submissions, making it easier to drive alignment around the process and coordinate go-to-market plans.
- Process integration across systems. Market authorization is one of the most critical pieces of information that go-to-market teams need. Maintaining that information across multiple products and multiple markets, and keeping it visible to go-to-market teams is a consistent challenge. RIM systems can integrate directly with ERP or CRM systems to feed authorization information directly into sales and distribution processes. Automating the in-country authorizations across systems can prevent noncompliance, and ensure that go-to-market teams are able to launch as soon as authorization is obtained. The same integration capabilities can be used to automatically retrieve product information from PLM and QMS systems, further speeding the application process.
- Automated regulation and standards tracking. This doesn’t necessarily directly impact time-to-market for new products, but it can definitely impact time in the market. Regulations and standards aren’t static, and regulatory affairs teams must keep on top of pending changes to ensure that products remain compliant and retain selling authorization. RIM systems can help to track changes, and flag products for potential compliance issues or that are at risk of losing authorization.In addition to regulatory changes, RIM systems can track authorization expirations, and other important events, helping companies maximize the revenue potential of their products by avoiding regulatory disruptions.
Accelerating regulatory approval and product go-to-market
MedTech companies are keenly aware of the role regulation plays in getting new products to market, but they aren’t always aware of the time and effort required to reach all of their target markets. Manual processes, disjointed information, and lack of coordination and visibility across teams can make it hard to obtain marketing authorization in a timely manner. This can significantly impact time-to-market—delaying return on investment, and even putting companies, themselves at risk.
RIM systems can eliminate a lot of the inefficiencies that slow down regulatory processes. By providing insight into regulatory requirements, access to digital templates, and integration across tools, they make it easier for companies to complete timely, successful regulatory submissions, and accelerate time-to-market.
RIM software from Rimsys
Rimsys is the only holistic RIM software designed specifically for medical technology companies. It helps companies digitize regulatory management by bringing together global UDI requirements, Essential Principles/GSPR, and regulatory registrations while monitoring products at the SKU level.
To learn more about RIM software from Rimsys, read our benefits datasheet.

Arena Solutions and Rimsys announce partnership to offer an end-to-end quality and product-centric regulatory solution
Foster City, Calif., January 12, 2021 – Arena Solutions, a leader providing cloud-based product development solutions for high tech, consumer electronics, and medical device industries, today announced a new partnership with Rimsys Inc., a world-leading provider of a holistic Regulatory Information Management (RIM) software platform designed specifically for medical technology (medtech) companies.
The Arena and Rimsys partnership offers a secure cloud-based, product-centric regulatory solution for the medtech industry. Rimsys seamlessly integrates with Arena’s QMS and PLM solutions by pulling product and documentation information directly into Rimsys to create, manage, and maintain marketing applications such as 510(k), Summary Technical Documentation (STED), and Table of Contents (ToCs).
The Arena product development platform connects product and quality processes allowing dispersed teams throughout the product design and manufacturing process to work together. Rimsys integrates with Arena’s platform by syncing product information so companies can better manage global registrations and selling status at the SKU level. Rimsys has the capability to pull in QMS records and documentation to create and compile regulatory applications from approved documentation. The integration automatically monitors for documentation changes and alerts users when updates occur with additional reporting based on document location ensuring a single and accurate source of truth.
"The seamless and deep integration between Rimsys and Arena solutions reduces the day-to-day regulatory management," said James Gianoutsos, Founder and President of Rimsys. "The administrative burden of compiling marketing applications and the maintenance of product data is completely eliminated, allowing for increased compliance, efficiency, and visibility throughout the organization."
"Our partnership with Rimsys makes it easier for MedTech companies to address regulatory affairs, product registration, and standards management more effectively," said George Lewis, VP of Business Development and Strategy for Arena Solutions. "This new integration streamlines regulatory compliance processes by accelerating the notification of updates to critical quality records and documents."
About Arena Solutions
Arena Solutions helps innovative electronic high tech and medical device companies create products that change the world. Arena unifies product lifecycle (PLM) and quality management (QMS) processes, allowing every participant throughout the product realization process from design to manufacturing to work together. With Arena, teams accelerate product development and delivery to increase profits. For more information, visit ArenaSolutions.com.
About Rimsys
Rimsys is a world-leading provider of Regulatory Information Management (RIM) software for medical technology companies. Built by and for regulatory affairs professionals, Rimsys digitizes, automates, and creates regulatory order to ensure products adhere to changing global regulations. It is the only holistic RIM software for medical devices, in-vitro diagnostics, and medical device software that makes it easy to manage global UDI requirements and navigate the pillars of regulatory affairs, including product registration, standards management, essential principles/GSPR, and regulatory intelligence. rimsys.io





