

Featured
Rimsys Announces Rimsys AI to Eliminate Repetitive Tasks and Enhance Decision-Making for MedTech Regulatory Teams
Rimsys, the leading Regulatory Information Management (RIM) platform for the MedTech industry, today announced the launch of Rimsys AI, a suite of embedded artificial intelligence (AI) agents.

The ultimate guide to the EU MDR and IVDR general safety and performance requirements (GSPR)
This article is an excerpt from The ultimate guide to the EU MDR and IVDR general safety and performance requirements (GSPR) ebook.
Table of contents
- Overview
- Terminology
- EU MDR/IVDR Annex I
- EU MDR/IVDR Annex II
- Proactive Monitoring & Maintenance
- Comparison Table: EU MDR/IVDR Annex I GSPRs vs EU MDD/IVDD Annex I Essential Principles
With the initial rollout of the European Medical Device Regulation (MDR) complete, medical device companies are shifting focus to the sister In Vitro Diagnostic Regulation (IVDR) which has rolling effective dates starting in May 2022. Like the MDR, the IVDR also includes new General Safety and Performance Requirements (GSPR). The expanded 2nd edition of this ebook includes a detailed summary of the IVDR GSPR regulations in addition to those of the MDR. It provides you with practical guidance on how to meet the GSPR requirements for all types of medical technology products. This ebook, however, should not take the place of reviewing the actual regulations and consulting regulatory experts when needed
Timeline
The EU MDR submission became mandatory from the previous MDD directive on May 26, 2021, and the EU IVDR effective date is quickly approaching. In fact, all submissions for new devices under the new EU IVDR must be implemented no later than May 25, 2022. Below is a high-level overview of key dates for both regulations.
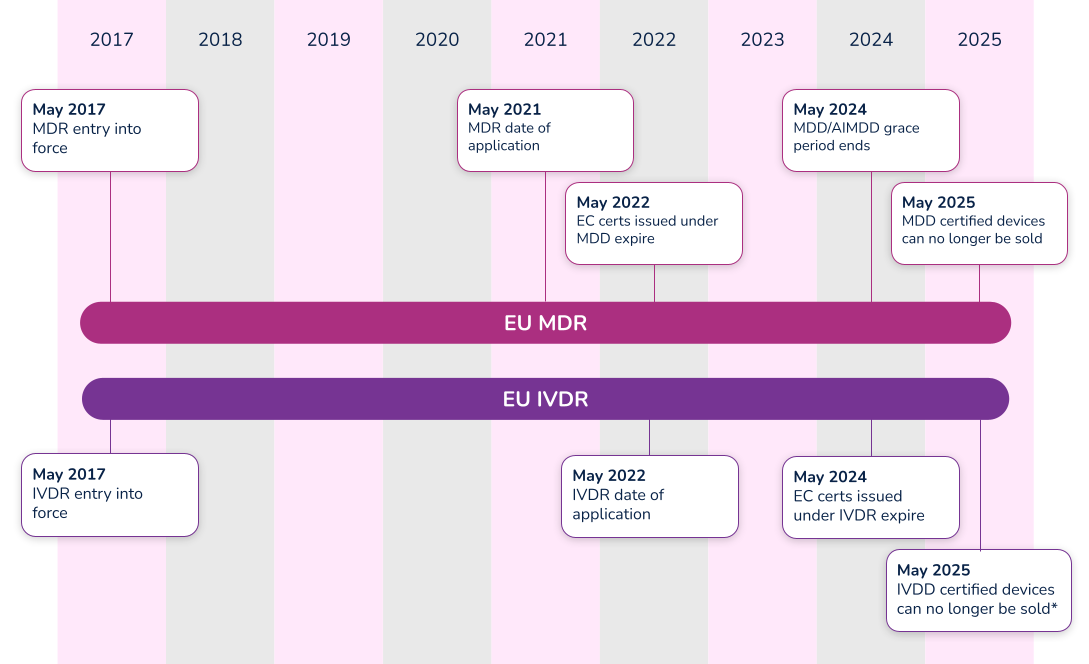
*Note that the timeline for compliance was extended in 2021. Class D (high-risk) devices have until 2025 to comply with IVDR, while Class C devices have until 2026. Class B and Class A sterile devices have until 2027 to comply with IVDR.
What’s the difference between Essential Requirements, General Safety and Performance Requirements (GSPR), and Essential Principles. In order to have a meaningful dialogue, let’s first discuss the three (3) main terms used in the industry.
#1 Essential requirements
The ‘Essential Requirements’ is the backbone for establishing conformity with the Medical Device Directive (MDD 93/42/EEC) and the Active Implantable Medical Device Directive (AIMDD 90/385/EEC). Detailed within Annex I of the MDD and AIMDD, the ‘Essential Requirements’ laid out the requirements that devices must meet in order to state compliance to the directives. With the implementation of the new EU Medical Device Regulation (MDR 2017/745), the ‘Essential Requirements’ will become superseded by the new EU MDR General Safety and Performance Requirements (GSPRs).
#2 Essential principles
The IMDRF laid out Essential Principles requirements in a document entitled Essential Principles of Safety and Performance of Medical Devices and IVD Medical Devices. From a high-level perspective, three basic tenets make up these ‘Essential Principles’:
- A device must be designed to be safe and perform effectively throughout its lifecycle.
- Device manufacturers must maintain all design characteristics.
- Devices must be used in a way that is consistent with how it was designed.
Many countries use the term ‘Essential Principles’ when compiling the documentation required to determine compliance to the law. For instance, the Australian Therapeutic Goods Administration (TGA) uses the term ‘Essential Principles Checklist’. Regardless of the term used, Essential Principles are of similar nature and overlap many of the Essential Requirements and new GSPRs.
#3 General safety and performance requirements (GSPR)
As of May 26, 2021, medical device manufacturers must start to comply with Annex I – General Safety and Performance Requirements (GSPRs) of the new EU Medical Device Regulation (MDR 2017/745). GSPRs are specific to the European MDR and IVDR. If you hear any other term (i.e. Essential Principles), it most likely means it is not referencing the European market.
Annex I of the EU MDR and IVDR details the specific requirements of the General Safety and Performance Requirements (GSPRs). The GSPRs are broken down into three (3) chapters in Annex I, MDR 2017/745 and IVDR 2017/746:
- Chapter 1 - General requirements
- Chapter 2 - Requirements regarding design and manufacture
- Chapter 3 - Requirements regarding the information supplied with the device
Chapter 1 - General requirements
Both the EU MDR and the EU IVDR outline General Safety and Performance Requirements (GSPRs) in great detail for medical device designers and manufacturers. The general requirements for each are almost identical and consist of the following:
- Devices must perform in a way that aligns with the intended design.
- They must not compromise the health or safety of a patient, user, or any other person associated with the device.
- Risks must be reduced as much as possible, but not so much that they negatively affect the risk-benefit ratio.
- Device manufacturers must implement and maintain a thorough, well-documented, and evaluative risk management system that continues to be updated throughout the life cycle of a device.
- Manufacturers and designers must include any necessary measures for protecting users in cases where risks cannot be completely eliminated.
- Manufacturers must provide users with information about any potential risks that remain. This information must be clear, easy to understand, and considerate of the users’ technical knowledge level, use environment, and any applicable medical conditions.
- Devices must withstand the stresses of normal use for the duration of their lifecycle. Devices must be designed, manufactured, and packaged in a way that protects them from damage during transport and storage.
- When it comes to risks and negative side effects that are known and foreseeable, designers and manufacturers must make every effort to minimize negative outcomes. They must also ensure that potential risks are acceptable when compared to the potential benefits of a device to its users.
Chapter 2 - Requirements regarding design and manufacture
The GSPRs also provide key details regarding specific information about the performance, design and manufacture of medical devices. As it relates to design inputs, the MDR and IVDR GSPRs provide highly detailed requirements relating to a device’s technical information. Further detail can be found in the comparison tables in Appendix A and Appendix B, where we have compared MDR to MDD and IVDR to IVDD.
Chapter 3 - Requirements regarding the information supplied with the device
The final key area of governance within the GSPRs relates to specific information a manufacturer must supply with a device. The general requirements for this information states that, “Each device shall be accompanied by the information needed to identify the device and its manufacturer, and by any safety and performance information relevant to the user, or any other person, as appropriate.” The requirements provide further detail as far as location - specific information that must be provided on the following:
- The device label includes its UDI.
- The user instructions.
- The packaging of a device that is intended to maintain its sterile condition.
Medical devices are subject to significant regulations and a full understanding of EU MDR and/or IVDR labeling as defined in Annex 1 Chapter 3.
In addition to the specific requirements identified within Annex I of the EU MDR and IVDR, Annex II, Technical Documentation, identifies additional requirements. Specifically, in both EU MDR and IVDR’s Section 4 – General Safety and Performance Requirements it states:
“the documentation shall contain information for the demonstration of conformity with the general safety and performance requirements set out in Annex I that are applicable to the device taking into account its intended purpose, and shall include a justification, validation and verification of the solutions adopted to meet those requirements. The demonstration of conformity shall include:
(a) the general safety and performance requirements that apply to the device and an explanation as to why others do not apply;
(b) the method or methods used to demonstrate conformity with each applicable general safety and performance requirement;
(c) the harmonised standards, CS or other solutions applied; and
(d) the precise identity of the controlled documents offering evidence of conformity with each harmonised standard, CS or other method applied to demonstrate conformity with the general safety and performance requirements. The information referred to under this point shall incorporate a cross reference to the location of such evidence within the full technical documentation and, if applicable, the summary technical documentation.”
Let’s break this down into each part.
Requirement
(a) the general safety and performance requirements that apply to the device and an explanation as to why others do not apply;
What needs to be documented for the requirements that apply or the requirements that do not apply?
Each and every section of the EU MDR GSPR or EU IVDR should be assessed in its own right as it pertains to your medical device. When a requirement applies, a simple statement may be made that this requirement applies to the device. In practice this is often achieved using a checklist or table, with a column for applicability and a Yes/No answer against each requirement. When a requirement applies, you can move on to the other parts of demonstrating conformity regarding methods used and standards applied.
When a requirement is not applicable, a statement must be made to that effect, i.e. a ‘No’ in the applicability column. Additionally, it must be fully and properly justified. Such a justification may be something like ‘The device is not powered and is therefore not an active device. This requirement does not apply.' The justification should clearly state why the requirement has been deemed not to apply so that your notified body can understand your reasoning
Requirement
(b) the method or methods used to demonstrate conformity with each applicable general safety and performance requirement;
What is meant by “method or methods used”?
This relates to the way you complied with that GSPR requirement, historically it would be listed as a standard or other documentation reference that you have applied to demonstrate compliance, however, the question of ‘method or methods used’ is new to the MDR and it is expected that a verbal description be provided such as:
i. Risk analysis weighed against clinical evaluation benefit
ii. Performance intended demonstrated by design requirements, verification and validation
Requirement
(c) the harmonized standards, common standards (CS) or other solutions applied;
What are harmonized standards, common specifications (CS), and “other solutions”?
Harmonized standards
These are standards that have been specifically developed and assessed for compliance to a regulation or directive. They are published in the Official Journal of the European Union (sometimes just referred to as ‘the OJ’) and if you comply with these standards then there is a ‘presumption of conformity’ with that directive or regulation to which they have been harmonized. These harmonized standards can only be created by a recognized European Standard Organization (such as CEN or CENELEC). When a standard is harmonized, an annex is added that describes how the standard conforms to the directive or regulation. When using harmonized standards, you should make sure that you understand how the standard conforms so that you do not claim compliance when the standard either does not meet that requirement or only partially meets that requirement.
If a standard does not meet a certain requirement of the directive or regulation, or indeed only partially meets it, then you must employ additional mechanisms for compliance. If a harmonized standard meets part of a directive or regulation, then by complying with that standard you also fully meet the corresponding requirement(s) The list of harmonized standards continues to grow - refer to the “Healthcare Engineering” section of the European Commission’s Harmonized Standards page for current information. In this case, using an MDD harmonized standard and documenting a justification for doing so (i.e. how you believe the standard demonstrates compliance with the GSPRs), should provide sufficient evidence
Common specifications
Common Specifications (CS) are a new concept in the MDR. They allow the European Union to add additional requirements that must be met in order to claim compliance where harmonized standards do not exist or where relevant standards are considered insufficient. The definition of a Common Specification is:
‘A set of technical and/or clinical requirements, other than a standard, that provides a means of complying with the legal obligations applicable to a device, process or system.’
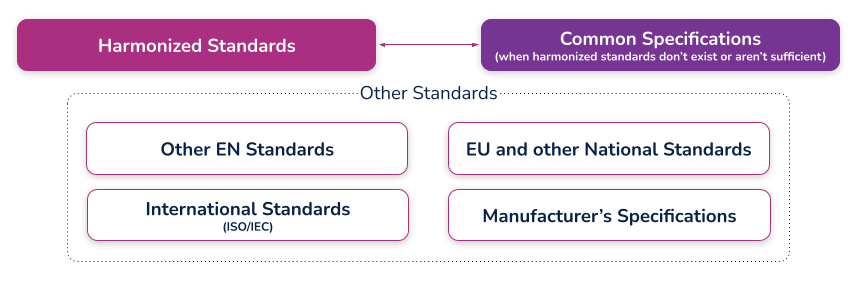
Requirement
(d) the precise identity of the controlled documents offering evidence of conformity with each harmonized standard, CS or other method applied to demonstrate conformity with the general safety and performance requirements. The information referred to under this point shall incorporate a cross- reference to the location of such evidence within the full technical documentation and, if applicable, the summary technical documentation;
What is the expectation for incorporating a "cross-reference to the location of such evidence within the full technical documentation"?
This means that someone looking at the document should be able to identify exactly where in the technical documentation that the compliance evidence can be found. For example, this may refer to test reports and their exact location, or it could even reference locations within a large document, depending on the GSPR and your particular documentation. (i.e. if you have included usability risks as part of a larger risk assessment, you may need to say ‘See Technical File XXX, Section XX, Doc RMF001 rev 3 lines 65-78’). In other cases it could just mean the whole document reference, i.e. Have you done risk management? – then yes, it is RMF001 rev 3. What the specific reference actually is depends on how you have managed your technical documentation and how defined it is (i.e. separate reports or one big one). There should be no ambiguity as to where the document is located
An example of a completed GSPR checklist could look something like this (applicable and nonapplicable examples are shown):
Specification developers and manufacturers must continually maintain their technical documentation to stay compliant. Part of this process is to ensure that they take into account the "generally acknowledged state of the art".
Proactive monitoring
'State of the art'
There is no formal definition of ‘state of the art’ within the EU MDR or IVDR, although it is mentioned many times. ‘State of the art’ is an ongoing debate; however, it generally means that it embodies what is currently and generally accepted as good practice in the medtech industry. The ‘state of the art’ does not necessarily imply the most technologically advanced solution.
One consensus on state of the art is being up to date and compliant with the current and in effect standards that are applicable to your device. This means that if a standard is updated that your medical device is compliant with, you must evaluate that update to ensure that it would meet the EU MDR or EU IVDR ‘state of the art’ requirement. This is not a new requirement from the EU MDD but it is spelled out more clearly in the EU MDR.
The specification developer or manufacturer is ultimately responsible for determining if the updated standard applies or does not apply to their device(s). Either way, the justification should be documented within a gap analysis.
Monitoring for changes
Of course, 'state of the art' only applies if you actually know if something changed. This is why you need to develop a process for monitoring the standards that compliance is claimed. Every single standard that is associated with your technical documentation must be actively monitored, reviewed, and reported on.
If you have a product on the market and need a better way to monitor and maintain your General Safety and Performance Requirements (GSPR) or Essential Principles, Rimsys can help. Rimsys digitizes and automates GSPR and Essential Requirements so you can dynamically update and proactively monitor changing standards and evidence files.
When a standard or evidence file changes, you will automatically be notified and can update one GSPR or all of your GSPRs as applicable with a single click of a button. If additional information is needed, such as testing, it’s also invaluable to ensure that all devices are identified. What used to take weeks of manual, error-prone administrative tasks is now done in seconds within a fully validated, secure, maintenance-free, cloud-based solution
Maintenance
Maintaining and updating your technical documentation is generally the hardest part of staying compliant. Robust processes must be established to ensure nothing slips through the cracks and show up as nonconformances during regulatory audits.
Gap analysis
In addition to meeting the ‘state of the art’ requirements and the continuous proactive monitoring of standards, once a change has been detected that affects the technical documentation, a proper and thorough gap analysis must be completed.
The gap analysis between the old versions and the new versions, or an evaluation of a brand new standard, must occur and be properly documented. The gap analysis should detail what is applicable and what is not applicable, with your supporting justification.
If something within the new or revised standard was applicable to your device, additional engineering testing, documentation, justification, and, in some instances design changes, may be needed to ensure compliance
GSPR updates
Once the gap analysis has been properly documented, specification developers and manufacturers must update their GSPRs.
These updates include finding the withdrawn or superseded standard or evidence file throughout each row within your GSPR table, for every single device on the market on which this change is applicable. This could be one table or dozens of tables depending on the complexity of the products and your product mix.
Without a holistic RIM system to help you, this is an error-prone process as is it tedious, administrative, and extremely easy to miss an inappropriate referenced standard or evidence file.
Extreme diligence on the regulatory or engineering team must occur to ensure these critical updates to the GSPRs are not missed and a gap analysis must be properly referenced throughout. Any justification for including or excluding a new standard or evidence file will be scrutinized by regulatory auditors, and without proper maintenance, may lead to additional review time.
To continue reading this eBook including Comparison Table of the EU MDR Annex I GSPR vs. the EU MDD Annex I Essential Requirements, please register to download the full version.

The beginner's guide to the FDA PMA submission process
This article is an excerpt from The beginner's guide to the FDA PMA submission process ebook.
Table of Contents
- Introduction
- PMA basics
- FDA interactions
- Contents of a traditional PMA submission
- PMA supplements and amendments
- PMA Quality Management System (QMS)
- Review process and timeline
If your organization is planning to market a new medical device in the United States, you first need to determine which regulatory class the device falls under. The vast majority of medical devices regulated by the FDA are either Class I or Class II medical devices, requiring a 510(k) premarket notification or a simple registration if exempt from 510(k) requirements. However, if your device sustains or supports life, is implanted, or presents a “potential unreasonable risk of illness or injury,” your device is likely a Class III device which will require Premarket Approval (PMA) from the FDA before it can be marketed in the United States. Novel devices, for which there are no existing substantially equivalent devices, are automatically classified as Class III as well. Novel devices with a lower risk profile, however, may qualify for the De Novo process instead of the PMA. Just 10% of devices regulated by the FDA are Class III devices.
This ebook provides an overview of the PMA process and its requirements, but it is not designed to be the only resource used in compiling a PMA submission. The FDA provides significant documentation on this process, starting with the regulation governing premarket approval that is located in Title 21 Code of Federal Regulations (CFR) Part 814.
FDA: Background and device oversight
Before we explain what a PMA is, let’s first talk generally about the Food and Drug Administration (FDA) and device oversight. The FDA is the U.S. governmental agency responsible for overseeing medical devices, drugs, food, and tobacco products. When it comes to medical devices, the FDA’s mission is to “protect the public health by ensuring the safety, efficacy, and security of...medical devices.” At the same time, the FDA also has an interest in “advancing public health by helping to speed innovations.” In other words, the FDA’s goal is to make sure devices are safe and effective for public use, while also ensuring that devices have a quick and efficient path to market.
In order to achieve this balance of safety and efficiency, the FDA has three different levels of oversight depending on the risk level of the device: (1) exempt from premarket notification, (2) Premarket Notification, also known as 510(k), and (3) Premarket Approval (PMA).
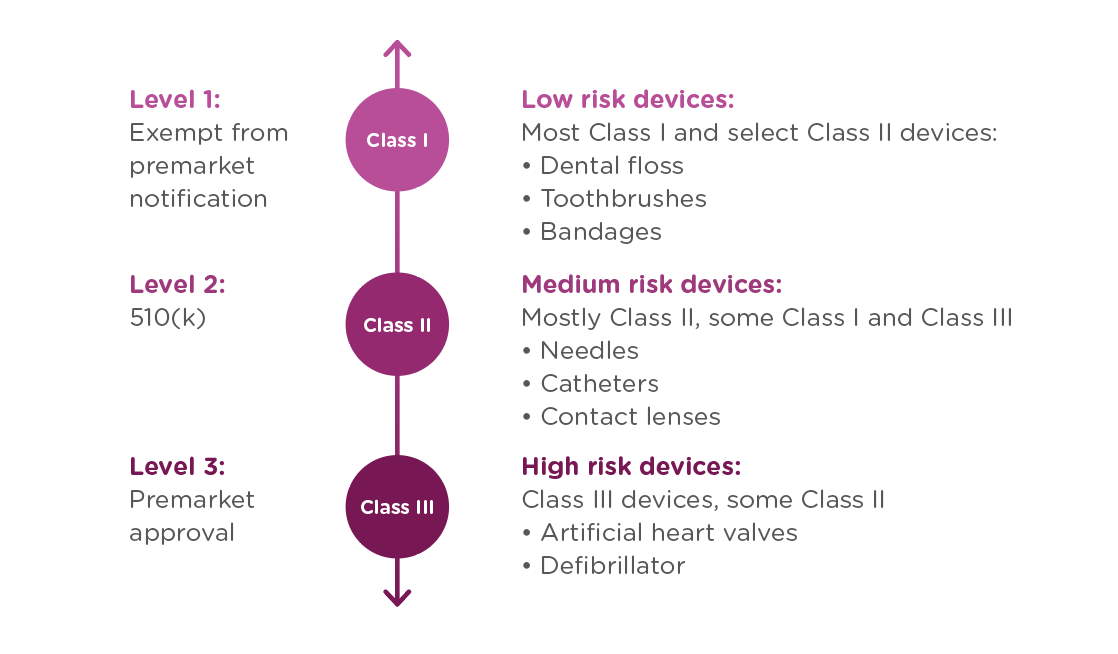
When is a PMA required?
The PMA process is the most stringent regulatory process for medical device approval under the FDA and applies to almost all Class III devices. To determine whether your device requires a PMA, you must first Classify your device by searching the Product Classification Database. The database will provide you with similar devices; their name, classification, and link to the Code of Federal Regulations (CFR) if applicable.
- If a substantial equivalent is found in the Product Classification Database with a submission type of 510(k), you should submit a 510(k), not a PMA.
- If the product classification database identifies your device as Class III and/or requiring a PMA - you should submit a PMA.
- If your device involves a new concept and does not have a classification regulation in the CFR, the database will list only the device type name and product code. In this case, the three-letter product code can be used to search the PMA database and the 510(k).
- If your device cannot be found in the product classification database because it is a new type of device and should be classified as a Class III device because of the level of risk it presents*.
Class III devices support or sustain human life, are of substantial importance in preventing impairment of human health, or present a potential and unreasonable risk of illness or injury.
Note that if your device is a new concept without a substantial equivalent, but does not present the level of risk of a class III device, it may be eligible for the De Novo process as a class I or class II device.
PMA vs 510(k)
Not only are PMA and 510(k) processes applicable to different types of devices, they have different purposes.
510(k): A 510(k) is intended to demonstrate that the device for which approval is being sought is as safe and effective as a currently marketed device that does not require a PMA.
PMA: A PMA is intended to prove that a new device is safe and effective for the end user. A PMA is much more detailed and in-depth than a 510(k). Device manufacturers are typically required to present human clinical trial data, in addition to laboratory testing data.
The difference in complexity between a PMA and 510(k) also affects the time needed to process the submissions. The FDA typically accepts or rejects a 510(k) submission within 30-90 days, at which point the device is posted to the FDA’s 510(k) database. A PMA submission can take up to 180 days to be processed, at which point the FDA can approve or deny the application. The FDA may also issue an “approvable” or “not approvable” letter, which the applicant can choose to respond to, thereby adding time to the submission process.
PMA application methods
There are a number of types of PMA application methods. While most devices which require a PMA will follow the traditional process, be sure to verify that you are using the correct application process to maximize your chances for success and avoid unnecessary delays:
Traditional PMA
The most common method for attaining FDA clearance for Class III devices, the traditional PMA is the appropriate option for most devices that have completed clinical testing.
Modular PMA
The modular PMA is the appropriate application method for devices that have not yet completed clinical testing. Applicants complete individual “modules,” with final confirmation granted once all sections are completed. For additional information on specific requirements of a modular PMA, read the FDA’s Premarket Approval Application Modular Review.
Product Development Protocol
Use the Product Development Protocol (PDP) with medical devices that are based on well-established technology. The PDP process for gaining market approval merges the clinical evaluation and development of information, and involves an agreement between the manufacturer and the FDA. The process provides the advantage of early predictability for the manufacturer and allows early interaction that can identifyFDA concerns as soon as possible in the development process. Because the PDP identifies the agreed upon design and development details, a completed PDP is considered to have an approved PMA. For additional information, read more about the FDA’s PMA Application Methods.
Humanitarian Device Exemption
A Humanitarian Use Device (HUD) is specifically defined as a device intended to benefit patients that are affected by a disease or condition that affects less than 8,000 individuals in the U.S. per year. TheHumanitarian Device Exemption (HDE) approval process is designed to encourage clinical activity around rare conditions, and does have certain restrictions, including:
- After receiving HDE approval, a HUD is eligible to be sold for profit only if the device is intended to address a disease or condition that occurs primarily in pediatric patients, or occurs in pediatric patients in small numbers.
- If an HDE is approved to be sold for profit, the FDA will determine an annual distribution number(ADN). Any devices sold beyond the ADN limit are required to be sold for no profit.
For more information see the FDA’s explanation of the Humanitarian Device Exemption.
CBER Submissions
There are two centers within the FDA responsible for evaluating medical devices. While the majority of devices will go through the Center for Devices and Radiological Health (CDRH), some will be managed by The Center for Biologics Evaluation and Research (CBER). CBER regulates medical devices related to blood and cellular products, including blood collection and processing procedures as well as cellular therapies. This ebook focuses on submissions made through the CDRH, but you can view CBER Regulatory Submissions – Electronic and Paper for more information on the CBER process.
To continue reading this eBook, including a walk through of the different types of required and optional FDA meetings and communications, a detailed list of the contents of a traditional PMA submission, and an overview of quality management system requirements, please register to download the full version.

An overview of 21 CFR Part 11 regulations for medical device companies
What is 21 CFR Part 11?
21 CFR Part 11 refers to the federal regulation that address electronic records and electronic signatures associated with FDA requirements. This single, relatively small, part of the Code of Federal Regulations is extremely significant for companies with FDA-regulated products because it impacts every document signature, electronic file, and FDA submission. Codified in 1997, interpretations of this FDA-issued regulation continue to be debated and re-evaluated as the technology supporting electronic records and signatures changes. In this article, we’ll discuss the regulation and generally accepted interpretations.
Note that discussions and statements in this document are our observations only and should not be taken as fact. You can refer directly to the regulation here.
Part 11: General Provisions
The General Provisions section of 21CFR11 addresses the scope of the regulation, when and how it should be implemented, and defines some of the key terms used. It states that the purpose of Part 11 is to define the criteria under which electronic records, electronic signatures, and handwritten signatures attached to electronic records are equivalent to, and as reliable as, handwritten signatures on paper documents.
Fundamentally, any record that is maintained, used, or submitted under any FDA records regulation is subject to Part 11, and the FDA will accept electronic records in lieu of paper records if an organization can prove that their records and systems meet the Part 11 requirements.
The General Provisions subpart also sets forth a number of definitions, and we’ve listed the ones that are most significant to our discussion here:
- Closed System: A computer system or software whose access is controlled by the same people who are responsible for the information stored in the system. Because the opposite of a closed system, and “open system,” is subject to additional scrutiny be sure that you are able to thoroughly explain and provide documentation for a decision to classify your system as a “closed system.”
- Open System: A computer system or software whose access is not controlled by the same people who are responsible for the information stored in the system.
- Digital Signature: An electronic signature created in a manner that can be verified, ensures the identity of the signer, and maintains the integrity of the document and signature. This often involves the use of cryptography and/or biometric data.
- Electronic Signature: Symbols that represent a legally binding equivalent to an individual’s handwritten signature (as adopted and authorized by the signer).
Part 11: Electronic Records
The Electronic Records section sets forth the requirements for administration of closed and open electronic record-keeping systems, then discusses signature manifestations and requirements for establishing a link between signatures and records.
Part 11 defines a “closed system” as any computer system in which the users controlling access to the system are the same people who are responsible for the data in the system. Today, most systems can be classified as closed systems, but take special care to document control procedures around software that is hosted offsite or classified as a SaaS solution.
This section of the regulation deals with the controls that need to be in place for all applicable electronic record systems by defining:
- Procedures to ensure that all electronic records are authentic, have integrity, and can ensure confidentiality (where that is appropriate).
- Validation requirements for systems that maintain electronic records to ensure that all records are accurate, reliable, and that the system performs consistently according to regulatory requirements.
- Audit trail requirements for all regulated records to ensure a complete history of all changes to records are maintained.
- Controls around system access and document signatures.
Part 11: Electronic Signatures
The Electronic Signatures section defines the components of electronic signatures and the required controls and procedures necessary for using them.
In general, an organization must be able to demonstrate that electronic signatures:
- Are unique to each individual, and that the individual assigned an electronic signature has had their identity and level of authorization verified.
- Must be based either on biometric data (such as fingerprints) or made up of two distinct pieces (ie: a User ID and password)
- Require appropriate controls to ensure that they are verified periodically, cannot be used by someone other than the intended user, and are immediately deactivated if compromised in any way.
Practical application of 21CFR Part 11 for regulatory affairs professionals
21 CFR Part 11 is a critical regulation, and one that can be open to interpretation. Below, we cover some of the key areas that should be of concern for RA professionals. This is an overview of key areas only, and should not be taken as complete instruction or guidance for 21CFR part 11 compliance.
System compliance and validation
Any system that you are using to store electronic records that fall under FDA regulations needs to be compliant with Part 11. This includes everything from spreadsheets to full-featured RIM and document management systems.
Software vendors will often document how their systems are developed to be compliant, and may even support system validation during implementation - but it is ultimately the responsibility of the user organization to ensure that their systems and processes are compliant with Part 11. System validation is the process of documenting that your system meets all of the Part 11 requirements. Software vendors can support this process by ensuring that their systems are built on a highly secured infrastructure that can be demonstrated and proven.
The Rimsys system was built from the ground up to meet the stringent requirements of not only 21 CFR Part 11, but other industry standards and good practices guidelines (GxP). We have put in place a rigorous validation program, built by industry experts and supported by a secure and well-documented infrastructure. For more information, visit the Rimsys Security and Privacy page.
Audit trails
Audit trails are the required system logs that track the who, when, and what of every change made to data that falls under Part 11. Audit trails should be generated and time-stamped by the system, with no ability for users to change that information. Audit trails serve two purposes under 21 CFR Part 11:
- To demonstrate that documented policies and procedures are being followed, including that only users with the appropriate authority are managing data.
- To prove that data retention policies are being adhered to (see below).
At any time, you should be able to view the history of any record, from a Design History File to a submission document, in order to determine what changes have been made, when they were made, and by whom.
Record retention
21 CFR Part 11 specifies that electronic records must be protected and readily available throughout the defined record retention period. Additionally, 21 CFR Part 820 specifies that records related to the quality, manufacturer, regulatory submissions, or any other data that falls under FDA regulation, should be maintained for the life of the medical device and for a minimum of two years from the date of first commercial distribution. This is often referred to as “cradle to grave” tracking.
This means that regulatory professionals need to not only be aware of their company’s record retention policy, but need to ensure that any system being used to track regulatory submissions or other data subject to audit meets Part 11 and Part 820 requirements. Note that record retention requirements apply also to paper records where they are the source document.
Electronic and digital signatures
An important piece of 21 CFR Part 11 is its definition of electronic and digital signatures. “Electronic signature” is used to define any set of symbols that are used in place of a handwritten signature, whereas a “digital signature” is an electronic signature based on methods that ensure the identity of the signer where the integrity of the data can be verified. A digital signature can be based on biometric data (such as fingerprints) or secure user IDs and passwords that are controlled to ensure only one authorized user can use the signature.
As a regulatory affairs professional, you should ensure that:
- Everyone on your team who needs to sign documents has their own unique digital signature and understands the importance of protecting it. Sharing of electronic credentials is a common FDA audit observation. Also ensure that users who are not required to sign documents have appropriate access to data to discourage other users from sharing login credentials with them.
- You are following your company’s policies concerning electronic signature audits so that passwords remain updated and strong and signatures are revoked when a user leaves or changes positions.
- You immediately report any possible loss, theft, or sharing of user credentials or devices that generate identification codes.
While 21 CFR Part 11 is usually considered more of a “quality regulation,” it is important that regulatory teams within medical device organizations fully understand this regulation and its compliance implications. To learn more about the regulations, click below to read our regulatory brief.
.avif)
Rimsys Becomes the Trusted Regulatory Partner for 6 of the Top 12 Global MedTech Manufacturers
“Adoption by half of the top global MedTech manufacturers is a powerful validation that we’re not just a solution, we’re setting the new gold standard for regulatory excellence.”
Pittsburgh, PA - August 7, 2025 - Rimsys, the leading Regulatory Information Management (RIM) software purpose-built for the MedTech industry, today announced a significant milestone: 6 of the world’s top 12 medical device manufacturers now rely on Rimsys to manage and streamline their global regulatory operations.
This milestone further solidifies Rimsys’ position as the trusted partner to the world’s most innovative and quality-focused MedTech companies.
Click here for the full list of the top 12 global MedTech companies.
“Today’s regulatory environment demands more than spreadsheets. Leading manufacturers recognize that regulatory operations are mission-critical, revenue-generating departments and need systems to match that level of importance,” said James Gianoutsos, Founder and CEO of Rimsys.
Rimsys’ unified, enterprise-grade RIM platform centralizes and automates critical regulatory processes—including market registrations, Unique Device Identification (UDI), essential principles/GSPR, and submissions management—reducing compliance risk and accelerating market access. Specifically tailored to the needs of medical device and diagnostics companies, Rimsys enables seamless collaboration across RA, QA, and commercial teams while delivering the audit-ready transparency global regulators demand.
“As more organizations embrace regulatory digital transformation, Rimsys is proud to lead the industry forward,” added Gianoutsos. “Adoption by half of the top global MedTech manufacturers is a powerful validation that we’re not just a solution, we’re setting the new gold standard for regulatory excellence.”
To learn more about the Rimsys, please visit www.rimsys.io.
About Rimsys
Rimsys is the leading provider of Regulatory Information Management (RIM) software purpose-built for MedTech manufacturers. The comprehensive platform digitizes and automates regulatory activities, helping MedTech regulatory affairs teams to efficiently achieve regulatory compliance and get products to market faster. Rimsys is designed around MedTech workflows and supports a full breadth of regulatory functions including registrations, submissions, UDI, EUDAMED compliance, essential principles, and standards management in a unified platform. Rimsys is trusted by half of the world’s top 12 MedTech companies to power their global regulatory operations. For more information, visit www.rimsys.io.
%2520(855%2520x%2520268%2520px).png)
Rimsys Announces Bulk UDI Submission and Rimsys Connect™ to Empower MedTech Regulatory Teams
New solutions deliver enterprise-grade data access and streamlined EUDAMED compliance, driving smarter, faster decisions across the business
Pittsburgh - April 29th, 2025 - Rimsys, the global leader in Regulatory Information Management (RIM) software for the MedTech industry, today announced two major enhancements to its platform: expanded Unique Device Identification (UDI) capabilities to support EUDAMED machine-to-machine (M2M) bulk transmission and Rimsys Connect™, a new enterprise Change Data Capture (CDC) solution that provides near real-time synchronization of Rimsys data with customers’ Business Intelligence (BI) solutions.
Together, these capabilities are designed to help MedTech organizations streamline compliance, reduce manual effort, and unlock the full strategic value of their regulatory data.
New UDI Capabilities Support EUDAMED Readiness
The UDI enhancements extend Rimsys’ industry-leading Universal UDI® framework, enabling MedTech teams to manage complex, global UDI programs in one unified RIM system. Key new capabilities include:
- Approving multiple records simultaneously via a simple, scalable workflow
- EU data governance support with all required attributes for EUDAMED transmission
- Bulk submission of records to both the GUDID and EUDAMED databases
These features allow teams to eliminate time-consuming, record-by-record processing, helping them meet the mandatory January 2026 EUDAMED compliance deadline with confidence.
“We’ve partnered closely with our customers to develop a UDI offering that meets increasing regulatory complexity and is easily scalable as new regulations come online,” said Adam Price, Director of Regulatory and Technical Programs at Rimsys. "We’re not only giving customers the ability to meet EUDAMED compliance but enabling them to manage their global UDI program in a single-sourced RIM solution for complete visibility.”
Introducing Rimsys Connect™: Enterprise Data Access, Redefined
Rimsys Connect™ offers enterprise customers a powerful new way to leverage regulatory data across the business. Built on a scalable, event-driven architecture, it provides secure, structured, near real-time streaming of Rimsys data into any modern data warehouse solution—such as Snowflake, Amazon S3, and Salesforce Bulk API 2.0.
“Rimsys Connect™ is not just a connector—it’s a strategic enabler,” said James Gianoutsos, Founder and CEO of Rimsys. “We’re giving regulatory affairs teams the ability to deliver insights that influence launches, accelerate tender responses, and align compliance with business impact. With Connect, RA teams become true strategic partners.”
By providing full access to customer data—registrations, UDI, projects, tasks, and custom attributes — Rimsys Connect™ supports a wide variety of enterprise use cases with customers’ own business intelligence solutions:
- Tracking on-time submission and decision KPIs
- Aligning registration timelines with product launch dates
- Conducting ROI analysis for renewals and market prioritization
- Accelerating tender readiness by combining RIM and PLM data
- Supporting post-market surveillance dashboards
While the initial release will focus on data access, Rimsys plans to expand Connect with curated BI templates and best practices to further accelerate enterprise customer time-to-value.
Solving the Data Fragmentation Problem for MedTech
Many regulatory affairs teams remain constrained by outdated tools, fragmented data sources, and increasing demands to deliver strategic insights to executive and commercial stakeholders. Rimsys Connect™ addresses these challenges by eliminating manual reporting workflows and enabling teams to analyze their regulatory data alongside financial, marketing, and quality systems.
“With Rimsys Connect™, regulatory teams can visualize and analyze their data in real time, assess launch readiness, and deliver more value to their organizations. This is how RA becomes a catalyst for better decisions—not just compliance,” said Gianoutsos.
Both Rimsys’ expanded UDI capabilities and Rimsys Connect™ will be available this summer. Those interested in learning more about these solutions and how they will enable greater automation, efficiency, and compliance can visit our booth at RAPS Euro Convergence May 13-15 in Brussels, Belgium, or sign up for Rimsys’ product update webinar on Thursday, May 22nd at 10 AM ET.
Read the press release here.
About Rimsys
Rimsys is improving global health by accelerating delivery and increasing availability of life-changing medical technologies. Rimsys Regulatory Information Management (RIM) software digitizes and automates regulatory activities, helping MedTech regulatory affairs teams to plan more effectively, execute more quickly, and confidently ensure global regulatory compliance. Rimsys is designed around MedTech workflows and supports a full breadth of regulatory activities including registrations, submissions, UDI, essential principles, and standards management in a unified platform. For more information, visit www.rimsys.io.
Contacts:
marketing@rimsys.io
.avif)
Rimsys Enters Strategic Alliance Relationship with KPMG
PITTSBURGH – March 11, 2025 -- Rimsys, the global leader of MedTech Regulatory Information Management (RIM) software, today announced that it has entered into a strategic alliance relationship with KPMG to advance digital transformation in the MedTech industry.
“KPMG’s deep experience in advisory and business transformation services and exceptional reputation make them a valuable alliance relationship for us,” said James Gianoutsos, Founder and CEO of Rimsys. “KPMG is on the cutting edge of industry trends and has a wide breadth of experience in helping companies innovate and scale. We are thrilled to work with them to help MedTech teams transform their regulatory management processes and leverage the benefits of automation and digitization as part of their broader transformation strategy.”
Founded for and by MedTech regulatory affairs professionals in 2017, Rimsys was created to bring efficiency to regulatory information management and fill an inhibitive technology gap in an underserved industry. Rimsys has since grown to support the world’s MedTech leaders backed by a staff that understands their complex workflows and a robust, secure technology infrastructure that allows customers to scale Rimsys software to support their changing regulatory needs and requirements.
“There is tremendous innovation happening in the MedTech industry, and we are excited to work with Rimsys to help clients transform how they manage regulatory information for getting new products to market and sustaining their existing product portfolios. It’s critical to approach these programs as a holistic business transformation across people, process, technology, data, and governance & controls,” said Dipan Karumsi, Principal, Consulting Sector Leader for Life Sciences at KPMG.
“Through our strategic alliance with KPMG, we can further expand our reach to large and enterprise MedTech companies and continue our exponential growth,” said James. “Combined with KPMG’s experience helping organizations mature their data collection and transformation processes to reach RIM readiness, we can enable the MedTech industry to innovate faster, strengthen compliance, and most importantly, improve the availability of life-changing medical technologies.”
See the full press release here.

FDA’s Final Rule on LDTs: What manufacturers need to know
In July 2024, the FDA's final rule in 21 CFR Part 809 on laboratory developed tests (LDTs) went into effect, amending its previous regulations to make it clear that IVDs, including those that are manufactured in laboratories, are classified as devices under the Federal Food, Drug, and Cosmetic Act. Our blog post provides an overview of LDTs, FDA’s final rule, the phase out policy schedule, and how LDT manufacturers can prepare themselves for compliance.
What are LDTs?
Simply put, LDTs are IVDs that are designed, manufactured, and utilized within a certified laboratory and are typically used for high-complexity testing.
Historically, FDA has used enforcement discretion only on LDTs, which means that most LDTs haven’t been subjected to specific regulatory requirements. However, the volume of and risks associated with LDTs have grown over the years. Some examples of modern LDTs include glucose tests, genetic tests for cancer and infectious diseases, and newborn screenings for early diagnostics. Without a regulatory framework in place, patients are at greater risk of receiving inaccurate test results, forgoing necessary or undergoing unnecessary treatment, and adhering to misleading or false product claims, possibly endangering patients and leading to higher healthcare costs.
FDA’s Final Rule on LDTs
Following the final rule that was issued, LDTs are now subject to the same regulatory requirements as other IVDs, including premarket reviews, quality system requirements, labeling requirements, adverse event reporting, and device listing and registration. To prevent disruptions in patient care, there is a four-year transition or phaseout period consisting of the following five stages:
- Stage 1 (May 6, 2025): LDT manufacturers will be expected to comply with FDA medical device reporting (MDR) requirements, correction and removal reporting requirements, and quality system (QS) requirements for complaint files.
- Stage 2 (May 6, 2026): LDT manufacturers will be required to comply with IVD registration and listing requirements, labeling requirements, and investigational use requirements.
- Stage 3 (May 6, 2027): LDT manufacturers will need to comply with all other QS requirements not covered in Stage 1.
- Stage 4 (November 6, 2027): Unless a premarket submission is received before the start of this stage, LDT manufacturers of high-risk products will need to comply with premarket review requirements for IVDs that may be classified into class III or that meet the requirements of section 351 of the Public Health Service Act.
- Stage 5 (May 6, 2028): LDT manufacturers of moderate and low –risk products will need to comply with premarket review requirements for IVDs unless a submission is received before the beginning of this stage.
Manufacturers of LDTs that don’t meet the requirements in each stage are deemed non-compliant to the regulations governing IVDs and may be subject to FDA 483 observations or warning letters, financial penalties, and even worse, involuntary removal of products from the market.
Note that some LDTs will be exempt from these requirements. Refer to the FDA’s website for more guidance.
Preparing for Compliance
Despite a four-year phaseout period, it’s crucial for LDT manufacturers to start assembling a compliance plan. Starting as early as May 2025, manufacturers will be required to comply with FDA Medical Device Reporting (MDR) requirements, correction and removal reporting requirements, and quality system requirements for complaint files.
It's good practice to conduct an internal regulatory assessment to ensure you have the resources, processes, and tools in place to successfully meet new requirements for LDT devices. It’s also essential to make sure your team is well-versed in these new requirements and the documentation and timelines involved. Including all relevant stakeholders early on, getting a comprehensive project plan in place, and meeting regularly to ensure all tasks are completed would be helpful during the phaseout period and beyond.
Regarding the LDT changes, the FDA has provided a Q&A sheet that you may find helpful.
If you're looking for guidance on FDA premarket submissions, see our Beginner’s Guides to the FDA 510(k), De Novo, and PMA processes.
How Regulatory Tools Can Help
FDA’s final rule on LDTs will add complexity to the regulatory information management of laboratory diagnostic tests. There are digital solutions that can help manufacturers stay current on updated regulations and manage the additional information and documentation needed because of these updates.
A regulatory intelligence database like Rimsys Intel can provide detailed global market entrance requirements, application timelines, fees, risk class specifications, and documentation needed for medical devices and IVDs so that manufacturers can start preparing their premarket strategies.
Regulatory Information Management (RIM) software like Rimsys can help boost efficiency, reduce compliance risk, and increase collaboration by centralizing regulatory information and automating time-consuming, manual processes. As a result, medical device manufacturers gain complete visibility into their submission management and selling status so that they can plan more effectively, avoid costly product delays, and execute faster.

Quick reference guide - global medical device UDI requirements and timelines
This article was last updated on February 10, 2025.
What is UDI?
UDI systems are intended to benefit healthcare providers, manufacturers, authorized health authorities, hospitals and institutions, and individual consumers by providing:
- Faster discovery of possible flawed medical device information by health authorities.
- Quicker access to recall information, and visibility into current inventory.
- A reduction in medical errors through consistently documented product expiration dates.
- Identification of any counterfeit products being used in healthcare facilities.
- Assurances that information regarding an implanted device is safely retained and traceable.
UDI timelines and deadlines vary by market, classification risk, and product and have been revised multiple times in some countries*. This article details the UDI deadlines for the countries which have announced specific programs (draft or implemented) and is current as of the date of this article.
*Note: these dates can change as participating countries adjust their plans. We do our best to update this as more information becomes available.
Quick Links to country-specific sections:
- Australia UDI
- Brazil UDI
- Canada UDI
- China UDI
- European Union UDI
- India UDI
- Japan UDI
- Saudi Arabia UDI
- Singapore UDI
- South Korea UDI
- Taiwan UDI
- United States UDI
- UDI databases by country
General UDI labeling requirements
There are two components to a medical device UDI: the UDI device identifier (UDI-DI) and the UDI production identifier (UDI-PI). The UDI is presented as a barcode label (human and machine readable) on device packaging or on the device itself and acts as the access key to all device UDI attributes.
UDI-DI: This is the static portion of the UDI which identifies the manufacturer along with the specific device version. The UDI-DI (device identifier), also known as the Global Trade Item Number (GTIN) is assigned by an approved organization, such as GS1, and contains:
- Company prefix
- Manufacturers internal product code
- Check character
The UDI-DI is the primary identifier to be used in looking up device attributes in country-specific databases and is assigned prior to placing a product on the market. Note that the device identifier is different for different packaging levels of the same device.
UDI-PI: This is the dynamic portion of the UDI which is assigned by the manufacturer and identifies one or more of the following:
- Manufacturer’s lot or batch number
- Serial number
- Manufacturing date
- Expiration date
- Other attributes as defined by country-specific regulations
The UDI-PI actual values do not appear in country-specific databases (with the exception of the EU vigilance database).
Australian UDIGuidelines
Reporting Database: AusUDID (pre-production)
The Australian government for medical devices, the TGA, has not launched any official regulations or timeline for mandatory UDI labeling. They do provide a wealth of information on their website that is worth reviewing. In the meantime, however, they are hoping for a Q1 2025 implementation. The AusUDID Pre-Production environment is available for sponsors and manufacturers of medical devices supplied in Australia. It is a test environment that allows testing of data submission, prior to submission to the AusUDID Production environment. Any sponsor or manufacturer with an active TBS account can access the database.
ANVISA UDI guidelines
Reporting database:TBD
RDC No. 591/2021 is the regulations guideline for the identification of medical devices regulated by ANVISA, implementing the Unique Identification of Medical Devices (UDI) system. In July 2024, ANVISA finalized amendment RDC No. 884/2024 which implemented various adjustments to RDC 591/2021. The biggest take-away regarding UDI is the extension of one year on the implementation deadlines.
Health Canada website
Reporting Database: N/A
Position paper on the current state of UDI implementation
Medtech Canada strongly supports the global initiative led by regulators under the guidance of the International Medical Devices Regulators Forum (IMDRF), which aims to standardize the identification of medical devices by requiring that certain medical devices carry an internationally recognized UDI. Currently, there is no process in place for UDI in Canada.
China (NMPA) website
Reporting Database: China National UDI Database
Announcement No 22 of 2023
On January 1, 2021, the NMPA implemented the UDI system for its first batch of medical devices, including 69 Class III devices. The following year, June 1, 2022, followed the implementation for the second batch of other Class III medical devices (including IVD reagents). Then in 2023, Order No. 22 announced the third batch of products to adopt the UDI system.
As of June 1, 2024, medical devices listed in the third batch implementation product catalog must have already had UDI implemented. According to the degree of risk and regulatory needs, some Class II medical devices in the third batch included high-demand single-use products, items selected for centralized procurement, and medical aesthetic products, totaling 103 types in 15 categories.
European Union UDI Information
Reporting Database: EUDAMED
Rimsys Updated EUDAMED Timeline Blog Post
The UDI & Devices module is expected to be declared fully functional by the end of Q2 2025 and mandatory for industry use on January 1, 2026. The EU continues to strongly recommend to the industry to establish its solution and to submit data on a voluntary basis.
Medical Devices Rules, 2017
Legal Metrology Act, 2009
Reporting Database: N/A
Rule 46 of Medical Device Rule 2017 was set to require UDI labeling by January 1, 2022. However, details on how the UDI needs to be implemented have not yet been released but India's labeling and traceability requirements must be met as per CDSCO regulations.
In addition to the Medical Device Rule 2017, the Legal Metrology Act, 2009 focuses on standardizing weights and measures and ensures that packaged commodities, including medical devices, are labeled with accurate and clear information.
Law to Ensure Quality, Efficacy and Safety of Pharmaceuticals, Medical Device, and Similar Products
Reporting Database: N/A
There are two regulatory authorities responsible for regulation of medical devices in Japan: The Ministry of Health, Labour and Welfare (MHLW) and the Pharmaceuticals and Medical Devices Agency (PMDA). The MHLW is responsible for the administrative actions such as guidance and approval, and judgment on whether or not a product is considered a medical device. The PMDA undertakes product review and post-market safety measures.
As of Dec 2022, bar code labeling based on international standards is required for immediate containers/wrappings/retail packages of medical devices. It is expected for barcodes to be displayed on every medical device in unit of use for patients. Japan was an early promoter of standardized barcodes and is still working towards harmonizing the requirements with global UDI expectations.
The Pharmaceuticals and Medical Devices Act (PMD Act) translates in Japanese meaning "Law to Ensure Quality, Efficacy and Safety of Pharmaceuticals, Medical Devices, and Similar Products," but is often shortened to Act on Pharmaceuticals and Medical Devices or just PMD Act.
Requirements for Unique Device Identification (UDI for Medical Devices)
Reporting Database: Saudi-DI
The SFDA requires compliance with the Unique Device Identification (UDI) regulations on all medical device companies in Saudi Arabia for all classifications. Medical device classifications include: devices, IVD, non-medical IVD, chemical for medical use, distillation device, general lab use, HCT/Ps product and radiation devices.
Guidance for UDI Implementation
Reporting Databases: Singapore Medical Device Register (SMDR) - For risk class B or higher, Class A Medical Device Database - Risk class A only
Singapore is now requiring compliance with UDI labeling and database registration. They will accept UDI labels for devices already marketed in the U.S. and the EU without any need for modification. However, if they are not marketed in either country, then they are required to implement via Singapore UDI regulations.
Companies are given an additional 6 months from the compliance date to deplete the respective medical devices that have been imported prior to the compliance date and exist in their current supply chain.
Note:
• UDIs will not be required for medical devices for clinical research, investigational testing or clinical trial and custom-made medical devices
• Medical devices authorized for supply via Special Access Routes (GN26, GN27, GN29) are required to comply with UDI requirement on a risk-calibrated approach
Act on In Vitro Diagnostic Medical Devices
Act on Medical Devices
Reporting Database: South Korean Integrated Medical Device Information System (IMDIS)
South Korea has already implemented UDI regulations by Article 20-23 of the Medical Device Act (No. 14330) and Article 54-2 of Enforcement Regulations of Medical Device Act (No. 1512).
Guidance document from Taiwan FDA
Reporting Database: TUDID
Taiwan has previously implemented UDI regulations, which include labeling and database reporting requirements.
FDA website for UDI
Reporting database: GUDID database
The United States has previously implemented UDI regulations, which includes labeling and database reporting requirements.
Each country has their own UDI database and varying requirements for the data stored in those databases. There is overlap in the data required among the various UDI databases, but each country also has unique data they require.
In addition, countries require that UDI-DI information be provided by “issuing entities.” Note that with the exception of China, all countries accept GS1, HIBCC, and ICCBA as issuing entities.
* Data attributes are approximations based on country UDI requirements and include mandatory, optional, mandatory if applicable, and country database auto generated elements.
** Expected to be similar to US GUDID requirements.
Keeping pace with UDI regulations
Keeping track of country-specific UDI requirements, implementation timelines, and affected devices can be a big challenge to RA teams—especially because the information is scattered across many sources and simply hard to find. In this guide, we have consolidated timeline information and device class requirements across multiple countries. While we make every effort to provide accurate and up to date information, it's always advised to check the government website for the country in question.
Additional UDI resources
Looking for more information? You can visit our EUDAMED resource center, where you will find videos and resources to help you plan for UDI requirements in Europe. In addition, you may enjoy our blog post that outlines our views on the recent EUDAMED timeline updates.
For a broader introduction to UDI, see our Rimsys UDI Overview blog post.
If you're looking for an automated, integrated solution to help you meet changing regulations and manage your global UDI program, request a custom Rimsys demo!
%2520(1755%2520x%2520550%2520px)%2520(855%2520x%2520268%2520px).avif)
Why a RIM System is Critical to Successfully Support MedTech M&A Activities
There was significant M&A activity in the MedTech sector in 2024, and the industry is predicting another big year for mergers and acquisitions. As MedTech companies aim to expand their product lines, enter new markets, innovate faster, and remain competitive in a rapidly evolving space, mergers and acquisitions can be attractive and cost-efficient options. Additionally, some manufacturers are choosing to divest parts of their business to hone their focus, drive additional investment in other key areas, and optimize operations.
For the regulatory affairs professional, M&A activities can be anything but efficient. An influx of new products, registrations, and regulatory information to maintain can wreak havoc on RA teams who are already struggling to effectively manage and maintain compliance amid a seemingly constant state of regulatory change.
In an increasingly complex regulatory landscape, many MedTech teams are turning to RIM systems to help them centralize regulatory information across the business, automate time-consuming, manual processes, and strengthen global compliance. These benefits are even more palpable for companies undergoing mergers, acquisitions, and divestitures, giving them streamlined, fully visible regulatory information management that can scale with their evolving business needs and additional regulatory information to manage.
Case Study: Large, Publicly Traded Device Manufacturer Navigates Product Line Divestiture with Ease
A large, publicly traded manufacturer of products for pain management, digestive health, and IV therapy was in the process of divesting one of its product lines to another company. As a result, some of their regulatory employees were transitioned to the company that purchased its product line. The customer leveraged Rimsys’ unique Linked Accounts feature, which allows users to grant external stakeholders controlled access to Rimsys, to give those impacted access to the 100+ registrations associated with the divested products. As a result, the transitioned employees lost no access to their respective products and are able to manage, review, and approve those registrations as they normally would.
“Linked Accounts is a fantastic feature that I didn’t originally appreciate as much. Rimsys made it easy for us to identify the products impacted by the divestiture and provide access to those who need them. It has been a bright spot in the sea of headaches both teams are experiencing when trying to review and approve information in other systems.”
-Program Manager, Regulatory Information Management
When the transaction is complete, the customer will easily be able to export the list of registrations by product tag and archive the registrations in Rimsys for easy management and visibility. The transitioned employees will also still retain access to the information they need in Rimsys as they work to implement their own Rimsys solution to manage those registrations.
Navigating Business and Regulatory Changes with RIM Systems
As the MedTech industry prepares for additional mergers, acquisitions, and divestitures this year, getting a solid regulatory information foundation in place is critical for a successful transition and ongoing compliance. Yet, many MedTech RA teams are using manual processes and siloed systems to manage regulatory information.
One of Rimsys’ goals is to serve as a strategic partner to MedTech RA teams, helping them better understand their current RIM state and the steps they need to advance their processes. This includes the implementation of a RIM system such as Rimsys to centralize their regulatory information, enable easy collaboration with internal and external stakeholders, and automate time-consuming manual processes for strengthened global compliance.
See our Guide to MedTech RIM Maturity, which provides our RIM Maturity Model Framework, for ways to better assess your organization’s current RIM state and incrementally reach new milestones.
Rimsys has helped global MedTech leaders navigate business and regulatory change with unified RIM software that provides full visibility into their regulatory activities. If you’re looking to stay ahead of upcoming strategic activities or are simply looking for a better way to manage your information amid increasing complexity, contact us to learn how Rimsys can help you streamline and automate your processes for long-term success.




